Buy article online - an online subscription or single-article purchase is required to access this article.
The structure of the title compound, [Co(C
4H
4NOS
2)
2(CH
4O)
2], has been determined at 180 (2) K. The molecule possesses
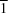
point symmetry with the Co
II atom adopting a Jahn–Teller-distorted octahedral geometry. The molecules are linked into chains by O—H

O hydrogen-bond interactions involving the H atom of the coordinated methanol molecules.
Supporting information
CCDC reference: 162796
Key indicators
- Single-crystal X-ray study
- T = 180 K
- Mean
![[sigma]](https://journals.iucr.org/logos/entities/sigma_rmgif.gif) (C-C) = 0.005 Å
(C-C) = 0.005 Å
- R factor = 0.034
- wR factor = 0.078
- Data-to-parameter ratio = 13.8
checkCIF results
No syntax errors found
ADDSYM reports no extra symmetry
 Alert Level C:
Alert Level C:
ABSTM_02 Alert C The ratio of Tmax/Tmin expected RT(exp) is > 1.10
Absorption corrections should be applied.
Tmin and Tmax expected: 0.862 0.969
RT(exp) = 1.124
PLAT_710 Alert C Delete 1-2-3 or 2-3-4 (CIF) Linear Torsion Angle # 5
S1 -CO1 -S1 -C1 88.00 70.00 3.565 1.555 1.555 1.555
PLAT_710 Alert C Delete 1-2-3 or 2-3-4 (CIF) Linear Torsion Angle # 6
O1 -CO1 -O1 -N1 -44.00100.00 3.565 1.555 1.555 1.555
PLAT_710 Alert C Delete 1-2-3 or 2-3-4 (CIF) Linear Torsion Angle # 13
O2 -CO1 -O2 -C5 51.00 65.00 3.565 1.555 1.555 1.555
0 Alert Level A = Potentially serious problem
0 Alert Level B = Potential problem
4 Alert Level C = Please check
HMTT was obtained from the Aldrich Chemical Co. and was recrystallized from
CH2Cl2. The sodium salt hydrate Na(MTT).xH2O was prepared by addition of
1 mol equivalent of NaOH to a suspension of HMTT in water, followed by
evaporation of the solvent. The cobalt(II) complex was prepared initially as a
monohydrate Co(MTT)2·H2O by combination of aqueous solutions of
CoCl2 and Na(MTT) in a 1:2 molar ratio at room temperature. The resulting
pink precipitate was removed by filtration under gravity and dried in air at
room temperature. Single crystals of (I) were grown by slow evaporation of a
solution of Co(MTT)2·H2O in methanol at room temperature.
All H atoms except H1 were placed geometrically and allowed to ride during
subsequent refinement. H1 was located in a difference Fourier map and refined
freely with an isotropic displacement parameter.
Data collection: COLLECT (Nonius, 1998); cell refinement: HKL SCALEPACK (Otwinowski & Minor, 1997); data reduction: HKL DENZO and SCALEPACK (Otwinowski & Minor, 1997); program(s) used to solve structure: SHELXS97 (Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); software used to prepare material for publication: SHELXL97.
Bis[3-hydroxy-4-methylthiazole-2(3
H)-thiolato-
S2,
O]bis(methanol-O)- cobalt(II)
top
Crystal data top
| [Co(C4H4NOS2)2(CH4O)2] | F(000) = 426 |
| Mr = 415.42 | Dx = 1.692 Mg m−3 |
| Monoclinic, P21/c | Mo Kα radiation, λ = 0.71073 Å |
| a = 5.0414 (4) Å | Cell parameters from 9582 reflections |
| b = 18.878 (2) Å | θ = 2.9–25.0° |
| c = 8.6814 (11) Å | µ = 1.58 mm−1 |
| β = 99.349 (7)° | T = 180 K |
| V = 815.23 (16) Å3 | Plate, red |
| Z = 2 | 0.13 × 0.08 × 0.02 mm |
Data collection top
Nonius KappaCCD
diffractometer | 1087 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.029 |
| Graphite monochromator | θmax = 25.0°, θmin = 4.0° |
| Thin–slice ω and ϕ scans | h = −5→5 |
| 2478 measured reflections | k = −20→22 |
| 1424 independent reflections | l = −10→10 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.034 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.078 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.01 | w = 1/[σ2(Fo2) + (0.0359P)2]
where P = (Fo2 + 2Fc2)/3 |
| 1424 reflections | (Δ/σ)max = 0.017 |
| 103 parameters | Δρmax = 0.31 e Å−3 |
| 0 restraints | Δρmin = −0.42 e Å−3 |
Crystal data top
| [Co(C4H4NOS2)2(CH4O)2] | V = 815.23 (16) Å3 |
| Mr = 415.42 | Z = 2 |
| Monoclinic, P21/c | Mo Kα radiation |
| a = 5.0414 (4) Å | µ = 1.58 mm−1 |
| b = 18.878 (2) Å | T = 180 K |
| c = 8.6814 (11) Å | 0.13 × 0.08 × 0.02 mm |
| β = 99.349 (7)° | |
Data collection top
Nonius KappaCCD
diffractometer | 1087 reflections with I > 2σ(I) |
| 2478 measured reflections | Rint = 0.029 |
| 1424 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.034 | 0 restraints |
| wR(F2) = 0.078 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.01 | Δρmax = 0.31 e Å−3 |
| 1424 reflections | Δρmin = −0.42 e Å−3 |
| 103 parameters | |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes)
are estimated using the full covariance matrix. The cell e.s.d.'s are taken
into account individually in the estimation of e.s.d.'s in distances, angles
and torsion angles; correlations between e.s.d.'s in cell parameters are only
used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s.
planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor
wR and goodness of fit S are based on F2, conventional
R-factors R are based on F, with F set to zero for
negative F2. The threshold expression of F2 >
σ(F2) is used only for calculating R-factors(gt) etc.
and is not relevant to the choice of reflections for refinement.
R-factors based on F2 are statistically about twice as large
as those based on F, and R- factors based on ALL data will be
even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Co1 | 0.0000 | 0.5000 | 0.0000 | 0.0228 (2) | |
| S1 | −0.19352 (14) | 0.38022 (4) | −0.02754 (9) | 0.0299 (2) | |
| S2 | 0.09944 (16) | 0.25946 (4) | 0.15912 (10) | 0.0412 (3) | |
| O1 | 0.2694 (3) | 0.45657 (10) | 0.1764 (2) | 0.0258 (5) | |
| H1 | 0.385 (8) | 0.4983 (19) | −0.163 (4) | 0.049 (13)* | |
| O2 | 0.2661 (4) | 0.47558 (12) | −0.1582 (2) | 0.0307 (5) | |
| N1 | 0.2553 (4) | 0.38537 (12) | 0.1892 (2) | 0.0238 (6) | |
| C1 | 0.0599 (5) | 0.34690 (15) | 0.1057 (3) | 0.0255 (7) | |
| C2 | 0.3815 (6) | 0.27863 (18) | 0.2935 (4) | 0.0413 (8) | |
| H2 | 0.4829 | 0.2442 | 0.3578 | 0.050* | |
| C3 | 0.4403 (5) | 0.34770 (16) | 0.2964 (3) | 0.0303 (7) | |
| C4 | 0.6625 (6) | 0.38600 (18) | 0.3956 (4) | 0.0378 (8) | |
| H4A | 0.7561 | 0.3536 | 0.4745 | 0.045* | |
| H4B | 0.5889 | 0.4258 | 0.4474 | 0.045* | |
| H4C | 0.7893 | 0.4038 | 0.3303 | 0.045* | |
| C5 | 0.2171 (6) | 0.43229 (18) | −0.2938 (4) | 0.0422 (9) | |
| H5A | 0.0743 | 0.3982 | −0.2836 | 0.051* | |
| H5B | 0.3818 | 0.4067 | −0.3055 | 0.051* | |
| H5C | 0.1616 | 0.4621 | −0.3858 | 0.051* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Co1 | 0.0191 (3) | 0.0204 (4) | 0.0276 (4) | 0.0001 (2) | 0.0001 (2) | 0.0020 (2) |
| S1 | 0.0272 (4) | 0.0246 (5) | 0.0353 (5) | −0.0039 (3) | −0.0026 (3) | 0.0020 (3) |
| S2 | 0.0464 (5) | 0.0230 (5) | 0.0511 (6) | 0.0011 (3) | −0.0009 (4) | 0.0063 (4) |
| O1 | 0.0263 (10) | 0.0187 (12) | 0.0316 (12) | −0.0012 (8) | 0.0020 (9) | 0.0021 (9) |
| O2 | 0.0235 (12) | 0.0357 (14) | 0.0331 (13) | −0.0060 (10) | 0.0052 (10) | −0.0071 (11) |
| N1 | 0.0229 (13) | 0.0224 (15) | 0.0259 (14) | 0.0025 (10) | 0.0037 (10) | 0.0034 (11) |
| C1 | 0.0264 (16) | 0.0189 (17) | 0.0325 (17) | 0.0004 (12) | 0.0087 (13) | 0.0022 (13) |
| C2 | 0.0437 (19) | 0.032 (2) | 0.046 (2) | 0.0051 (15) | 0.0006 (16) | 0.0125 (17) |
| C3 | 0.0257 (16) | 0.038 (2) | 0.0280 (17) | 0.0070 (13) | 0.0071 (13) | 0.0108 (15) |
| C4 | 0.0318 (18) | 0.043 (2) | 0.036 (2) | 0.0089 (14) | −0.0022 (15) | 0.0104 (16) |
| C5 | 0.0435 (19) | 0.046 (2) | 0.038 (2) | −0.0074 (15) | 0.0077 (16) | −0.0085 (17) |
Geometric parameters (Å, º) top
| Co1—O1 | 2.0459 (17) | N1—C1 | 1.339 (3) |
| Co1—O1i | 2.0459 (17) | N1—C3 | 1.400 (3) |
| Co1—O2i | 2.121 (2) | C2—C3 | 1.336 (4) |
| Co1—O2 | 2.121 (2) | C2—H2 | 0.9500 |
| Co1—S1i | 2.4590 (7) | C3—C4 | 1.485 (4) |
| Co1—S1 | 2.4590 (7) | C4—H4A | 0.9800 |
| S1—C1 | 1.699 (3) | C4—H4B | 0.9800 |
| S2—C1 | 1.717 (3) | C4—H4C | 0.9800 |
| S2—C2 | 1.725 (3) | C5—H5A | 0.9800 |
| O1—N1 | 1.352 (3) | C5—H5B | 0.9800 |
| O2—C5 | 1.421 (4) | C5—H5C | 0.9800 |
| O2—H1 | 0.75 (4) | | |
| | | |
| O1—Co1—O1i | 180.00 (9) | O1—N1—C3 | 121.5 (2) |
| O1—Co1—O2i | 90.88 (8) | N1—C1—S1 | 125.0 (2) |
| O1i—Co1—O2i | 89.12 (8) | N1—C1—S2 | 109.4 (2) |
| O1—Co1—O2 | 89.12 (8) | S1—C1—S2 | 125.54 (17) |
| O1i—Co1—O2 | 90.88 (8) | C3—C2—S2 | 111.9 (2) |
| O2i—Co1—O2 | 180.00 (12) | C3—C2—H2 | 124.0 |
| O1—Co1—S1i | 95.22 (5) | S2—C2—H2 | 124.0 |
| O1i—Co1—S1i | 84.78 (5) | C2—C3—N1 | 111.2 (3) |
| O2i—Co1—S1i | 91.14 (6) | C2—C3—C4 | 129.0 (3) |
| O2—Co1—S1i | 88.86 (6) | N1—C3—C4 | 119.8 (3) |
| O1—Co1—S1 | 84.78 (5) | C3—C4—H4A | 109.5 |
| O1i—Co1—S1 | 95.22 (5) | C3—C4—H4B | 109.5 |
| O2i—Co1—S1 | 88.86 (6) | H4A—C4—H4B | 109.5 |
| O2—Co1—S1 | 91.14 (6) | C3—C4—H4C | 109.5 |
| S1i—Co1—S1 | 180.0 | H4A—C4—H4C | 109.5 |
| C1—S1—Co1 | 91.82 (10) | H4B—C4—H4C | 109.5 |
| C1—S2—C2 | 91.59 (14) | O2—C5—H5A | 109.5 |
| N1—O1—Co1 | 115.02 (14) | O2—C5—H5B | 109.5 |
| C5—O2—Co1 | 128.20 (18) | H5A—C5—H5B | 109.5 |
| C5—O2—H1 | 109 (3) | O2—C5—H5C | 109.5 |
| Co1—O2—H1 | 121 (3) | H5A—C5—H5C | 109.5 |
| C1—N1—O1 | 122.6 (2) | H5B—C5—H5C | 109.5 |
| C1—N1—C3 | 115.9 (2) | | |
| | | |
| O1—Co1—S1—C1 | −6.19 (11) | Co1—O1—N1—C3 | 174.97 (18) |
| O1i—Co1—S1—C1 | 173.81 (11) | O1—N1—C1—S1 | 0.6 (4) |
| O2i—Co1—S1—C1 | −97.18 (11) | C3—N1—C1—S1 | 178.09 (19) |
| O2—Co1—S1—C1 | 82.82 (11) | O1—N1—C1—S2 | −177.41 (17) |
| S1i—Co1—S1—C1 | 88 (70) | C3—N1—C1—S2 | 0.1 (3) |
| O1i—Co1—O1—N1 | −44 (100) | Co1—S1—C1—N1 | 5.1 (2) |
| O2i—Co1—O1—N1 | 96.91 (16) | Co1—S1—C1—S2 | −177.21 (17) |
| O2—Co1—O1—N1 | −83.09 (16) | C2—S2—C1—N1 | 0.0 (2) |
| S1i—Co1—O1—N1 | −171.87 (14) | C2—S2—C1—S1 | −178.0 (2) |
| S1—Co1—O1—N1 | 8.13 (14) | C1—S2—C2—C3 | −0.1 (3) |
| O1—Co1—O2—C5 | 120.9 (3) | S2—C2—C3—N1 | 0.1 (3) |
| O1i—Co1—O2—C5 | −59.1 (3) | S2—C2—C3—C4 | 179.2 (3) |
| O2i—Co1—O2—C5 | 51 (65) | C1—N1—C3—C2 | −0.1 (3) |
| S1i—Co1—O2—C5 | −143.8 (2) | O1—N1—C3—C2 | 177.4 (2) |
| S1—Co1—O2—C5 | 36.2 (2) | C1—N1—C3—C4 | −179.3 (3) |
| Co1—O1—N1—C1 | −7.7 (3) | O1—N1—C3—C4 | −1.8 (4) |
| Symmetry code: (i) −x, −y+1, −z. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O2—H1···O1ii | 0.75 (4) | 1.96 (4) | 2.696 (3) | 171 (4) |
| Symmetry code: (ii) −x+1, −y+1, −z. |
Experimental details
| Crystal data |
| Chemical formula | [Co(C4H4NOS2)2(CH4O)2] |
| Mr | 415.42 |
| Crystal system, space group | Monoclinic, P21/c |
| Temperature (K) | 180 |
| a, b, c (Å) | 5.0414 (4), 18.878 (2), 8.6814 (11) |
| β (°) | 99.349 (7) |
| V (Å3) | 815.23 (16) |
| Z | 2 |
| Radiation type | Mo Kα |
| µ (mm−1) | 1.58 |
| Crystal size (mm) | 0.13 × 0.08 × 0.02 |
| |
| Data collection |
| Diffractometer | Nonius KappaCCD
diffractometer |
| Absorption correction | – |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 2478, 1424, 1087 |
| Rint | 0.029 |
| (sin θ/λ)max (Å−1) | 0.595 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.034, 0.078, 1.01 |
| No. of reflections | 1424 |
| No. of parameters | 103 |
| H-atom treatment | H atoms treated by a mixture of independent and constrained refinement |
| Δρmax, Δρmin (e Å−3) | 0.31, −0.42 |
Selected bond lengths (Å) top| Co1—O1 | 2.0459 (17) | S2—C2 | 1.725 (3) |
| Co1—O2 | 2.121 (2) | O1—N1 | 1.352 (3) |
| Co1—S1 | 2.4590 (7) | N1—C1 | 1.339 (3) |
| S1—C1 | 1.699 (3) | N1—C3 | 1.400 (3) |
| S2—C1 | 1.717 (3) | C2—C3 | 1.336 (4) |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O2—H1···O1i | 0.75 (4) | 1.96 (4) | 2.696 (3) | 171 (4) |
| Symmetry code: (i) −x+1, −y+1, −z. |

 Subscribe to Acta Crystallographica Section E: Crystallographic Communications
Subscribe to Acta Crystallographica Section E: Crystallographic Communications
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
[email protected] for assistance.
 O hydrogen-bond interactions involving the H atom of the coordinated methanol molecules.
O hydrogen-bond interactions involving the H atom of the coordinated methanol molecules.![[sigma]](https://journals.iucr.org/logos/entities/sigma_rmgif.gif) (C-C) = 0.005 Å
(C-C) = 0.005 ÅAlert Level C:

 Subscribe to Acta Crystallographica Section E: Crystallographic Communications
Subscribe to Acta Crystallographica Section E: Crystallographic Communications
 journal menu
journal menu









![[Figure 1]](https://journals.iucr.org/e/issues/2001/04/00/ya6012/ya6012fig1thm.gif)
![[Figure 2]](https://journals.iucr.org/e/issues/2001/04/00/ya6012/ya6012fig2thm.gif)
![[Figure 3]](https://journals.iucr.org/e/issues/2001/04/00/ya6012/ya6012fig3thm.gif)




Cyclic thiohydroxamic acids, such as 1-hydroxypyridine-2(1H)-thione (pyrithione, HPT), find extensive use as fungicides (Paulus, 1993). It has been suggested that their mode of action is based predominantly on chelate complex formation (Albert, 1973). 3-Hydroxy-4-methylthiazole-2(3H)-thione (methylthiazolethione, HMTT) contains a chelating unit similar to that in pyrithione and might, therefore, be expected to exhibit similar antimicrobial properties. We are currently studying the solid-state chemistry of cyclic thiohydroxamic acids (Bond & Jones, 2000) and their complexes with first-row d-block elements, and report here the crystal structure of the chelate complex Co(MTT)2(methanol)2, (I) (Fig. 1).
In (I), the Co1 atom occupies a centre of symmetry and adopts octahedral coordination via two bidentate MTT ligands and two methanol molecules. The Co1—S1 distance [2.4590 (7) Å] is significantly greater than the Co—S distances in comparable complexes [see, for example, Kang et al. (1993); Co—Save = 2.377 (5) Å]; this may be attributed to a tetragonal Jahn–Teller distortion about the CoII atom (d7). The C1—S1 and N1—O1 bond distances [1.699 (3) Å and 1.352 (3) Å, respectively] suggest that these bonds retain some multiple-bond character [cf. C—Save = 1.773 (9) Å and N—Oave = 1.396 (12) Å; Alen et al., 1987].
The H atom of the coordinated methanol molecule is involved in a hydrogen-bond interaction with the O atom of an MTT ligand in an adjacent molecule [H1···O1i = 1.96 (4) Å, O2—H1···O1i = 171 (4)°; symmetry code: (i) 1 - x, 1 - y, -z], giving rise to extended chains running parallel to the a direction (Fig. 2). These chains pack in a herring-bone-type arrangement (Fig. 3). The vinylic H2 atom may be expected to be sufficiently acidic to become involved in a hydrogen-bond interaction. H2 makes its closest approach to an S atom in an MTT molecule of an adjacent chain [H2···S1ii = 2.82 Å and C2—H2···S1ii = 163.3°; symmetry code: (ii) 1 + x, 1/2 - y, 1/2 + z]. The approximate linear geometry of this interaction may be indicative of a C—H···S-type hydrogen bond.