The distinctive feature of the crystal structure of 2,6-diphenylpyridine-4-carboxylic acid, C
18H
13NO
2, is the formation of intermolecular O—H

O hydrogen bonds that lead to the formation of centrosymmetric cyclic dimers with
R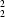
(8) topology. Molecules related by translation along the
b axis exhibit strong π–π stacking of aromatic rings, with an average interplanar distance of 3.3 Å.
Supporting information
CCDC reference: 180165
The title compound was prepared in one step from 3-benzoylacrilic acid and
N-phenacylpyridinium bromide in the presence of excess ammonium acetate
(Blumbergs et al., 1972). The compound was recrystallized from hot
acetic acid affording pale yellow crystals.
Data collection: XPREP (Bruker, 1997); cell refinement: XPREP (Bruker, 1997); data reduction: XPREP (Bruker, 1997); program(s) used to solve structure: SHELXS97 (Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: XP (Bruker, 1997); software used to prepare material for publication: SHELXL97 (Sheldrick, 1997).
Crystal data top
| C18H13NO2 | F(000) = 576 |
| Mr = 275.29 | Dx = 1.32 Mg m−3 |
| Monoclinic, P21/n | Mo Kα radiation, λ = 0.71073 Å |
| a = 16.524 (4) Å | Cell parameters from 25 reflections |
| b = 5.2898 (9) Å | θ = 8.0–12.9° |
| c = 17.021 (3) Å | µ = 0.09 mm−1 |
| β = 111.229 (17)° | T = 293 K |
| V = 1386.8 (5) Å3 | Block, pale yellow |
| Z = 4 | 0.23 × 0.20 × 0.17 mm |
Data collection top
Siemens R3m/V
diffractometer | Rint = 0.069 |
| Radiation source: X-ray tube | θmax = 25.1°, θmin = 1.5° |
| Graphite monochromator | h = 0→19 |
| θ/2θ scans | k = 0→6 |
| 2559 measured reflections | l = −20→18 |
| 2469 independent reflections | 2 standard reflections every 48 min |
| 980 reflections with I > 2σ(I) | intensity decay: no decay, variation 0.5% |
Refinement top
| Refinement on F2 | 0 restraints |
| Least-squares matrix: full | H atoms treated by a mixture of independent and constrained refinement |
| R[F2 > 2σ(F2)] = 0.045 | w = 1/[σ2(Fo2) + (0.025P)2]
where P = (Fo2 + 2Fc2)/3 |
| wR(F2) = 0.089 | (Δ/σ)max < 0.001 |
| S = 0.87 | Δρmax = 0.13 e Å−3 |
| 2469 reflections | Δρmin = −0.13 e Å−3 |
| 190 parameters | |
Crystal data top
| C18H13NO2 | V = 1386.8 (5) Å3 |
| Mr = 275.29 | Z = 4 |
| Monoclinic, P21/n | Mo Kα radiation |
| a = 16.524 (4) Å | µ = 0.09 mm−1 |
| b = 5.2898 (9) Å | T = 293 K |
| c = 17.021 (3) Å | 0.23 × 0.20 × 0.17 mm |
| β = 111.229 (17)° | |
Data collection top
Siemens R3m/V
diffractometer | Rint = 0.069 |
| 2559 measured reflections | 2 standard reflections every 48 min |
| 2469 independent reflections | intensity decay: no decay, variation 0.5% |
| 980 reflections with I > 2σ(I) | |
Refinement top
| R[F2 > 2σ(F2)] = 0.045 | 0 restraints |
| wR(F2) = 0.089 | H atoms treated by a mixture of independent and constrained refinement |
| S = 0.87 | Δρmax = 0.13 e Å−3 |
| 2469 reflections | Δρmin = −0.13 e Å−3 |
| 190 parameters | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| N1 | 0.92467 (13) | 0.1108 (4) | 0.69830 (13) | 0.0515 (6) | |
| O1 | 1.05832 (11) | 0.7793 (4) | 0.57999 (10) | 0.0709 (6) | |
| H1 | 1.0619 | 0.9040 | 0.5528 | 0.106* | |
| O2 | 0.91707 (11) | 0.8373 (4) | 0.50817 (11) | 0.0674 (6) | |
| C2 | 0.85978 (16) | 0.2316 (5) | 0.63842 (16) | 0.0483 (7) | |
| C3 | 0.87627 (16) | 0.4328 (5) | 0.59355 (16) | 0.0518 (8) | |
| H3 | 0.8307 | 0.5145 | 0.5523 | 0.062* | |
| C4 | 0.96062 (17) | 0.5099 (5) | 0.61078 (16) | 0.0481 (7) | |
| C5 | 1.02650 (16) | 0.3905 (5) | 0.67265 (16) | 0.0542 (8) | |
| H5 | 1.0836 | 0.4438 | 0.6863 | 0.065* | |
| C6 | 1.00715 (16) | 0.1875 (5) | 0.71508 (16) | 0.0513 (8) | |
| C7 | 0.97728 (18) | 0.7241 (5) | 0.56290 (17) | 0.0527 (8) | |
| C8 | 1.07541 (17) | 0.0392 (5) | 0.78092 (16) | 0.0518 (7) | |
| C9 | 1.16336 (18) | 0.0702 (7) | 0.79678 (18) | 0.0833 (11) | |
| H9 | 1.1815 | 0.1914 | 0.7671 | 0.100* | |
| C10 | 1.22390 (19) | −0.0766 (7) | 0.8559 (2) | 0.0853 (11) | |
| H10 | 1.2825 | −0.0517 | 0.8658 | 0.102* | |
| C11 | 1.20051 (19) | −0.2564 (6) | 0.90019 (17) | 0.0743 (10) | |
| H11 | 1.2422 | −0.3560 | 0.9394 | 0.089* | |
| C12 | 1.11401 (19) | −0.2885 (7) | 0.88584 (19) | 0.0910 (11) | |
| H12 | 1.0964 | −0.4100 | 0.9157 | 0.109* | |
| C13 | 1.05300 (18) | −0.1394 (6) | 0.8267 (2) | 0.0764 (10) | |
| H13 | 0.9946 | −0.1623 | 0.8181 | 0.092* | |
| C14 | 0.77008 (16) | 0.1425 (5) | 0.62523 (16) | 0.0490 (7) | |
| C15 | 0.75731 (16) | −0.0738 (6) | 0.66479 (16) | 0.0583 (8) | |
| H15 | 0.8051 | −0.1684 | 0.6974 | 0.070* | |
| C16 | 0.67528 (18) | −0.1521 (6) | 0.65700 (17) | 0.0688 (9) | |
| H16 | 0.6680 | −0.2968 | 0.6848 | 0.083* | |
| C17 | 0.60398 (19) | −0.0149 (7) | 0.60776 (19) | 0.0730 (10) | |
| H17 | 0.5484 | −0.0671 | 0.6022 | 0.088* | |
| C18 | 0.61493 (18) | 0.1974 (7) | 0.56717 (19) | 0.0743 (9) | |
| H18 | 0.5668 | 0.2902 | 0.5341 | 0.089* | |
| C19 | 0.69789 (17) | 0.2752 (6) | 0.57512 (17) | 0.0639 (8) | |
| H19 | 0.7048 | 0.4181 | 0.5464 | 0.077* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| N1 | 0.0433 (13) | 0.0622 (17) | 0.0502 (15) | 0.0008 (13) | 0.0184 (11) | 0.0015 (13) |
| O1 | 0.0584 (12) | 0.0761 (15) | 0.0817 (14) | −0.0043 (12) | 0.0294 (10) | 0.0206 (13) |
| O2 | 0.0562 (12) | 0.0744 (15) | 0.0683 (14) | −0.0015 (11) | 0.0185 (10) | 0.0202 (12) |
| C2 | 0.0446 (16) | 0.050 (2) | 0.0515 (18) | −0.0025 (15) | 0.0190 (15) | −0.0066 (16) |
| C3 | 0.0473 (17) | 0.054 (2) | 0.0554 (19) | 0.0028 (15) | 0.0194 (14) | 0.0021 (16) |
| C4 | 0.0496 (17) | 0.0456 (19) | 0.0532 (19) | −0.0039 (15) | 0.0237 (15) | −0.0001 (16) |
| C5 | 0.0451 (16) | 0.062 (2) | 0.0576 (19) | −0.0012 (16) | 0.0203 (15) | 0.0026 (17) |
| C6 | 0.0451 (17) | 0.056 (2) | 0.0532 (19) | −0.0005 (15) | 0.0179 (15) | 0.0026 (16) |
| C7 | 0.0495 (17) | 0.057 (2) | 0.0544 (19) | −0.0057 (17) | 0.0225 (15) | −0.0024 (18) |
| C8 | 0.0503 (18) | 0.056 (2) | 0.0505 (18) | −0.0026 (16) | 0.0203 (15) | 0.0002 (16) |
| C9 | 0.0500 (18) | 0.117 (3) | 0.084 (3) | 0.004 (2) | 0.0257 (18) | 0.037 (2) |
| C10 | 0.0491 (18) | 0.122 (3) | 0.083 (3) | 0.014 (2) | 0.0212 (18) | 0.028 (2) |
| C11 | 0.062 (2) | 0.084 (3) | 0.067 (2) | 0.009 (2) | 0.0097 (17) | 0.010 (2) |
| C12 | 0.061 (2) | 0.107 (3) | 0.090 (3) | −0.008 (2) | 0.0096 (18) | 0.041 (2) |
| C13 | 0.0474 (18) | 0.092 (3) | 0.080 (2) | −0.0061 (19) | 0.0124 (17) | 0.027 (2) |
| C14 | 0.0439 (16) | 0.054 (2) | 0.0500 (18) | −0.0015 (15) | 0.0182 (14) | −0.0049 (16) |
| C15 | 0.0478 (18) | 0.067 (2) | 0.061 (2) | −0.0035 (16) | 0.0209 (15) | 0.0004 (18) |
| C16 | 0.0587 (19) | 0.082 (2) | 0.069 (2) | −0.014 (2) | 0.0260 (17) | −0.0033 (19) |
| C17 | 0.054 (2) | 0.086 (3) | 0.083 (3) | −0.010 (2) | 0.0297 (19) | −0.015 (2) |
| C18 | 0.0463 (18) | 0.077 (3) | 0.094 (3) | 0.0000 (18) | 0.0182 (17) | −0.004 (2) |
| C19 | 0.0524 (17) | 0.067 (2) | 0.070 (2) | −0.0020 (18) | 0.0191 (16) | 0.0050 (18) |
Geometric parameters (Å, º) top
| N1—C2 | 1.343 (3) | C10—C11 | 1.354 (4) |
| N1—C6 | 1.350 (3) | C10—H10 | 0.9300 |
| O1—C7 | 1.297 (3) | C11—C12 | 1.371 (3) |
| O1—H1 | 0.8200 | C11—H11 | 0.9300 |
| O2—C7 | 1.243 (3) | C12—C13 | 1.384 (4) |
| C2—C3 | 1.393 (3) | C12—H12 | 0.9300 |
| C2—C14 | 1.493 (3) | C13—H13 | 0.9300 |
| C3—C4 | 1.378 (3) | C14—C19 | 1.381 (3) |
| C3—H3 | 0.9300 | C14—C15 | 1.382 (3) |
| C4—C5 | 1.365 (3) | C15—C16 | 1.377 (3) |
| C4—C7 | 1.478 (3) | C15—H15 | 0.9300 |
| C5—C6 | 1.394 (3) | C16—C17 | 1.379 (4) |
| C5—H5 | 0.9300 | C16—H16 | 0.9300 |
| C6—C8 | 1.493 (3) | C17—C18 | 1.364 (4) |
| C8—C13 | 1.359 (3) | C17—H17 | 0.9300 |
| C8—C9 | 1.389 (3) | C18—C19 | 1.390 (3) |
| C9—C10 | 1.374 (4) | C18—H18 | 0.9300 |
| C9—H9 | 0.9300 | C19—H19 | 0.9300 |
| | | |
| C2—N1—C6 | 119.0 (2) | C9—C10—H10 | 119.1 |
| C7—O1—H1 | 109.5 | C10—C11—C12 | 118.5 (3) |
| N1—C2—C3 | 121.2 (2) | C10—C11—H11 | 120.7 |
| N1—C2—C14 | 116.1 (3) | C12—C11—H11 | 120.7 |
| C3—C2—C14 | 122.6 (3) | C11—C12—C13 | 119.8 (3) |
| C4—C3—C2 | 119.5 (3) | C11—C12—H12 | 120.1 |
| C4—C3—H3 | 120.2 | C13—C12—H12 | 120.1 |
| C2—C3—H3 | 120.2 | C8—C13—C12 | 122.4 (3) |
| C5—C4—C3 | 119.3 (3) | C8—C13—H13 | 118.8 |
| C5—C4—C7 | 121.6 (3) | C12—C13—H13 | 118.8 |
| C3—C4—C7 | 119.0 (3) | C19—C14—C15 | 118.1 (3) |
| C4—C5—C6 | 119.2 (2) | C19—C14—C2 | 121.4 (3) |
| C4—C5—H5 | 120.4 | C15—C14—C2 | 120.4 (3) |
| C6—C5—H5 | 120.4 | C16—C15—C14 | 121.4 (3) |
| N1—C6—C5 | 121.7 (2) | C16—C15—H15 | 119.3 |
| N1—C6—C8 | 115.6 (3) | C14—C15—H15 | 119.3 |
| C5—C6—C8 | 122.8 (2) | C15—C16—C17 | 119.6 (3) |
| O2—C7—O1 | 122.8 (3) | C15—C16—H16 | 120.2 |
| O2—C7—C4 | 121.6 (2) | C17—C16—H16 | 120.2 |
| O1—C7—C4 | 115.6 (3) | C18—C17—C16 | 120.0 (3) |
| C13—C8—C9 | 117.0 (3) | C18—C17—H17 | 120.0 |
| C13—C8—C6 | 120.5 (3) | C16—C17—H17 | 120.0 |
| C9—C8—C6 | 122.5 (3) | C17—C18—C19 | 120.1 (3) |
| C10—C9—C8 | 120.6 (3) | C17—C18—H18 | 120.0 |
| C10—C9—H9 | 119.7 | C19—C18—H18 | 120.0 |
| C8—C9—H9 | 119.7 | C14—C19—C18 | 120.7 (3) |
| C11—C10—C9 | 121.7 (3) | C14—C19—H19 | 119.6 |
| C11—C10—H10 | 119.1 | C18—C19—H19 | 119.6 |
| | | |
| C6—N1—C2—C3 | 0.2 (4) | C13—C8—C9—C10 | 0.5 (5) |
| C6—N1—C2—C14 | −178.0 (2) | C6—C8—C9—C10 | −177.7 (3) |
| N1—C2—C3—C4 | 0.0 (4) | C8—C9—C10—C11 | 0.5 (5) |
| C14—C2—C3—C4 | 178.1 (2) | C9—C10—C11—C12 | −0.9 (5) |
| C2—C3—C4—C5 | −1.2 (4) | C10—C11—C12—C13 | 0.4 (5) |
| C2—C3—C4—C7 | −179.8 (2) | C9—C8—C13—C12 | −1.0 (5) |
| C3—C4—C5—C6 | 2.1 (4) | C6—C8—C13—C12 | 177.2 (3) |
| C7—C4—C5—C6 | −179.2 (2) | C11—C12—C13—C8 | 0.6 (5) |
| C2—N1—C6—C5 | 0.8 (4) | N1—C2—C14—C19 | 170.0 (2) |
| C2—N1—C6—C8 | −178.5 (2) | C3—C2—C14—C19 | −8.2 (4) |
| C4—C5—C6—N1 | −2.0 (4) | N1—C2—C14—C15 | −8.6 (3) |
| C4—C5—C6—C8 | 177.3 (2) | C3—C2—C14—C15 | 173.3 (2) |
| C5—C4—C7—O2 | −178.0 (3) | C19—C14—C15—C16 | −2.0 (4) |
| C3—C4—C7—O2 | 0.7 (4) | C2—C14—C15—C16 | 176.6 (2) |
| C5—C4—C7—O1 | 4.1 (4) | C14—C15—C16—C17 | 1.0 (4) |
| C3—C4—C7—O1 | −177.2 (2) | C15—C16—C17—C18 | −0.1 (5) |
| N1—C6—C8—C13 | −7.6 (4) | C16—C17—C18—C19 | 0.2 (5) |
| C5—C6—C8—C13 | 173.0 (3) | C15—C14—C19—C18 | 2.1 (4) |
| N1—C6—C8—C9 | 170.5 (3) | C2—C14—C19—C18 | −176.4 (2) |
| C5—C6—C8—C9 | −8.9 (4) | C17—C18—C19—C14 | −1.3 (4) |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1—H1···O2i | 0.82 | 1.83 | 2.640 (3) | 172 |
| C11—H11···O2ii | 0.93 | 2.70 | 3.406 (3) | 134 |
| Symmetry codes: (i) −x+2, −y+2, −z+1; (ii) x+1/2, −y+1/2, z+1/2. |
Experimental details
| Crystal data |
| Chemical formula | C18H13NO2 |
| Mr | 275.29 |
| Crystal system, space group | Monoclinic, P21/n |
| Temperature (K) | 293 |
| a, b, c (Å) | 16.524 (4), 5.2898 (9), 17.021 (3) |
| β (°) | 111.229 (17) |
| V (Å3) | 1386.8 (5) |
| Z | 4 |
| Radiation type | Mo Kα |
| µ (mm−1) | 0.09 |
| Crystal size (mm) | 0.23 × 0.20 × 0.17 |
| |
| Data collection |
| Diffractometer | Siemens R3m/V
diffractometer |
| Absorption correction | – |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 2559, 2469, 980 |
| Rint | 0.069 |
| (sin θ/λ)max (Å−1) | 0.596 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.045, 0.089, 0.87 |
| No. of reflections | 2469 |
| No. of parameters | 190 |
| H-atom treatment | H atoms treated by a mixture of independent and constrained refinement |
| Δρmax, Δρmin (e Å−3) | 0.13, −0.13 |
Selected geometric parameters (Å, º) top| N1—C2 | 1.343 (3) | C3—C4 | 1.378 (3) |
| N1—C6 | 1.350 (3) | C4—C5 | 1.365 (3) |
| O1—C7 | 1.297 (3) | C4—C7 | 1.478 (3) |
| O2—C7 | 1.243 (3) | C5—C6 | 1.394 (3) |
| C2—C3 | 1.393 (3) | | |
| | | |
| O2—C7—O1 | 122.8 (3) | O1—C7—C4 | 115.6 (3) |
| O2—C7—C4 | 121.6 (2) | | |
| | | |
| C3—C4—C7—O2 | 0.7 (4) | N1—C6—C8—C13 | −7.6 (4) |
| C5—C4—C7—O1 | 4.1 (4) | N1—C2—C14—C15 | −8.6 (3) |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1—H1···O2i | 0.82 | 1.83 | 2.640 (3) | 172 |
| C11—H11···O2ii | 0.93 | 2.70 | 3.406 (3) | 134 |
| Symmetry codes: (i) −x+2, −y+2, −z+1; (ii) x+1/2, −y+1/2, z+1/2. |
 O hydrogen bonds that lead to the formation of centrosymmetric cyclic dimers with R
O hydrogen bonds that lead to the formation of centrosymmetric cyclic dimers with R
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2002/01/00/na1541/na1541fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2002/01/00/na1541/na1541fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/c/issues/2002/01/00/na1541/na1541fig3thm.gif)




The most common packing motif of carboxylic acids is based on the formation of cyclic dimers through a pair of O—H···O bonds (Leiserowitz, 1976). By contrast, the solid state chemistry of pyridinecarboxylic acids is dominated by the formation of an intermolecular O—H···N hydrogen-bond between the carboxylic group and the pyridine nitrogen atom of a second molecule (Takusagawa & Shimada, 1976; Wright & King, 1953). The latter H-bonding interaction can be described more accurately as occurring via O—H···N or +N—H···O- bonds depending on the tautomeric form present. Similar arguments also apply to other heterocyclic aromatic carboxylic acids such as the pyrazinic acid (Takusagawa et al., 1974) and the quinoline-4-carboxylic acid (Dobson & Gerkin, 1998). The directional nature of the O—H···N bonding interaction for the isonicotinic acid has been exploited in the promotion of highly oriented structures (O'Shea et al., 2001). On the other hand, when carboxylic groups occupy both α and α' positions of the pyridine ring - as in the dipicolinic acid - the nitrogen atom is not involved in head-to-tail interactions of the type described, although O—H···N hydrogen-bonds with a cocrystallized water molecule are still present (Takusagawa et al., 1973).
The molecular structure of (I) is shown in Figure 1. Geometric parameters are normal (Table 1) and compare well with those of other 2,6-diphenyl-pyridines (Krygowski et al., 1994; Silva et al., 1997). The whole molecule is nearly planar, as clearly shown by the values of the torsion angles reported in Table 1. Dihedral angles between the best-fit plane of the pyridine and those of the phenyl substituents are 8.7 (1)° (ring atoms C8,···,C13) and 9.8 (1)° (ring atoms C14,···,C19), respectively.
The supramolecular structure of (I) can be described in terms of π-π interactions and intermolecular H-bonding (Table 2). In the crystal packing the molecules are stacked along the b axis (Figure 2). The shortest interplanar distance of 2.919 (2) Å is found between the phenyl ring C14,···C19 and its translation-related one. The stacks are organized in a pseudo-herringbone fashion since the π-H interactions do not seem to be dominant like in a classical herringbone stacking mode (Gavezzotti & Desiraju, 1988; Andre et al., 1997). The dominant supramolecular interaction is the couple of O—H···O hydrogen bonds between centrosymmetric carboxylic acid groups (Figure 3). The corresponding graph-set is therefore R22(8) (Etter et al., 1990), which is typical for carboxyl dimers. In addition, a second motif with graph-set R46(44) can be identify in the crystal packing when weaker hydrogen bonds of the C—H···O type are considered (Fig. 3).