The title compound, UCu
0.60(4)Sb
2, crystallizes in the tetragonal space group
P4/
nmm. The U atom and one independent Sb atom have 4
mm site symmetry, whereas the Cu atom and the other Sb atom have
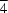
2
m site symmetry. Zigzag USb sheets stack perpendicular to the
c axis. These sheets are separated by square-planar nets of Sb atoms and Cu atoms. The length of the
a axis for UCu
xSb
2 is invariant to
x, whereas there is a linear relationship between Cu occupancy and the length of the
c axis (following Vegard's Law) that holds for
x between 0 and 1. This is explained in terms of the crystal structure.
Supporting information
USb2 was obtained from the reaction of U powder [0.63 mmol, ORNL, prepared
from turnings as described elsewhere (Haneveld & Jellinek, 1969)] and
Sb (1.26 mmol, Aldrich, 99.5%) in a carbon-coated fused-silica tube. The tube was
heated to 1273 K in 20 h, held there for 111 h, cooled to 773 K in 20 h, and
then air-cooled to 293 K. Single crystals of uranium copper antimonide,
UCu0.60 (4)Sb2, were obtained from the reaction of USb2 (0.093 mmol) and
CuO (0.093 mmol, Aldrich, 99.99%) in a carbon-coated fused-silica tube. The
tube was heated to 773 K in 10 h, further heated to 973 K in 60 h, held there
for 70 h, heated to 1173 K in 20 h, held there for 180 h, cooled to 773 K over
a period of 100 h, and finally cooled to 293 K in 60 h. The product consisted
mainly of UO2 powder and elemental Sb, among which a few small black plates
were found. Energy-dispersive X-ray analysis showed the presence of U, Cu, and
Sb in these plates.
A numerical face-indexed absorption correction was performed using the program
SADABS (Bruker, 2003). A correction was made for extinction. All
atoms
were given anisotropic displacement parameters. Data sets on the same crystal
were collected at 100, 120, 200, and 298 K. The corresponding refined Cu
occupancies were 0.634 (12), 0.600 (13), 0.608 (13) and 0.586 (12). We choose to
report its value as 0.60 (4) and we report only the structure determinations at
100 and 120 K here. The structure was standardized by means of the program
STRUCTURETIDY (Gelato & Parthé, 1987). The final difference
electron
densities show no significant residual peaks. The highest peaks are 1.24 and
0.07 Å from U, and the deepest holes are 1.29 and 0.89 Å from U, at 100
and 120 K, respectively.
For both compounds, data collection: SMART (Bruker, 2003); cell refinement: SAINT (Bruker, 2003); data reduction: SAINT (Bruker, 2003); program(s) used to solve structure: SHELXS97 (Sheldrick, 2008); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008); molecular graphics: CrystalMaker (Palmer, 2008); software used to prepare material for publication: SHELXTL (Sheldrick, 2008).
(I_100K) uranium copper diantimonide
top
Crystal data top
| UCu0.60(4)Sb2 | Dx = 9.943 Mg m−3 |
| Mr = 521.84 | Mo Kα radiation, λ = 0.71073 Å |
| Tetragonal, P4/nmm | Cell parameters from 1655 reflections |
| Hall symbol: -P 4a 2a | θ = 4.4–28.5° |
| a = 4.320 (4) Å | µ = 65.20 mm−1 |
| c = 9.341 (9) Å | T = 100 K |
| V = 174.3 (3) Å3 | Plate, black |
| Z = 2 | 0.14 × 0.08 × 0.05 mm |
| F(000) = 424.80 | |
Data collection top
Bruker SMART 1000 CCD
diffractometer | 155 independent reflections |
| Radiation source: fine-focus sealed tube | 155 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.075 |
| ω scans | θmax = 28.1°, θmin = 2.2° |
Absorption correction: numerical
face-indexed (SADABS; Bruker, 2003) | h = −5→5 |
| Tmin = 0.020, Tmax = 0.086 | k = −5→5 |
| 1951 measured reflections | l = −12→12 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.024 | w = 1/[σ2(Fo2) + (0.0306P)2 + 1.6422P]
where P = (Fo2 + 2Fc2)/3 |
| wR(F2) = 0.058 | (Δ/σ)max < 0.001 |
| S = 1.20 | Δρmax = 1.32 e Å−3 |
| 155 reflections | Δρmin = −3.32 e Å−3 |
| 13 parameters | Extinction correction: SHELXL97 (Sheldrick, 2008), Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| 0 restraints | Extinction coefficient: 0.0174 (16) |
Crystal data top
| UCu0.60(4)Sb2 | Z = 2 |
| Mr = 521.84 | Mo Kα radiation |
| Tetragonal, P4/nmm | µ = 65.20 mm−1 |
| a = 4.320 (4) Å | T = 100 K |
| c = 9.341 (9) Å | 0.14 × 0.08 × 0.05 mm |
| V = 174.3 (3) Å3 | |
Data collection top
Bruker SMART 1000 CCD
diffractometer | 155 independent reflections |
Absorption correction: numerical
face-indexed (SADABS; Bruker, 2003) | 155 reflections with I > 2σ(I) |
| Tmin = 0.020, Tmax = 0.086 | Rint = 0.075 |
| 1951 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.024 | 13 parameters |
| wR(F2) = 0.058 | 0 restraints |
| S = 1.20 | Δρmax = 1.32 e Å−3 |
| 155 reflections | Δρmin = −3.32 e Å−3 |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| U1 | 0.2500 | 0.2500 | 0.25716 (6) | 0.0067 (3) | |
| Sb1 | 0.2500 | 0.2500 | 0.65348 (12) | 0.0087 (4) | |
| Sb2 | 0.7500 | 0.2500 | 0.0000 | 0.0062 (4) | |
| Cu1 | 0.7500 | 0.2500 | 0.5000 | 0.0080 (13) | 0.634 (12) |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| U1 | 0.0045 (4) | 0.0045 (4) | 0.0113 (5) | 0.000 | 0.000 | 0.000 |
| Sb1 | 0.0054 (4) | 0.0054 (4) | 0.0153 (7) | 0.000 | 0.000 | 0.000 |
| Sb2 | 0.0045 (5) | 0.0045 (5) | 0.0097 (7) | 0.000 | 0.000 | 0.000 |
| Cu1 | 0.0095 (15) | 0.0095 (15) | 0.0051 (18) | 0.000 | 0.000 | 0.000 |
Geometric parameters (Å, º) top
| U1—Cu1i | 3.132 (2) | Sb1—U1ii | 3.167 (3) |
| U1—Cu1 | 3.132 (2) | Sb2—Sb2viii | 3.055 (3) |
| U1—Cu1ii | 3.132 (2) | Sb2—Sb2vi | 3.055 (3) |
| U1—Cu1iii | 3.132 (2) | Sb2—Sb2vii | 3.055 (3) |
| U1—Sb1i | 3.167 (3) | Sb2—Sb2ix | 3.055 (3) |
| U1—Sb1iv | 3.167 (3) | Sb2—U1vi | 3.231 (2) |
| U1—Sb1v | 3.167 (3) | Sb2—U1x | 3.231 (2) |
| U1—Sb1ii | 3.167 (3) | Sb2—U1vii | 3.231 (2) |
| U1—Sb2iii | 3.231 (2) | Cu1—Sb1ii | 2.593 (2) |
| U1—Sb2vi | 3.231 (2) | Cu1—Sb1x | 2.593 (2) |
| U1—Sb2 | 3.231 (2) | Cu1—Sb1i | 2.593 (2) |
| U1—Sb2vii | 3.231 (2) | Cu1—Cu1xi | 3.055 (3) |
| Sb1—Cu1ii | 2.593 (2) | Cu1—Cu1ii | 3.055 (3) |
| Sb1—Cu1iii | 2.593 (2) | Cu1—Cu1i | 3.055 (3) |
| Sb1—Cu1i | 2.593 (2) | Cu1—Cu1xii | 3.055 (3) |
| Sb1—Cu1 | 2.593 (2) | Cu1—U1i | 3.132 (2) |
| Sb1—U1i | 3.167 (3) | Cu1—U1ii | 3.132 (2) |
| Sb1—U1iv | 3.167 (3) | Cu1—U1x | 3.132 (2) |
| Sb1—U1v | 3.167 (3) | | |
| | | |
| Cu1i—U1—Cu1 | 58.37 (5) | U1i—Sb1—U1 | 105.28 (3) |
| Cu1i—U1—Cu1ii | 87.20 (8) | U1iv—Sb1—U1 | 105.28 (3) |
| Cu1—U1—Cu1ii | 58.37 (5) | U1v—Sb1—U1 | 105.28 (3) |
| Cu1i—U1—Cu1iii | 58.37 (5) | U1ii—Sb1—U1 | 105.28 (3) |
| Cu1—U1—Cu1iii | 87.20 (8) | Sb2viii—Sb2—Sb2vi | 180.0 |
| Cu1ii—U1—Cu1iii | 58.37 (5) | Sb2viii—Sb2—Sb2vii | 90.0 |
| Cu1i—U1—Sb1i | 48.603 (15) | Sb2vi—Sb2—Sb2vii | 90.0 |
| Cu1—U1—Sb1i | 48.603 (15) | Sb2viii—Sb2—Sb2ix | 90.0 |
| Cu1ii—U1—Sb1i | 106.23 (5) | Sb2vi—Sb2—Sb2ix | 90.0 |
| Cu1iii—U1—Sb1i | 106.23 (5) | Sb2vii—Sb2—Sb2ix | 180.0 |
| Cu1i—U1—Sb1iv | 106.23 (5) | Sb2viii—Sb2—U1vi | 118.22 (2) |
| Cu1—U1—Sb1iv | 106.23 (5) | Sb2vi—Sb2—U1vi | 61.78 (2) |
| Cu1ii—U1—Sb1iv | 48.603 (15) | Sb2vii—Sb2—U1vi | 118.22 (2) |
| Cu1iii—U1—Sb1iv | 48.603 (15) | Sb2ix—Sb2—U1vi | 61.78 (2) |
| Sb1i—U1—Sb1iv | 149.44 (6) | Sb2viii—Sb2—U1x | 61.78 (2) |
| Cu1i—U1—Sb1v | 48.603 (15) | Sb2vi—Sb2—U1x | 118.22 (2) |
| Cu1—U1—Sb1v | 106.23 (5) | Sb2vii—Sb2—U1x | 118.22 (2) |
| Cu1ii—U1—Sb1v | 106.23 (5) | Sb2ix—Sb2—U1x | 61.78 (2) |
| Cu1iii—U1—Sb1v | 48.603 (15) | U1vi—Sb2—U1x | 123.56 (5) |
| Sb1i—U1—Sb1v | 86.016 (15) | Sb2viii—Sb2—U1 | 118.22 (2) |
| Sb1iv—U1—Sb1v | 86.016 (15) | Sb2vi—Sb2—U1 | 61.78 (2) |
| Cu1i—U1—Sb1ii | 106.23 (5) | Sb2vii—Sb2—U1 | 61.78 (2) |
| Cu1—U1—Sb1ii | 48.603 (15) | Sb2ix—Sb2—U1 | 118.22 (2) |
| Cu1ii—U1—Sb1ii | 48.603 (15) | U1vi—Sb2—U1 | 123.56 (5) |
| Cu1iii—U1—Sb1ii | 106.23 (5) | U1x—Sb2—U1 | 83.93 (8) |
| Sb1i—U1—Sb1ii | 86.016 (15) | Sb2viii—Sb2—U1vii | 61.78 (2) |
| Sb1iv—U1—Sb1ii | 86.016 (15) | Sb2vi—Sb2—U1vii | 118.22 (2) |
| Sb1v—U1—Sb1ii | 149.44 (6) | Sb2vii—Sb2—U1vii | 61.78 (2) |
| Cu1i—U1—Sb2iii | 122.58 (5) | Sb2ix—Sb2—U1vii | 118.22 (2) |
| Cu1—U1—Sb2iii | 178.364 (13) | U1vi—Sb2—U1vii | 83.93 (8) |
| Cu1ii—U1—Sb2iii | 122.58 (5) | U1x—Sb2—U1vii | 123.56 (5) |
| Cu1iii—U1—Sb2iii | 94.43 (8) | U1—Sb2—U1vii | 123.56 (5) |
| Sb1i—U1—Sb2iii | 130.699 (18) | Sb1ii—Cu1—Sb1x | 107.80 (4) |
| Sb1iv—U1—Sb2iii | 74.92 (5) | Sb1ii—Cu1—Sb1i | 112.86 (8) |
| Sb1v—U1—Sb2iii | 74.92 (5) | Sb1x—Cu1—Sb1i | 107.80 (4) |
| Sb1ii—U1—Sb2iii | 130.699 (18) | Sb1ii—Cu1—Sb1 | 107.80 (4) |
| Cu1i—U1—Sb2vi | 178.364 (13) | Sb1x—Cu1—Sb1 | 112.86 (8) |
| Cu1—U1—Sb2vi | 122.58 (5) | Sb1i—Cu1—Sb1 | 107.80 (4) |
| Cu1ii—U1—Sb2vi | 94.43 (8) | Sb1ii—Cu1—Cu1xi | 126.10 (2) |
| Cu1iii—U1—Sb2vi | 122.58 (5) | Sb1x—Cu1—Cu1xi | 53.90 (2) |
| Sb1i—U1—Sb2vi | 130.699 (18) | Sb1i—Cu1—Cu1xi | 53.90 (2) |
| Sb1iv—U1—Sb2vi | 74.92 (5) | Sb1—Cu1—Cu1xi | 126.10 (2) |
| Sb1v—U1—Sb2vi | 130.699 (18) | Sb1ii—Cu1—Cu1ii | 53.90 (2) |
| Sb1ii—U1—Sb2vi | 74.92 (5) | Sb1x—Cu1—Cu1ii | 126.10 (2) |
| Sb2iii—U1—Sb2vi | 56.44 (5) | Sb1i—Cu1—Cu1ii | 126.10 (2) |
| Cu1i—U1—Sb2 | 122.58 (5) | Sb1—Cu1—Cu1ii | 53.90 (2) |
| Cu1—U1—Sb2 | 94.43 (8) | Cu1xi—Cu1—Cu1ii | 180.0 |
| Cu1ii—U1—Sb2 | 122.58 (5) | Sb1ii—Cu1—Cu1i | 126.10 (2) |
| Cu1iii—U1—Sb2 | 178.364 (13) | Sb1x—Cu1—Cu1i | 126.10 (2) |
| Sb1i—U1—Sb2 | 74.92 (5) | Sb1i—Cu1—Cu1i | 53.90 (2) |
| Sb1iv—U1—Sb2 | 130.699 (18) | Sb1—Cu1—Cu1i | 53.90 (2) |
| Sb1v—U1—Sb2 | 130.699 (18) | Cu1xi—Cu1—Cu1i | 90.0 |
| Sb1ii—U1—Sb2 | 74.92 (5) | Cu1ii—Cu1—Cu1i | 90.0 |
| Sb2iii—U1—Sb2 | 83.93 (8) | Sb1ii—Cu1—Cu1xii | 53.90 (2) |
| Sb2vi—U1—Sb2 | 56.44 (5) | Sb1x—Cu1—Cu1xii | 53.90 (2) |
| Cu1i—U1—Sb2vii | 94.43 (8) | Sb1i—Cu1—Cu1xii | 126.10 (2) |
| Cu1—U1—Sb2vii | 122.58 (5) | Sb1—Cu1—Cu1xii | 126.10 (2) |
| Cu1ii—U1—Sb2vii | 178.364 (13) | Cu1xi—Cu1—Cu1xii | 90.0 |
| Cu1iii—U1—Sb2vii | 122.58 (5) | Cu1ii—Cu1—Cu1xii | 90.0 |
| Sb1i—U1—Sb2vii | 74.92 (5) | Cu1i—Cu1—Cu1xii | 180.0 |
| Sb1iv—U1—Sb2vii | 130.699 (18) | Sb1ii—Cu1—U1i | 167.17 (2) |
| Sb1v—U1—Sb2vii | 74.92 (5) | Sb1x—Cu1—U1i | 66.39 (4) |
| Sb1ii—U1—Sb2vii | 130.699 (18) | Sb1i—Cu1—U1i | 79.97 (8) |
| Sb2iii—U1—Sb2vii | 56.44 (5) | Sb1—Cu1—U1i | 66.39 (4) |
| Sb2vi—U1—Sb2vii | 83.93 (8) | Cu1xi—Cu1—U1i | 60.81 (2) |
| Sb2—U1—Sb2vii | 56.44 (5) | Cu1ii—Cu1—U1i | 119.19 (2) |
| Cu1ii—Sb1—Cu1iii | 72.20 (4) | Cu1i—Cu1—U1i | 60.81 (2) |
| Cu1ii—Sb1—Cu1i | 112.86 (8) | Cu1xii—Cu1—U1i | 119.19 (2) |
| Cu1iii—Sb1—Cu1i | 72.20 (4) | Sb1ii—Cu1—U1 | 66.39 (4) |
| Cu1ii—Sb1—Cu1 | 72.20 (4) | Sb1x—Cu1—U1 | 167.17 (2) |
| Cu1iii—Sb1—Cu1 | 112.86 (8) | Sb1i—Cu1—U1 | 66.39 (4) |
| Cu1i—Sb1—Cu1 | 72.20 (4) | Sb1—Cu1—U1 | 79.97 (8) |
| Cu1ii—Sb1—U1i | 135.568 (16) | Cu1xi—Cu1—U1 | 119.19 (2) |
| Cu1iii—Sb1—U1i | 135.568 (16) | Cu1ii—Cu1—U1 | 60.81 (2) |
| Cu1i—Sb1—U1i | 65.00 (4) | Cu1i—Cu1—U1 | 60.81 (2) |
| Cu1—Sb1—U1i | 65.00 (4) | Cu1xii—Cu1—U1 | 119.19 (2) |
| Cu1ii—Sb1—U1iv | 65.00 (4) | U1i—Cu1—U1 | 121.63 (5) |
| Cu1iii—Sb1—U1iv | 65.00 (4) | Sb1ii—Cu1—U1ii | 79.97 (8) |
| Cu1i—Sb1—U1iv | 135.568 (16) | Sb1x—Cu1—U1ii | 66.39 (4) |
| Cu1—Sb1—U1iv | 135.568 (16) | Sb1i—Cu1—U1ii | 167.17 (2) |
| U1i—Sb1—U1iv | 149.44 (6) | Sb1—Cu1—U1ii | 66.39 (4) |
| Cu1ii—Sb1—U1v | 135.568 (16) | Cu1xi—Cu1—U1ii | 119.19 (2) |
| Cu1iii—Sb1—U1v | 65.00 (4) | Cu1ii—Cu1—U1ii | 60.81 (2) |
| Cu1i—Sb1—U1v | 65.00 (4) | Cu1i—Cu1—U1ii | 119.19 (2) |
| Cu1—Sb1—U1v | 135.568 (16) | Cu1xii—Cu1—U1ii | 60.81 (2) |
| U1i—Sb1—U1v | 86.016 (15) | U1i—Cu1—U1ii | 87.20 (8) |
| U1iv—Sb1—U1v | 86.016 (15) | U1—Cu1—U1ii | 121.63 (5) |
| Cu1ii—Sb1—U1ii | 65.00 (4) | Sb1ii—Cu1—U1x | 66.39 (4) |
| Cu1iii—Sb1—U1ii | 135.568 (16) | Sb1x—Cu1—U1x | 79.97 (8) |
| Cu1i—Sb1—U1ii | 135.568 (16) | Sb1i—Cu1—U1x | 66.39 (4) |
| Cu1—Sb1—U1ii | 65.00 (4) | Sb1—Cu1—U1x | 167.17 (2) |
| U1i—Sb1—U1ii | 86.016 (15) | Cu1xi—Cu1—U1x | 60.81 (2) |
| U1iv—Sb1—U1ii | 86.016 (15) | Cu1ii—Cu1—U1x | 119.19 (2) |
| U1v—Sb1—U1ii | 149.43 (6) | Cu1i—Cu1—U1x | 119.19 (2) |
| Cu1ii—Sb1—U1 | 56.43 (4) | Cu1xii—Cu1—U1x | 60.81 (2) |
| Cu1iii—Sb1—U1 | 56.43 (4) | U1i—Cu1—U1x | 121.63 (5) |
| Cu1i—Sb1—U1 | 56.43 (4) | U1—Cu1—U1x | 87.20 (8) |
| Cu1—Sb1—U1 | 56.43 (4) | U1ii—Cu1—U1x | 121.63 (5) |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) −x+1, −y, −z+1; (iii) x−1, y, z; (iv) −x, −y, −z+1; (v) −x, −y+1, −z+1; (vi) −x+1, −y, −z; (vii) −x+1, −y+1, −z; (viii) −x+2, −y+1, −z; (ix) −x+2, −y, −z; (x) x+1, y, z; (xi) −x+2, −y+1, −z+1; (xii) −x+2, −y, −z+1. |
(I_120K) uranium copper diantimonide
top
Crystal data top
| UCu0.60(4)Sb2 | Dx = 9.834 Mg m−3 |
| Mr = 519.65 | Mo Kα radiation, λ = 0.71073 Å |
| Tetragonal, P4/nmm | Cell parameters from 1591 reflections |
| Hall symbol: -P 4a 2a | θ = 4.4–28.6° |
| a = 4.331 (4) Å | µ = 64.56 mm−1 |
| c = 9.355 (9) Å | T = 120 K |
| V = 175.5 (3) Å3 | Plate, black |
| Z = 2 | 0.14 × 0.08 × 0.05 mm |
| F(000) = 422.8 | |
Data collection top
Bruker SMART 1000 CCD
diffractometer | 160 independent reflections |
| Radiation source: fine-focus sealed tube | 157 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.061 |
| ω scans | θmax = 28.8°, θmin = 2.2° |
Absorption correction: numerical
face-indexed (SADABS; Bruker, 2003) | h = −5→5 |
| Tmin = 0.016, Tmax = 0.086 | k = −5→5 |
| 1966 measured reflections | l = −12→12 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.027 | w = 1/[σ2(Fo2) + (0.0382P)2 + 0.8954P]
where P = (Fo2 + 2Fc2)/3 |
| wR(F2) = 0.066 | (Δ/σ)max = 0.012 |
| S = 1.32 | Δρmax = 3.26 e Å−3 |
| 160 reflections | Δρmin = −4.14 e Å−3 |
| 13 parameters | Extinction correction: SHELXL97 (Sheldrick, 2008), Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| 0 restraints | Extinction coefficient: 0.021 (2) |
Crystal data top
| UCu0.60(4)Sb2 | Z = 2 |
| Mr = 519.65 | Mo Kα radiation |
| Tetragonal, P4/nmm | µ = 64.56 mm−1 |
| a = 4.331 (4) Å | T = 120 K |
| c = 9.355 (9) Å | 0.14 × 0.08 × 0.05 mm |
| V = 175.5 (3) Å3 | |
Data collection top
Bruker SMART 1000 CCD
diffractometer | 160 independent reflections |
Absorption correction: numerical
face-indexed (SADABS; Bruker, 2003) | 157 reflections with I > 2σ(I) |
| Tmin = 0.016, Tmax = 0.086 | Rint = 0.061 |
| 1966 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.027 | 13 parameters |
| wR(F2) = 0.066 | 0 restraints |
| S = 1.32 | Δρmax = 3.26 e Å−3 |
| 160 reflections | Δρmin = −4.14 e Å−3 |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| U1 | 0.2500 | 0.2500 | 0.25724 (6) | 0.0069 (4) | |
| Sb1 | 0.2500 | 0.2500 | 0.65345 (12) | 0.0091 (4) | |
| Sb2 | 0.7500 | 0.2500 | 0.0000 | 0.0063 (4) | |
| Cu1 | 0.7500 | 0.2500 | 0.5000 | 0.0056 (14) | 0.600 (13) |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| U1 | 0.0056 (4) | 0.0056 (4) | 0.0096 (5) | 0.000 | 0.000 | 0.000 |
| Sb1 | 0.0064 (5) | 0.0064 (5) | 0.0144 (7) | 0.000 | 0.000 | 0.000 |
| Sb2 | 0.0057 (5) | 0.0057 (5) | 0.0074 (7) | 0.000 | 0.000 | 0.000 |
| Cu1 | 0.0073 (16) | 0.0073 (16) | 0.0021 (19) | 0.000 | 0.000 | 0.000 |
Geometric parameters (Å, º) top
| U1—Cu1i | 3.138 (2) | Sb1—U1iv | 3.174 (3) |
| U1—Cu1 | 3.138 (2) | Sb1—U1v | 3.174 (3) |
| U1—Cu1ii | 3.138 (2) | Sb1—U1ii | 3.174 (3) |
| U1—Cu1iii | 3.138 (2) | Sb2—Sb2viii | 3.062 (3) |
| U1—Sb1i | 3.174 (3) | Sb2—Sb2vi | 3.062 (3) |
| U1—Sb1iv | 3.174 (3) | Sb2—Sb2vii | 3.062 (3) |
| U1—Sb1v | 3.174 (3) | Sb2—Sb2ix | 3.062 (3) |
| U1—Sb1ii | 3.174 (3) | Sb2—U1vi | 3.237 (2) |
| U1—Sb2iii | 3.237 (2) | Sb2—U1x | 3.237 (2) |
| U1—Sb2vi | 3.237 (2) | Sb2—U1vii | 3.237 (2) |
| U1—Sb2 | 3.237 (2) | Cu1—Sb1ii | 2.598 (2) |
| U1—Sb2vii | 3.237 (2) | Cu1—Sb1x | 2.598 (2) |
| Sb1—Cu1ii | 2.598 (2) | Cu1—Sb1i | 2.598 (2) |
| Sb1—Cu1iii | 2.598 (2) | Cu1—U1i | 3.138 (2) |
| Sb1—Cu1i | 2.598 (2) | Cu1—U1x | 3.138 (2) |
| Sb1—Cu1 | 2.598 (2) | Cu1—U1ii | 3.138 (2) |
| Sb1—U1i | 3.174 (3) | | |
| | | |
| Cu1i—U1—Cu1 | 58.41 (5) | Cu1—Sb1—U1iv | 135.563 (16) |
| Cu1i—U1—Cu1ii | 87.28 (8) | U1i—Sb1—U1iv | 149.48 (6) |
| Cu1—U1—Cu1ii | 58.41 (5) | Cu1ii—Sb1—U1v | 135.563 (16) |
| Cu1i—U1—Cu1iii | 58.41 (5) | Cu1iii—Sb1—U1v | 64.97 (4) |
| Cu1—U1—Cu1iii | 87.28 (8) | Cu1i—Sb1—U1v | 64.97 (4) |
| Cu1ii—U1—Cu1iii | 58.41 (5) | Cu1—Sb1—U1v | 135.563 (16) |
| Cu1i—U1—Sb1i | 48.605 (16) | U1i—Sb1—U1v | 86.028 (15) |
| Cu1—U1—Sb1i | 48.605 (16) | U1iv—Sb1—U1v | 86.028 (15) |
| Cu1ii—U1—Sb1i | 106.28 (5) | Cu1ii—Sb1—U1ii | 64.97 (4) |
| Cu1iii—U1—Sb1i | 106.28 (5) | Cu1iii—Sb1—U1ii | 135.563 (16) |
| Cu1i—U1—Sb1iv | 106.28 (5) | Cu1i—Sb1—U1ii | 135.563 (16) |
| Cu1—U1—Sb1iv | 106.28 (5) | Cu1—Sb1—U1ii | 64.97 (4) |
| Cu1ii—U1—Sb1iv | 48.605 (16) | U1i—Sb1—U1ii | 86.028 (15) |
| Cu1iii—U1—Sb1iv | 48.605 (16) | U1iv—Sb1—U1ii | 86.028 (15) |
| Sb1i—U1—Sb1iv | 149.48 (6) | U1v—Sb1—U1ii | 149.48 (6) |
| Cu1i—U1—Sb1v | 48.605 (16) | Cu1ii—Sb1—U1 | 56.46 (4) |
| Cu1—U1—Sb1v | 106.28 (5) | Cu1iii—Sb1—U1 | 56.46 (4) |
| Cu1ii—U1—Sb1v | 106.28 (5) | Cu1i—Sb1—U1 | 56.46 (4) |
| Cu1iii—U1—Sb1v | 48.605 (16) | Cu1—Sb1—U1 | 56.46 (4) |
| Sb1i—U1—Sb1v | 86.028 (15) | U1i—Sb1—U1 | 105.26 (3) |
| Sb1iv—U1—Sb1v | 86.028 (15) | U1iv—Sb1—U1 | 105.26 (3) |
| Cu1i—U1—Sb1ii | 106.28 (5) | U1v—Sb1—U1 | 105.26 (3) |
| Cu1—U1—Sb1ii | 48.605 (16) | U1ii—Sb1—U1 | 105.26 (3) |
| Cu1ii—U1—Sb1ii | 48.605 (16) | Sb2viii—Sb2—Sb2vi | 180.0 |
| Cu1iii—U1—Sb1ii | 106.28 (5) | Sb2viii—Sb2—Sb2vii | 90.0 |
| Sb1i—U1—Sb1ii | 86.028 (15) | Sb2vi—Sb2—Sb2vii | 90.0 |
| Sb1iv—U1—Sb1ii | 86.028 (15) | Sb2viii—Sb2—Sb2ix | 90.0 |
| Sb1v—U1—Sb1ii | 149.48 (6) | Sb2vi—Sb2—Sb2ix | 90.0 |
| Cu1i—U1—Sb2iii | 122.55 (5) | Sb2vii—Sb2—Sb2ix | 180.0 |
| Cu1—U1—Sb2iii | 178.345 (14) | Sb2viii—Sb2—U1vi | 118.23 (2) |
| Cu1ii—U1—Sb2iii | 122.55 (5) | Sb2vi—Sb2—U1vi | 61.77 (2) |
| Cu1iii—U1—Sb2iii | 94.38 (8) | Sb2vii—Sb2—U1vi | 118.23 (2) |
| Sb1i—U1—Sb2iii | 130.689 (19) | Sb2ix—Sb2—U1vi | 61.77 (2) |
| Sb1iv—U1—Sb2iii | 74.89 (5) | Sb2viii—Sb2—U1x | 61.77 (2) |
| Sb1v—U1—Sb2iii | 74.89 (5) | Sb2vi—Sb2—U1x | 118.23 (2) |
| Sb1ii—U1—Sb2iii | 130.689 (19) | Sb2vii—Sb2—U1x | 118.23 (2) |
| Cu1i—U1—Sb2vi | 178.345 (14) | Sb2ix—Sb2—U1x | 61.77 (2) |
| Cu1—U1—Sb2vi | 122.55 (5) | U1vi—Sb2—U1x | 123.54 (5) |
| Cu1ii—U1—Sb2vi | 94.38 (8) | Sb2viii—Sb2—U1 | 118.23 (2) |
| Cu1iii—U1—Sb2vi | 122.55 (5) | Sb2vi—Sb2—U1 | 61.77 (2) |
| Sb1i—U1—Sb2vi | 130.689 (19) | Sb2vii—Sb2—U1 | 61.77 (2) |
| Sb1iv—U1—Sb2vi | 74.89 (5) | Sb2ix—Sb2—U1 | 118.23 (2) |
| Sb1v—U1—Sb2vi | 130.689 (19) | U1vi—Sb2—U1 | 123.54 (5) |
| Sb1ii—U1—Sb2vi | 74.89 (5) | U1x—Sb2—U1 | 83.96 (8) |
| Sb2iii—U1—Sb2vi | 56.46 (5) | Sb2viii—Sb2—U1vii | 61.77 (2) |
| Cu1i—U1—Sb2 | 122.55 (5) | Sb2vi—Sb2—U1vii | 118.23 (2) |
| Cu1—U1—Sb2 | 94.38 (8) | Sb2vii—Sb2—U1vii | 61.77 (2) |
| Cu1ii—U1—Sb2 | 122.55 (5) | Sb2ix—Sb2—U1vii | 118.23 (2) |
| Cu1iii—U1—Sb2 | 178.345 (14) | U1vi—Sb2—U1vii | 83.96 (8) |
| Sb1i—U1—Sb2 | 74.89 (5) | U1x—Sb2—U1vii | 123.54 (5) |
| Sb1iv—U1—Sb2 | 130.689 (19) | U1—Sb2—U1vii | 123.54 (5) |
| Sb1v—U1—Sb2 | 130.689 (19) | Sb1ii—Cu1—Sb1x | 107.78 (4) |
| Sb1ii—U1—Sb2 | 74.89 (5) | Sb1ii—Cu1—Sb1i | 112.92 (8) |
| Sb2iii—U1—Sb2 | 83.96 (8) | Sb1x—Cu1—Sb1i | 107.78 (4) |
| Sb2vi—U1—Sb2 | 56.46 (5) | Sb1ii—Cu1—Sb1 | 107.78 (4) |
| Cu1i—U1—Sb2vii | 94.38 (8) | Sb1x—Cu1—Sb1 | 112.92 (8) |
| Cu1—U1—Sb2vii | 122.55 (5) | Sb1i—Cu1—Sb1 | 107.78 (4) |
| Cu1ii—U1—Sb2vii | 178.345 (14) | Sb1ii—Cu1—U1 | 66.43 (4) |
| Cu1iii—U1—Sb2vii | 122.55 (5) | Sb1x—Cu1—U1 | 167.18 (2) |
| Sb1i—U1—Sb2vii | 74.89 (5) | Sb1i—Cu1—U1 | 66.43 (4) |
| Sb1iv—U1—Sb2vii | 130.689 (19) | Sb1—Cu1—U1 | 79.90 (8) |
| Sb1v—U1—Sb2vii | 74.89 (5) | Sb1ii—Cu1—U1i | 167.18 (2) |
| Sb1ii—U1—Sb2vii | 130.689 (19) | Sb1x—Cu1—U1i | 66.43 (4) |
| Sb2iii—U1—Sb2vii | 56.46 (5) | Sb1i—Cu1—U1i | 79.90 (8) |
| Sb2vi—U1—Sb2vii | 83.96 (8) | Sb1—Cu1—U1i | 66.43 (4) |
| Sb2—U1—Sb2vii | 56.46 (5) | U1—Cu1—U1i | 121.59 (5) |
| Cu1ii—Sb1—Cu1iii | 72.22 (4) | Sb1ii—Cu1—U1x | 66.43 (4) |
| Cu1ii—Sb1—Cu1i | 112.92 (8) | Sb1x—Cu1—U1x | 79.90 (8) |
| Cu1iii—Sb1—Cu1i | 72.22 (4) | Sb1i—Cu1—U1x | 66.43 (4) |
| Cu1ii—Sb1—Cu1 | 72.22 (4) | Sb1—Cu1—U1x | 167.18 (2) |
| Cu1iii—Sb1—Cu1 | 112.92 (8) | U1—Cu1—U1x | 87.28 (8) |
| Cu1i—Sb1—Cu1 | 72.22 (4) | U1i—Cu1—U1x | 121.59 (5) |
| Cu1ii—Sb1—U1i | 135.563 (16) | Sb1ii—Cu1—U1ii | 79.90 (8) |
| Cu1iii—Sb1—U1i | 135.563 (16) | Sb1x—Cu1—U1ii | 66.43 (4) |
| Cu1i—Sb1—U1i | 64.97 (4) | Sb1i—Cu1—U1ii | 167.18 (2) |
| Cu1—Sb1—U1i | 64.97 (4) | Sb1—Cu1—U1ii | 66.43 (4) |
| Cu1ii—Sb1—U1iv | 64.97 (4) | U1—Cu1—U1ii | 121.59 (5) |
| Cu1iii—Sb1—U1iv | 64.97 (4) | U1i—Cu1—U1ii | 87.28 (8) |
| Cu1i—Sb1—U1iv | 135.563 (16) | U1x—Cu1—U1ii | 121.59 (5) |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) −x+1, −y, −z+1; (iii) x−1, y, z; (iv) −x, −y, −z+1; (v) −x, −y+1, −z+1; (vi) −x+1, −y, −z; (vii) −x+1, −y+1, −z; (viii) −x+2, −y+1, −z; (ix) −x+2, −y, −z; (x) x+1, y, z. |
Experimental details
| | (I_100K) | (I_120K) |
| Crystal data |
| Chemical formula | UCu0.60(4)Sb2 | UCu0.60(4)Sb2 |
| Mr | 521.84 | 519.65 |
| Crystal system, space group | Tetragonal, P4/nmm | Tetragonal, P4/nmm |
| Temperature (K) | 100 | 120 |
| a, c (Å) | 4.320 (4), 9.341 (9) | 4.331 (4), 9.355 (9) |
| V (Å3) | 174.3 (3) | 175.5 (3) |
| Z | 2 | 2 |
| Radiation type | Mo Kα | Mo Kα |
| µ (mm−1) | 65.20 | 64.56 |
| Crystal size (mm) | 0.14 × 0.08 × 0.05 | 0.14 × 0.08 × 0.05 |
| |
| Data collection |
| Diffractometer | Bruker SMART 1000 CCD
diffractometer | Bruker SMART 1000 CCD
diffractometer |
| Absorption correction | Numerical
face-indexed (SADABS; Bruker, 2003) | Numerical
face-indexed (SADABS; Bruker, 2003) |
| Tmin, Tmax | 0.020, 0.086 | 0.016, 0.086 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 1951, 155, 155 | 1966, 160, 157 |
| Rint | 0.075 | 0.061 |
| (sin θ/λ)max (Å−1) | 0.663 | 0.677 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.024, 0.058, 1.20 | 0.027, 0.066, 1.32 |
| No. of reflections | 155 | 160 |
| No. of parameters | 13 | 13 |
| Δρmax, Δρmin (e Å−3) | 1.32, −3.32 | 3.26, −4.14 |
Selected geometric parameters (Å, º) for (I_100K) top| U1—Cu1i | 3.132 (2) | Sb1—Cu1iii | 2.593 (2) |
| U1—Sb1i | 3.167 (3) | Sb2—Sb2iv | 3.055 (3) |
| U1—Sb2ii | 3.231 (2) | Cu1—Cu1v | 3.055 (3) |
| | | |
| Sb1iii—Cu1—Sb1i | 112.86 (8) | | |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) x−1, y, z; (iii) −x+1, −y, −z+1; (iv) −x+2, −y+1, −z; (v) −x+2, −y+1, −z+1. |

 journal menu
journal menu








![[Figure 1]](https://journals.iucr.org/c/issues/2008/08/00/sq3154/sq3154fig1thm.gif)
![[Figure 2]](https://journals.iucr.org/c/issues/2008/08/00/sq3154/sq3154fig2thm.gif)
![[Figure 3]](https://journals.iucr.org/c/issues/2008/08/00/sq3154/sq3154fig3thm.gif)




UCu0.60 (4)Sb2 crystallizes with two formula units in the tetragonal space group P4/nmm. It possesses the HfCuSi2 structure type (Andrukhiv et al., 1975). Zigzag U1Sb1 sheets stack perpendicular to the c axis (Fig. 1). These sheets are separated by square-planar nets of Sb2 atoms and Cu1 atoms, the repeating pattern being (a) Sb2 planar net, (b) U1Sb1 sheet, (c) Cu1 planar net and (d) U1Sb1 sheet [aligned antiparallel in the c direction to the (b) sheet]. The U1 and Sb1 atoms have 4mm site symmetry, whereas the Cu1 and Sb2 atoms have 42m site symmetry. The U1 atoms are surrounded by a square antiprism of Sb1 and Sb2 atoms. The geometry around the Sb1 atom is a square antiprism of Cu1 and U1 atoms. The Cu1 atoms are coordinated to four Sb1 atoms in a tetrahedral arrangement, and Sb2 atoms are coordinated to four U1 atoms, also in a tetrahedral arrangement.
Since the UMSb2 compounds (M is a transition metal) were first reported [M = Ni, Cu (Kaczorowski, 1992); M = Fe, Ru, Co, Pd, Ag, Au (Kaczorowski et al., 1998)], they have received considerable attention, mainly because of their interesting magnetic properties, including Kondo behaviour (Kaczorowski et al., 1998; Bukowski et al., 2005). Large stoichiometric variations are frequently encountered in these UMSb2 compounds, examples being UCo0.46Sb2 (Bukowski et al., 2004) and UNi0.5Sb2. Such variations are also common among rare-earth analogues (Wollesen et al., 1996). The compound UCu0.60 (4)Sb2 reported here is another stoichiometric variation in the UCuxSb2 system, for which the known compounds are UCu0.44Sb2 (Bobev et al., 2006), UCu0.83Sb2 (Bukowski et al., 2005) and UCuSb2 (Kaczorowski et al., 1998). Reaction conditions and starting compositions are presumably the determining factors in the final Cu occupancy. This family of compounds obeys Vegard's Law (Vegard, 1921): the cell constant c increases linearly with Cu occupancy, as shown in Fig. 2, even though the present structures were determined at 100 and 120 K and the previous structures were determined at 298 K. This linear relationship holds at all Cu occupancies, including the `zero occupancy', i.e. USb2. The structures of USb2 and UCuSb2 are indeed related (Fig. 3). In UCuSb2, the voids between adjacent USb zigzag sheets are filled with Cu atoms, effectively pulling the sheets apart and preventing interlayer U—Sb bonding along the c axis. This simple geometric consideration explains why the c axis lengthens but the a axis remains unchanged when the Cu occupancy is increased.