Buy article online - an online subscription or single-article purchase is required to access this article.
In 4,4′-biphenol–1,4-diazabicyclo[2.2.2]octane (1/1), C12H10O2.C6H12N2 (1), monoclinic, C2/c, a = 12.1510 (9), b = 6.3202 (4), c = 21.051 (2) Å, β = 98.875 (10)° with Z = 4, the biphenol units lie across twofold rotation axes and the diamine units are disordered across centres of inversion. The molecular components are linked by means of O—H N hydrogen bonds into zigzag chains with graph set C
N hydrogen bonds into zigzag chains with graph set C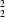 (16) running parallel to the [201] direction; the chains are not coiled and there are no interactions between neighbouring chains. In 4,4′-biphenol–1,2-diaminoethane–methanol (2/1/1), (C12H10O2)2.C2H8N2.CH4O (2), triclinic, P1, a = 9.4592 (7), b = 10.6311 (10), c = 12.2085 (14) Å, α = 91.812 (9), β = 93.091 (6), γ = 90.652 (6)° with Z = 2, there are seven independent hydrogen bonds of O—H
(16) running parallel to the [201] direction; the chains are not coiled and there are no interactions between neighbouring chains. In 4,4′-biphenol–1,2-diaminoethane–methanol (2/1/1), (C12H10O2)2.C2H8N2.CH4O (2), triclinic, P1, a = 9.4592 (7), b = 10.6311 (10), c = 12.2085 (14) Å, α = 91.812 (9), β = 93.091 (6), γ = 90.652 (6)° with Z = 2, there are seven independent hydrogen bonds of O—H O, O—H
O, O—H N and N—H
N and N—H O types and each oxygen and each nitrogen acts as a single donor and single acceptor of hydrogen bonds. The four independent components are linked by means of O—H
O types and each oxygen and each nitrogen acts as a single donor and single acceptor of hydrogen bonds. The four independent components are linked by means of O—H O and O—H
O and O—H N hydrogen bonds into a linear aggregate, and further O—H
N hydrogen bonds into a linear aggregate, and further O—H N and O—H
N and O—H O hydrogen bonds generate chains in the [011] direction containing alternating biphenol and diamine units, and in the [021] direction containing methanol and pairs of biphenol molecules. These two types of chain intersect to form a continuous two-dimensional net, graph set
O hydrogen bonds generate chains in the [011] direction containing alternating biphenol and diamine units, and in the [021] direction containing methanol and pairs of biphenol molecules. These two types of chain intersect to form a continuous two-dimensional net, graph set 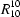 (62), with biphenol molecules at the nodes: pairs of these nets are linked into bilayers by N—H
(62), with biphenol molecules at the nodes: pairs of these nets are linked into bilayers by N—H O hydrogen bonds, but the nets are not interwoven. In 4,4′-biphenol–1,2-diaminoethane (1/1), C12H10O2.C2H8N2 (3), monoclinic, P21/a, a = 8.2121 (5), b = 5.3401 (3), c = 14.6014 (9) Å, β = 99.962 (7)° with Z = 2, each of the components lies across a centre of inversion. The O atoms act as single donors and double acceptors of hydrogen bonds and the N atoms as double donors and single acceptors, so that both oxygen and nitrogen are effectively four-coordinate; all the hydrogen bonds are of O—H
O hydrogen bonds, but the nets are not interwoven. In 4,4′-biphenol–1,2-diaminoethane (1/1), C12H10O2.C2H8N2 (3), monoclinic, P21/a, a = 8.2121 (5), b = 5.3401 (3), c = 14.6014 (9) Å, β = 99.962 (7)° with Z = 2, each of the components lies across a centre of inversion. The O atoms act as single donors and double acceptors of hydrogen bonds and the N atoms as double donors and single acceptors, so that both oxygen and nitrogen are effectively four-coordinate; all the hydrogen bonds are of O—H N or N—H
N or N—H O type. Chains built from N—H
O type. Chains built from N—H O and O—H
O and O—H N hydrogen bonds run parallel to [100] and [010] and the intersection of these gives two-dimensional nets built from
N hydrogen bonds run parallel to [100] and [010] and the intersection of these gives two-dimensional nets built from  (12) rings: these nets are linked by the —C6H4C6H4— and —CH2CH2— spacer units into a continuous three-dimensional diamondoid structure.
(12) rings: these nets are linked by the —C6H4C6H4— and —CH2CH2— spacer units into a continuous three-dimensional diamondoid structure.
 N hydrogen bonds into zigzag chains with graph set C
N hydrogen bonds into zigzag chains with graph set C O, O—H
O, O—H N and N—H
N and N—H O types and each oxygen and each nitrogen acts as a single donor and single acceptor of hydrogen bonds. The four independent components are linked by means of O—H
O types and each oxygen and each nitrogen acts as a single donor and single acceptor of hydrogen bonds. The four independent components are linked by means of O—H O and O—H
O and O—H N hydrogen bonds into a linear aggregate, and further O—H
N hydrogen bonds into a linear aggregate, and further O—H N and O—H
N and O—H O hydrogen bonds generate chains in the [011] direction containing alternating biphenol and diamine units, and in the [021] direction containing methanol and pairs of biphenol molecules. These two types of chain intersect to form a continuous two-dimensional net, graph set
O hydrogen bonds generate chains in the [011] direction containing alternating biphenol and diamine units, and in the [021] direction containing methanol and pairs of biphenol molecules. These two types of chain intersect to form a continuous two-dimensional net, graph set  O hydrogen bonds, but the nets are not interwoven. In 4,4′-biphenol–1,2-diaminoethane (1/1), C12H10O2.C2H8N2 (3), monoclinic, P21/a, a = 8.2121 (5), b = 5.3401 (3), c = 14.6014 (9) Å, β = 99.962 (7)° with Z = 2, each of the components lies across a centre of inversion. The O atoms act as single donors and double acceptors of hydrogen bonds and the N atoms as double donors and single acceptors, so that both oxygen and nitrogen are effectively four-coordinate; all the hydrogen bonds are of O—H
O hydrogen bonds, but the nets are not interwoven. In 4,4′-biphenol–1,2-diaminoethane (1/1), C12H10O2.C2H8N2 (3), monoclinic, P21/a, a = 8.2121 (5), b = 5.3401 (3), c = 14.6014 (9) Å, β = 99.962 (7)° with Z = 2, each of the components lies across a centre of inversion. The O atoms act as single donors and double acceptors of hydrogen bonds and the N atoms as double donors and single acceptors, so that both oxygen and nitrogen are effectively four-coordinate; all the hydrogen bonds are of O—H N or N—H
N or N—H O type. Chains built from N—H
O type. Chains built from N—H O and O—H
O and O—H N hydrogen bonds run parallel to [100] and [010] and the intersection of these gives two-dimensional nets built from
N hydrogen bonds run parallel to [100] and [010] and the intersection of these gives two-dimensional nets built from 
 journal menu
journal menu









 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials


