The crystal structure of the title complex, bis(tetra-
n-butylammonium) bis(
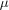
-

-hydroxybenzeneacetato)-1κ
2O1,
O2:2κ
O2;1κ
O2:2κ
2O1,
O2-bis[oxo(peroxo)vanadium(V)] α-hydroxybenzeneacetic acid solvate, (C
16H
36N)
2[V
2O
2(O
2)
2(C
8H
6O
3)
2]·C
8H
8O
3, consists of dimeric anions with twofold rotation symmetry, cations and molecules of mandelic acid. Deprotonated hydroxyl O atoms of the mandelate dianion ligands bridge the V atoms to give a non-planar V
2O
2 ring. Each V atom has distorted pentagonal pyramidal coordination geometry, with an oxo ligand in the axial position. The mandelic acid molecule is disordered and coordinates weakly to the second axial site of each V atom through a carboxylate O atom.
Supporting information
CCDC reference: 142724
A solution of NH4VO3 (2.5 mmol) in water (10 ml) was mixed with a solution of mandelic acid (2.5 mmol) in water (5 ml). After cooling of the solution to 273 K, H2O2 (30%, 5 ml) and ethanol (2.5 mmol, 10 ml) were added with stirring. Russet (dark red below?) crystals were isolated after a few days at 278 K.
All C atoms of the neutral disordered mandelic acid molecule have a site occupation factor of 1/2, while O8 lies on the twofold rotation axis and O7 represents two different O atoms in the two disordered orientations. H atoms of the neutral disordered mandelic acid were set in an idealized positions and their parameters were calculated. Other H-atom parameters were refined independently.
Data collection: KM-4 Software (Galdecki et al., 1996); cell refinement: KM-4 Software; data reduction: KM-4 Software; program(s) used to solve structure: SHELXS97 (Sheldrick, 1990); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: ORTEPIII (Johnson & Burnett, 1996); software used to prepare material for publication: SHELXL97.
Bis(tetra-n-butylammonium) Bis[µ-2-hydroxy-2-phenyl- ethanoato(2-)]-bis[oxomonoperoxovanadate(V)]
top
Crystal data top
| (C16H36N)2[V2O2(O2)2(C8H6O3)2]·C8H8O3 | F(000) = 2432 |
| Mr = 1135.20 | Dx = 1.272 Mg m−3 |
| Orthorhombic, Pbcn | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: -P 2n 2ab | Cell parameters from 41 reflections |
| a = 19.657 (12) Å | θ = 1.6–25.1° |
| b = 17.066 (3) Å | µ = 0.38 mm−1 |
| c = 17.670 (5) Å | T = 150 K |
| V = 5928 (4) Å3 | Prism, dark red |
| Z = 4 | 0.4 × 0.3 × 0.3 mm |
Data collection top
4-circle κ-axis Kuma KM-4
diffractometer with Oxford Cryosytems Cryostream
cooler | Rint = 0.023 |
| Radiation source: fine-focus sealed tube | θmax = 25.1°, θmin = 1.6° |
| Graphite monochromator | h = −23→0 |
| ω/2θ scans | k = −19→3 |
| 5680 measured reflections | l = 0→21 |
| 5210 independent reflections | 3 standard reflections every 300 reflections |
| 3329 reflections with I > 2σ(I) | intensity decay: 2.0% |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.052 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.168 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.03 | w = 1/[σ2(Fo2) + (0.0838P)2 + 5.65P]
where P = (Fo2 + 2Fc2)/3 |
| 5210 reflections | (Δ/σ)max = 0.001 |
| 383 parameters | Δρmax = 0.66 e Å−3 |
| 0 restraints | Δρmin = −0.40 e Å−3 |
Crystal data top
| (C16H36N)2[V2O2(O2)2(C8H6O3)2]·C8H8O3 | V = 5928 (4) Å3 |
| Mr = 1135.20 | Z = 4 |
| Orthorhombic, Pbcn | Mo Kα radiation |
| a = 19.657 (12) Å | µ = 0.38 mm−1 |
| b = 17.066 (3) Å | T = 150 K |
| c = 17.670 (5) Å | 0.4 × 0.3 × 0.3 mm |
Data collection top
4-circle κ-axis Kuma KM-4
diffractometer with Oxford Cryosytems Cryostream
cooler | Rint = 0.023 |
| 5680 measured reflections | 3 standard reflections every 300 reflections |
| 5210 independent reflections | intensity decay: 2.0% |
| 3329 reflections with I > 2σ(I) | |
Refinement top
| R[F2 > 2σ(F2)] = 0.052 | 0 restraints |
| wR(F2) = 0.168 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.03 | Δρmax = 0.66 e Å−3 |
| 5210 reflections | Δρmin = −0.40 e Å−3 |
| 383 parameters | |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| V1 | 0.57814 (3) | 0.69623 (3) | 0.22800 (3) | 0.03233 (18) | |
| O1 | 0.61333 (11) | 0.77895 (14) | 0.21963 (14) | 0.0415 (6) | |
| N1 | 0.77877 (14) | 0.85962 (17) | 0.34708 (16) | 0.0378 (7) | |
| C1 | 0.53831 (19) | 0.6788 (2) | 0.07320 (19) | 0.0407 (8) | |
| O2 | 0.62799 (11) | 0.64963 (15) | 0.30686 (13) | 0.0420 (6) | |
| C2 | 0.47521 (17) | 0.7142 (2) | 0.10889 (17) | 0.0362 (7) | |
| H2 | 0.4358 | 0.6784 | 0.0997 | 0.043* | |
| O3 | 0.64933 (11) | 0.62301 (14) | 0.23374 (13) | 0.0390 (5) | |
| O4 | 0.58532 (12) | 0.66062 (15) | 0.12102 (13) | 0.0441 (6) | |
| O5 | 0.48649 (11) | 0.71961 (13) | 0.18755 (11) | 0.0325 (5) | |
| C5 | 0.45873 (16) | 0.7933 (2) | 0.07630 (19) | 0.0367 (8) | |
| O6 | 0.54194 (15) | 0.66761 (17) | 0.00506 (14) | 0.0556 (7) | |
| C6 | 0.47267 (18) | 0.8614 (2) | 0.1156 (2) | 0.0442 (8) | |
| H6 | 0.4914 | 0.8587 | 0.1651 | 0.053* | |
| C7 | 0.4594 (2) | 0.9332 (2) | 0.0833 (2) | 0.0545 (10) | |
| H7 | 0.4687 | 0.9799 | 0.1107 | 0.065* | |
| O7 | 0.41079 (14) | 0.4824 (2) | 0.2623 (2) | 0.0767 (11) | |
| H7O | 0.398 (4) | 0.528 (5) | 0.278 (4) | 0.15 (3)* | |
| C8 | 0.4327 (2) | 0.9378 (3) | 0.0113 (2) | 0.0568 (11) | |
| H8 | 0.4240 | 0.9875 | −0.0109 | 0.068* | |
| O8 | 0.5000 | 0.5791 (2) | 0.2500 | 0.0514 (10) | |
| C9 | 0.4186 (2) | 0.8706 (3) | −0.0281 (2) | 0.0563 (10) | |
| H9 | 0.4006 | 0.8737 | −0.0779 | 0.068* | |
| C10 | 0.4304 (2) | 0.7987 (2) | 0.0040 (2) | 0.0503 (10) | |
| H10 | 0.4193 | 0.7523 | −0.0230 | 0.060* | |
| C11 | 0.5264 (3) | 0.5147 (4) | 0.2526 (5) | 0.0482 (19) | 0.50 |
| C12 | 0.4826 (6) | 0.4549 (6) | 0.2915 (5) | 0.076 (3) | 0.50 |
| H12 | 0.4858 | 0.4606 | 0.3477 | 0.091* | 0.50 |
| C13 | 0.4975 (8) | 0.3697 (6) | 0.2677 (4) | 0.037 (4) | 0.50 |
| C14 | 0.5005 (5) | 0.3119 (9) | 0.3216 (7) | 0.042 (4) | 0.50 |
| H14 | 0.4989 | 0.3238 | 0.3741 | 0.050* | 0.50 |
| C15 | 0.5062 (14) | 0.2333 (16) | 0.2955 (15) | 0.069 (8) | 0.50 |
| H15 | 0.5100 | 0.1917 | 0.3309 | 0.083* | 0.50 |
| C16 | 0.5063 (13) | 0.2180 (14) | 0.2221 (13) | 0.071 (11) | 0.50 |
| H16 | 0.5137 | 0.1653 | 0.2068 | 0.086* | 0.50 |
| C17 | 0.4967 (8) | 0.2723 (10) | 0.1675 (10) | 0.053 (4) | 0.50 |
| H17 | 0.4909 | 0.2590 | 0.1157 | 0.064* | 0.50 |
| C18 | 0.4960 (6) | 0.3492 (8) | 0.1932 (8) | 0.036 (3) | 0.50 |
| H18 | 0.4944 | 0.3900 | 0.1567 | 0.043* | 0.50 |
| C31 | 0.83151 (19) | 0.9095 (2) | 0.3872 (2) | 0.0461 (9) | |
| H31A | 0.8715 | 0.8762 | 0.3983 | 0.055* | |
| H31B | 0.8466 | 0.9510 | 0.3519 | 0.055* | |
| C32 | 0.8094 (3) | 0.9481 (2) | 0.4600 (2) | 0.0624 (12) | |
| H32A | 0.8022 | 0.9081 | 0.4998 | 0.075* | |
| H32B | 0.7662 | 0.9768 | 0.4522 | 0.075* | |
| C33 | 0.8663 (3) | 1.0058 (3) | 0.4843 (3) | 0.0884 (18) | |
| H33A | 0.9106 | 0.9781 | 0.4842 | 0.106* | |
| H33B | 0.8691 | 1.0492 | 0.4472 | 0.106* | |
| C34 | 0.8533 (4) | 1.0384 (4) | 0.5607 (4) | 0.126 (3) | |
| H34A | 0.8894 | 1.0756 | 0.5738 | 0.189* | |
| H34B | 0.8526 | 0.9957 | 0.5979 | 0.189* | |
| H34C | 0.8093 | 1.0653 | 0.5610 | 0.189* | |
| C41 | 0.71599 (18) | 0.9076 (2) | 0.3270 (2) | 0.0435 (8) | |
| H41A | 0.6847 | 0.8740 | 0.2974 | 0.052* | |
| H41B | 0.6924 | 0.9220 | 0.3745 | 0.052* | |
| C42 | 0.7292 (2) | 0.9821 (3) | 0.2821 (3) | 0.0661 (12) | |
| H42A | 0.7484 | 0.9682 | 0.2320 | 0.079* | |
| H42B | 0.7631 | 1.0145 | 0.3091 | 0.079* | |
| C43 | 0.6643 (2) | 1.0294 (3) | 0.2709 (3) | 0.0671 (12) | |
| H43A | 0.6413 | 1.0358 | 0.3203 | 0.081* | |
| H43B | 0.6763 | 1.0823 | 0.2520 | 0.081* | |
| C44 | 0.6163 (3) | 0.9918 (3) | 0.2166 (3) | 0.0921 (18) | |
| H44A | 0.6005 | 0.9419 | 0.2374 | 0.138* | |
| H44B | 0.6397 | 0.9824 | 0.1684 | 0.138* | |
| H44C | 0.5773 | 1.0265 | 0.2081 | 0.138* | |
| C51 | 0.81340 (17) | 0.8282 (2) | 0.27666 (19) | 0.0421 (8) | |
| H51A | 0.8259 | 0.8730 | 0.2439 | 0.051* | |
| H51B | 0.8561 | 0.8017 | 0.2919 | 0.051* | |
| C52 | 0.77127 (18) | 0.7716 (3) | 0.2311 (2) | 0.0513 (10) | |
| H52A | 0.7599 | 0.7256 | 0.2629 | 0.062* | |
| H52B | 0.7281 | 0.7973 | 0.2163 | 0.062* | |
| C53 | 0.8079 (2) | 0.7444 (3) | 0.1612 (2) | 0.0544 (10) | |
| H53A | 0.8514 | 0.7194 | 0.1759 | 0.065* | |
| H53B | 0.8185 | 0.7902 | 0.1289 | 0.065* | |
| C54 | 0.7654 (2) | 0.6867 (3) | 0.1168 (2) | 0.0610 (12) | |
| H54B | 0.7581 | 0.6394 | 0.1472 | 0.091* | |
| H54A | 0.7891 | 0.6728 | 0.0699 | 0.091* | |
| H54C | 0.7214 | 0.7104 | 0.1045 | 0.091* | |
| C61 | 0.75488 (19) | 0.7932 (2) | 0.39740 (19) | 0.0416 (8) | |
| H61A | 0.7305 | 0.8158 | 0.4413 | 0.050* | |
| H61B | 0.7219 | 0.7610 | 0.3687 | 0.050* | |
| C62 | 0.8101 (2) | 0.7405 (2) | 0.4264 (2) | 0.0526 (10) | |
| H62A | 0.8399 | 0.7702 | 0.4613 | 0.063* | |
| H62B | 0.8383 | 0.7220 | 0.3835 | 0.063* | |
| C63 | 0.7804 (3) | 0.6709 (3) | 0.4672 (3) | 0.0642 (12) | |
| H63A | 0.7573 | 0.6363 | 0.4303 | 0.077* | |
| H63B | 0.7459 | 0.6893 | 0.5041 | 0.077* | |
| C64 | 0.8348 (3) | 0.6252 (3) | 0.5082 (4) | 0.103 (2) | |
| H64A | 0.8145 | 0.5792 | 0.5324 | 0.155* | |
| H64B | 0.8559 | 0.6585 | 0.5469 | 0.155* | |
| H64C | 0.8696 | 0.6082 | 0.4720 | 0.155* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| V1 | 0.0297 (3) | 0.0336 (3) | 0.0337 (3) | −0.0011 (2) | 0.0004 (2) | 0.0004 (2) |
| O1 | 0.0365 (12) | 0.0364 (13) | 0.0517 (14) | −0.0064 (10) | 0.0022 (10) | 0.0036 (11) |
| N1 | 0.0406 (14) | 0.0392 (17) | 0.0336 (14) | −0.0022 (12) | 0.0016 (12) | 0.0026 (13) |
| C1 | 0.053 (2) | 0.035 (2) | 0.0334 (18) | 0.0023 (16) | 0.0016 (16) | −0.0028 (15) |
| O2 | 0.0375 (12) | 0.0493 (15) | 0.0393 (13) | 0.0053 (11) | −0.0024 (10) | 0.0027 (11) |
| C2 | 0.0428 (18) | 0.0383 (19) | 0.0276 (16) | −0.0008 (15) | −0.0022 (14) | −0.0006 (14) |
| O3 | 0.0356 (12) | 0.0419 (14) | 0.0394 (13) | 0.0047 (10) | 0.0023 (10) | 0.0058 (11) |
| O4 | 0.0429 (13) | 0.0509 (15) | 0.0385 (13) | 0.0107 (11) | 0.0010 (11) | −0.0047 (12) |
| O5 | 0.0337 (11) | 0.0356 (13) | 0.0281 (11) | 0.0032 (9) | −0.0021 (9) | −0.0001 (10) |
| C5 | 0.0346 (16) | 0.044 (2) | 0.0318 (16) | 0.0017 (15) | 0.0003 (13) | 0.0001 (15) |
| O6 | 0.0731 (18) | 0.0633 (18) | 0.0304 (13) | 0.0188 (15) | 0.0026 (12) | −0.0054 (12) |
| C6 | 0.052 (2) | 0.042 (2) | 0.0385 (19) | 0.0011 (17) | −0.0006 (16) | 0.0014 (16) |
| C7 | 0.065 (3) | 0.043 (2) | 0.055 (2) | 0.0013 (19) | 0.006 (2) | −0.0002 (19) |
| O7 | 0.0360 (14) | 0.083 (3) | 0.111 (3) | −0.0040 (15) | 0.0001 (16) | −0.031 (2) |
| C8 | 0.057 (2) | 0.054 (3) | 0.059 (2) | 0.011 (2) | 0.002 (2) | 0.019 (2) |
| O8 | 0.055 (2) | 0.037 (2) | 0.062 (2) | 0.000 | −0.0204 (18) | 0.000 |
| C9 | 0.059 (2) | 0.064 (3) | 0.046 (2) | 0.007 (2) | −0.0118 (19) | 0.013 (2) |
| C10 | 0.058 (2) | 0.054 (2) | 0.039 (2) | 0.0027 (19) | −0.0101 (17) | 0.0029 (18) |
| C11 | 0.035 (3) | 0.038 (4) | 0.072 (5) | 0.004 (3) | −0.009 (4) | 0.002 (4) |
| C12 | 0.122 (9) | 0.063 (6) | 0.043 (5) | −0.022 (6) | −0.004 (5) | 0.003 (4) |
| C13 | 0.059 (5) | 0.029 (4) | 0.022 (12) | −0.011 (4) | −0.014 (10) | 0.006 (4) |
| C14 | 0.037 (4) | 0.060 (14) | 0.027 (7) | −0.011 (9) | 0.001 (3) | 0.032 (10) |
| C15 | 0.068 (11) | 0.085 (18) | 0.055 (13) | −0.027 (11) | −0.006 (11) | 0.047 (10) |
| C16 | 0.049 (10) | 0.041 (6) | 0.12 (3) | 0.007 (8) | 0.011 (12) | −0.013 (9) |
| C17 | 0.053 (6) | 0.075 (13) | 0.032 (8) | 0.015 (8) | 0.010 (5) | −0.015 (8) |
| C18 | 0.040 (5) | 0.045 (9) | 0.022 (9) | 0.001 (6) | 0.006 (5) | −0.004 (6) |
| C31 | 0.053 (2) | 0.040 (2) | 0.044 (2) | −0.0088 (17) | −0.0102 (17) | 0.0015 (17) |
| C32 | 0.098 (3) | 0.043 (2) | 0.046 (2) | −0.001 (2) | −0.009 (2) | −0.0035 (19) |
| C33 | 0.120 (4) | 0.061 (3) | 0.085 (4) | 0.018 (3) | −0.052 (3) | −0.020 (3) |
| C34 | 0.194 (7) | 0.080 (4) | 0.104 (5) | 0.036 (5) | −0.076 (5) | −0.031 (4) |
| C41 | 0.0414 (18) | 0.044 (2) | 0.045 (2) | 0.0002 (16) | 0.0034 (15) | 0.0076 (17) |
| C42 | 0.070 (3) | 0.061 (3) | 0.068 (3) | −0.002 (2) | 0.002 (2) | 0.026 (2) |
| C43 | 0.071 (3) | 0.060 (3) | 0.071 (3) | 0.000 (2) | −0.007 (2) | 0.026 (2) |
| C44 | 0.122 (5) | 0.069 (4) | 0.085 (4) | 0.019 (3) | −0.032 (3) | 0.003 (3) |
| C51 | 0.0362 (17) | 0.053 (2) | 0.0367 (18) | −0.0030 (16) | 0.0037 (15) | −0.0004 (17) |
| C52 | 0.0383 (18) | 0.070 (3) | 0.046 (2) | −0.0120 (18) | 0.0031 (16) | −0.009 (2) |
| C53 | 0.050 (2) | 0.077 (3) | 0.036 (2) | −0.012 (2) | 0.0030 (16) | −0.004 (2) |
| C54 | 0.057 (2) | 0.092 (3) | 0.0346 (19) | −0.014 (2) | 0.0040 (17) | −0.010 (2) |
| C61 | 0.051 (2) | 0.038 (2) | 0.0359 (17) | −0.0039 (16) | 0.0065 (16) | 0.0026 (15) |
| C62 | 0.066 (2) | 0.041 (2) | 0.050 (2) | 0.0069 (19) | 0.0015 (19) | 0.0053 (18) |
| C63 | 0.091 (3) | 0.038 (2) | 0.064 (3) | 0.008 (2) | 0.006 (2) | 0.007 (2) |
| C64 | 0.120 (5) | 0.063 (3) | 0.128 (5) | 0.028 (3) | 0.004 (4) | 0.040 (4) |
Geometric parameters (Å, º) top
| V1—O1 | 1.579 (2) | C31—C32 | 1.509 (6) |
| V1—O3 | 1.879 (2) | C31—H31A | 0.9900 |
| V1—O2 | 1.880 (2) | C31—H31B | 0.9900 |
| V1—O5 | 1.979 (2) | C32—C33 | 1.550 (7) |
| V1—O4 | 1.991 (2) | C32—H32A | 0.9900 |
| V1—O5i | 2.000 (2) | C32—H32B | 0.9900 |
| N1—C61 | 1.516 (4) | C33—C34 | 1.484 (8) |
| N1—C51 | 1.516 (4) | C33—H33A | 0.9900 |
| N1—C31 | 1.518 (4) | C33—H33B | 0.9900 |
| N1—C41 | 1.523 (4) | C34—H34A | 0.9800 |
| C1—O6 | 1.221 (4) | C34—H34B | 0.9800 |
| C1—O4 | 1.290 (4) | C34—H34C | 0.9800 |
| C1—C2 | 1.517 (5) | C41—C42 | 1.521 (5) |
| O2—O3 | 1.432 (3) | C41—H41A | 0.9900 |
| C2—O5 | 1.410 (4) | C41—H41B | 0.9900 |
| C2—C5 | 1.502 (5) | C42—C43 | 1.523 (6) |
| C2—H2 | 1.0000 | C42—H42A | 0.9900 |
| O5—V1i | 2.000 (2) | C42—H42B | 0.9900 |
| C5—C6 | 1.382 (5) | C43—C44 | 1.491 (7) |
| C5—C10 | 1.396 (5) | C43—H43A | 0.9900 |
| C6—C7 | 1.378 (5) | C43—H43B | 0.9900 |
| C6—H6 | 0.9500 | C44—H44A | 0.9800 |
| C7—C8 | 1.378 (6) | C44—H44B | 0.9800 |
| C7—H7 | 0.9500 | C44—H44C | 0.9800 |
| O7—C11i | 1.378 (8) | C51—C52 | 1.505 (5) |
| O7—C12 | 1.576 (12) | C51—H51A | 0.9900 |
| O7—H7O | 0.86 (8) | C51—H51B | 0.9900 |
| C8—C9 | 1.371 (6) | C52—C53 | 1.503 (5) |
| C8—H8 | 0.9500 | C52—H52A | 0.9900 |
| O8—C11i | 1.216 (8) | C52—H52B | 0.9900 |
| O8—C11 | 1.216 (8) | C53—C54 | 1.512 (6) |
| C9—C10 | 1.371 (6) | C53—H53A | 0.9900 |
| C9—H9 | 0.9500 | C53—H53B | 0.9900 |
| C10—H10 | 0.9500 | C54—H54B | 0.9800 |
| C11—O7i | 1.378 (8) | C54—H54A | 0.9800 |
| C11—C12 | 1.501 (12) | C54—H54C | 0.9800 |
| C12—C13 | 1.541 (15) | C61—C62 | 1.501 (5) |
| C12—H12 | 1.0000 | C61—H61A | 0.9900 |
| C13—C18 | 1.362 (16) | C61—H61B | 0.9900 |
| C13—C14 | 1.372 (14) | C62—C63 | 1.507 (6) |
| C14—C15 | 1.42 (3) | C62—H62A | 0.9900 |
| C14—H14 | 0.9500 | C62—H62B | 0.9900 |
| C15—C16 | 1.32 (3) | C63—C64 | 1.510 (7) |
| C15—H15 | 0.9500 | C63—H63A | 0.9900 |
| C16—C17 | 1.35 (4) | C63—H63B | 0.9900 |
| C16—H16 | 0.9500 | C64—H64A | 0.9800 |
| C17—C18 | 1.390 (18) | C64—H64B | 0.9800 |
| C17—H17 | 0.9500 | C64—H64C | 0.9800 |
| C18—H18 | 0.9500 | | |
| | | |
| O1—V1—O3 | 105.87 (12) | C31—C32—C33 | 107.8 (4) |
| O1—V1—O2 | 102.67 (12) | C31—C32—H32A | 110.2 |
| O3—V1—O2 | 44.80 (10) | C33—C32—H32A | 110.2 |
| O1—V1—O5 | 100.65 (11) | C31—C32—H32B | 110.2 |
| O3—V1—O5 | 146.27 (10) | C33—C32—H32B | 110.2 |
| O2—V1—O5 | 145.84 (10) | H32A—C32—H32B | 108.5 |
| O1—V1—O4 | 98.80 (12) | C34—C33—C32 | 111.4 (6) |
| O3—V1—O4 | 78.20 (10) | C34—C33—H33A | 109.3 |
| O2—V1—O4 | 122.53 (11) | C32—C33—H33A | 109.3 |
| O5—V1—O4 | 77.47 (9) | C34—C33—H33B | 109.3 |
| O1—V1—O5i | 99.78 (11) | C32—C33—H33B | 109.3 |
| O3—V1—O5i | 124.43 (10) | H33A—C33—H33B | 108.0 |
| O2—V1—O5i | 82.09 (10) | C33—C34—H34A | 109.5 |
| O5—V1—O5i | 69.57 (10) | C33—C34—H34B | 109.5 |
| O4—V1—O5i | 144.53 (10) | H34A—C34—H34B | 109.5 |
| C61—N1—C51 | 110.8 (3) | C33—C34—H34C | 109.5 |
| C61—N1—C31 | 110.9 (3) | H34A—C34—H34C | 109.5 |
| C51—N1—C31 | 106.0 (3) | H34B—C34—H34C | 109.5 |
| C61—N1—C41 | 106.7 (2) | C42—C41—N1 | 115.7 (3) |
| C51—N1—C41 | 111.2 (3) | C42—C41—H41A | 108.4 |
| C31—N1—C41 | 111.2 (3) | N1—C41—H41A | 108.4 |
| O6—C1—O4 | 124.5 (3) | C42—C41—H41B | 108.4 |
| O6—C1—C2 | 121.3 (3) | N1—C41—H41B | 108.4 |
| O4—C1—C2 | 114.2 (3) | H41A—C41—H41B | 107.4 |
| O3—O2—V1 | 67.55 (13) | C41—C42—C43 | 111.6 (3) |
| O5—C2—C5 | 110.7 (3) | C41—C42—H42A | 109.3 |
| O5—C2—C1 | 107.9 (3) | C43—C42—H42A | 109.3 |
| C5—C2—C1 | 112.0 (3) | C41—C42—H42B | 109.3 |
| O5—C2—H2 | 108.7 | C43—C42—H42B | 109.3 |
| C5—C2—H2 | 108.7 | H42A—C42—H42B | 108.0 |
| C1—C2—H2 | 108.7 | C44—C43—C42 | 112.6 (5) |
| O2—O3—V1 | 67.65 (13) | C44—C43—H43A | 109.1 |
| C1—O4—V1 | 119.9 (2) | C42—C43—H43A | 109.1 |
| C2—O5—V1 | 119.08 (19) | C44—C43—H43B | 109.1 |
| C2—O5—V1i | 128.5 (2) | C42—C43—H43B | 109.1 |
| V1—O5—V1i | 105.58 (10) | H43A—C43—H43B | 107.8 |
| C6—C5—C10 | 118.9 (3) | C43—C44—H44A | 109.5 |
| C6—C5—C2 | 121.3 (3) | C43—C44—H44B | 109.5 |
| C10—C5—C2 | 119.7 (3) | H44A—C44—H44B | 109.5 |
| C7—C6—C5 | 120.1 (3) | C43—C44—H44C | 109.5 |
| C7—C6—H6 | 119.9 | H44A—C44—H44C | 109.5 |
| C5—C6—H6 | 119.9 | H44B—C44—H44C | 109.5 |
| C6—C7—C8 | 120.4 (4) | C52—C51—N1 | 114.7 (3) |
| C6—C7—H7 | 119.8 | C52—C51—H51A | 108.6 |
| C8—C7—H7 | 119.8 | N1—C51—H51A | 108.6 |
| C11i—O7—C12 | 51.6 (5) | C52—C51—H51B | 108.6 |
| C11i—O7—H7O | 87 (5) | N1—C51—H51B | 108.6 |
| C12—O7—H7O | 115 (5) | H51A—C51—H51B | 107.6 |
| C9—C8—C7 | 119.9 (4) | C53—C52—C51 | 112.0 (3) |
| C9—C8—H8 | 120.1 | C53—C52—H52A | 109.2 |
| C7—C8—H8 | 120.1 | C51—C52—H52A | 109.2 |
| C11i—O8—C11 | 50.6 (7) | C53—C52—H52B | 109.2 |
| C8—C9—C10 | 120.3 (4) | C51—C52—H52B | 109.2 |
| C8—C9—H9 | 119.8 | H52A—C52—H52B | 107.9 |
| C10—C9—H9 | 119.8 | C52—C53—C54 | 111.3 (3) |
| C9—C10—C5 | 120.4 (4) | C52—C53—H53A | 109.4 |
| C9—C10—H10 | 119.8 | C54—C53—H53A | 109.4 |
| C5—C10—H10 | 119.8 | C52—C53—H53B | 109.4 |
| O8—C11—O7i | 137.4 (7) | C54—C53—H53B | 109.4 |
| O8—C11—C12 | 112.9 (7) | H53A—C53—H53B | 108.0 |
| O7i—C11—C12 | 109.2 (7) | C53—C54—H54B | 109.5 |
| C11—C12—C13 | 114.1 (9) | C53—C54—H54A | 109.5 |
| C11—C12—O7 | 99.2 (7) | H54B—C54—H54A | 109.5 |
| C13—C12—O7 | 111.2 (8) | C53—C54—H54C | 109.5 |
| C11—C12—H12 | 110.6 | H54B—C54—H54C | 109.5 |
| C13—C12—H12 | 110.6 | H54A—C54—H54C | 109.5 |
| O7—C12—H12 | 110.6 | C62—C61—N1 | 115.1 (3) |
| C18—C13—C14 | 119.2 (12) | C62—C61—H61A | 108.5 |
| C18—C13—C12 | 120.1 (10) | N1—C61—H61A | 108.5 |
| C14—C13—C12 | 119.8 (8) | C62—C61—H61B | 108.5 |
| C13—C14—C15 | 117.1 (11) | N1—C61—H61B | 108.5 |
| C13—C14—H14 | 121.4 | H61A—C61—H61B | 107.5 |
| C15—C14—H14 | 121.4 | C61—C62—C63 | 110.7 (4) |
| C16—C15—C14 | 120 (2) | C61—C62—H62A | 109.5 |
| C16—C15—H15 | 119.8 | C63—C62—H62A | 109.5 |
| C14—C15—H15 | 119.8 | C61—C62—H62B | 109.5 |
| C15—C16—C17 | 124 (3) | C63—C62—H62B | 109.5 |
| C15—C16—H16 | 117.8 | H62A—C62—H62B | 108.1 |
| C17—C16—H16 | 117.8 | C62—C63—C64 | 111.2 (4) |
| C16—C17—C18 | 114.5 (19) | C62—C63—H63A | 109.4 |
| C16—C17—H17 | 122.7 | C64—C63—H63A | 109.4 |
| C18—C17—H17 | 122.7 | C62—C63—H63B | 109.4 |
| C13—C18—C17 | 123.9 (17) | C64—C63—H63B | 109.4 |
| C13—C18—H18 | 118.0 | H63A—C63—H63B | 108.0 |
| C17—C18—H18 | 118.0 | C63—C64—H64A | 109.5 |
| C32—C31—N1 | 116.5 (3) | C63—C64—H64B | 109.5 |
| C32—C31—H31A | 108.2 | H64A—C64—H64B | 109.5 |
| N1—C31—H31A | 108.2 | C63—C64—H64C | 109.5 |
| C32—C31—H31B | 108.2 | H64A—C64—H64C | 109.5 |
| N1—C31—H31B | 108.2 | H64B—C64—H64C | 109.5 |
| H31A—C31—H31B | 107.3 | | |
| Symmetry code: (i) −x+1, y, −z+1/2. |
Experimental details
| Crystal data |
| Chemical formula | (C16H36N)2[V2O2(O2)2(C8H6O3)2]·C8H8O3 |
| Mr | 1135.20 |
| Crystal system, space group | Orthorhombic, Pbcn |
| Temperature (K) | 150 |
| a, b, c (Å) | 19.657 (12), 17.066 (3), 17.670 (5) |
| V (Å3) | 5928 (4) |
| Z | 4 |
| Radiation type | Mo Kα |
| µ (mm−1) | 0.38 |
| Crystal size (mm) | 0.4 × 0.3 × 0.3 |
| |
| Data collection |
| Diffractometer | 4-circle κ-axis Kuma KM-4
diffractometer with Oxford Cryosytems Cryostream
cooler |
| Absorption correction | – |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 5680, 5210, 3329 |
| Rint | 0.023 |
| (sin θ/λ)max (Å−1) | 0.596 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.052, 0.168, 1.03 |
| No. of reflections | 5210 |
| No. of parameters | 383 |
| H-atom treatment | H atoms treated by a mixture of independent and constrained refinement |
| Δρmax, Δρmin (e Å−3) | 0.66, −0.40 |
Selected geometric parameters (Å, º) top| V1—O1 | 1.579 (2) | V1—O5i | 2.000 (2) |
| V1—O3 | 1.879 (2) | C1—O4 | 1.290 (4) |
| V1—O2 | 1.880 (2) | C1—C2 | 1.517 (5) |
| V1—O5 | 1.979 (2) | O2—O3 | 1.432 (3) |
| V1—O4 | 1.991 (2) | C2—O5 | 1.410 (4) |
| | | |
| O1—V1—O3 | 105.87 (12) | O2—V1—O5i | 82.09 (10) |
| O1—V1—O2 | 102.67 (12) | O5—V1—O5i | 69.57 (10) |
| O3—V1—O2 | 44.80 (10) | O4—V1—O5i | 144.53 (10) |
| O1—V1—O5 | 100.65 (11) | O4—C1—C2 | 114.2 (3) |
| O3—V1—O5 | 146.27 (10) | O3—O2—V1 | 67.55 (13) |
| O2—V1—O5 | 145.84 (10) | C5—C2—C1 | 112.0 (3) |
| O1—V1—O4 | 98.80 (12) | O2—O3—V1 | 67.65 (13) |
| O3—V1—O4 | 78.20 (10) | C1—O4—V1 | 119.9 (2) |
| O2—V1—O4 | 122.53 (11) | C2—O5—V1 | 119.08 (19) |
| O5—V1—O4 | 77.47 (9) | C2—O5—V1i | 128.5 (2) |
| O1—V1—O5i | 99.78 (11) | V1—O5—V1i | 105.58 (10) |
| O3—V1—O5i | 124.43 (10) | | |
| Symmetry code: (i) −x+1, y, −z+1/2. |
![[Figure 1]](http://journals.iucr.org/c/issues/2000/02/00/cf1306/cf1306fig1thm.gif)

 journal menu
journal menu












Vanadium compounds are important in biochemistry and biology in many ways (Sigel & Sigel, 1995; Djordjevic et al., 1995). Peroxo complexes form a new category of compounds, with effective insulin-mimetic activity (Sigel & Sigel, 1995). Vanadium peroxo complexes are formed as intermediates in oxidation reactions catalysed by haloperoxidase, which contains V in the active site (Messerschmidt et al., 1997). This enzyme has been found in a variety of seaweeds (Wever et al., 1991). Anions of α-hydroxycarboxylic acids also represent an interesting class of biogenic ligands. Several dinuclear vanadium monoperoxo complexes with biogenic heteroligands such as citrate (Djordjevic et al., 1989), malate (Djordjevic et al., 1995), and tartrate ions (Schwendt et al., 1998) have been prepared recently. We report here a related complex with the mandelate dianion as ligand, bis(tetra-n-butylammonium) bis[(mandelato)oxo(peroxo)vanadate(V)] mandelic acid solvate, (I). \scheme
The crystal structure of (I) consists of dimeric anions with crystallographic twofold rotation symmetry (Fig. 1), unexceptional (Bu4N)+ cations and one molecule of neutral mandelic acid per dimeric anion. The component cations, anions and mandelic acid molecules are held together by electrostatic forces and hydrogen bonds.
The two V atoms in the anion are doubly bridged by the deprotonated hydroxyl O atoms of two mandelato ligands, O5 and O5i [symmetry code: (i) 1 − x, y, 1/2 − z], to give a central V2O2 ring. This ring is not planar, but has a dihedral angle of folding of 151.79 (8)° about the O5···O5i line. Each mandelato anion also coordinates terminally to one V atom through its carboxylate group to form five-membered chelate rings. The primary coordination of V is distorted pentagonal pyramidal by six O atoms. The equatorial plane involves the peroxo group [O2 and O3, with O—O = 1.432 (3) Å], the two bridging O atoms and the carboxylate atom O4, which have a maximum deviation of 0.10 (7) Å from this plane. The axial position is occupied by the doubly-bonded oxo ligand O1, 1.89 (7) Å out of the equatorial plane, with V—O1 = 1.578 (2) Å. The V atom is displaced 0.32 (1) Å out of the equatorial plane, towards the axial oxo ligand.
The neutral mandelic acid molecule is disordered about the twofold rotation axis in such a way that the O atoms coincide in the two orientations. One carboxylate O atom, on the twofold axis, is coordinated to the two V atoms in an axial position opposite the oxo ligand, the V—O8 distance being 2.550 (3) Å.