The crystal structures of the dimeric pentahalides (MX5)2 have a close-packed arrangement of halogen atoms in which one fifth of the octahedral holes are occupied by metal atoms. From considerations of the geometrical possibilities of arranging the molecules and thereby resulting symmetry restrictions, the possible space groups and structures are elucidated with the aid of group-subgroup relationships. It is shown that only certain space groups can be achieved. Among the centrosymmetric space groups these include: 1. C2/m, C2/c and C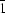 for hexagonal close packing when octahedral holes are occupied between layers A and B but not between B and A; 2. Pnma, Pnmn, Pnab, C2/m, P2/m, P21/m, P21/a and P
for hexagonal close packing when octahedral holes are occupied between layers A and B but not between B and A; 2. Pnma, Pnmn, Pnab, C2/m, P2/m, P21/m, P21/a and P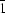 for h.c.p. when holes between all pairs of layers are occupied; 3. P21/m, P21/a and P
for h.c.p. when holes between all pairs of layers are occupied; 3. P21/m, P21/a and P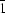 for the double hexagonal close packing; 4. I41/a, Bbmb, Fddd and the monoclinic and triclinic space groups for cubic close packing. The more important arrangements are illustrated and their expected cell dimensions are given. The results are compared with the known structures of the pentachlorides of Nb, Ta, Mo, W, U and Re, (NbBr5)2, β-(PaBr5)2, and also (WSCl4)2 and (WSBr4)2. It is deduced that the previously published structure of niobium pentaiodide is probably in error. Predictions are made for likely structures of (TaI5)2, α-(PaBr5)2, (PaI5)2 and (UBr5)2.
for the double hexagonal close packing; 4. I41/a, Bbmb, Fddd and the monoclinic and triclinic space groups for cubic close packing. The more important arrangements are illustrated and their expected cell dimensions are given. The results are compared with the known structures of the pentachlorides of Nb, Ta, Mo, W, U and Re, (NbBr5)2, β-(PaBr5)2, and also (WSCl4)2 and (WSBr4)2. It is deduced that the previously published structure of niobium pentaiodide is probably in error. Predictions are made for likely structures of (TaI5)2, α-(PaBr5)2, (PaI5)2 and (UBr5)2.

 journal menu
journal menu











