In both 2,5-dimethyl-6,7-dihydrobenzo[
h]pyrazolo[1,5-
a]quinazoline, C
16H
15N
3, (I), and 2-
tert-butyl-5-methyl-6,7-dihydrobenzo[
h]pyrazolo[1,5-
a]quinazoline, C
19H
21N
3, (II), which crystallizes with
Z' = 2 in the space group
P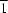
, the non-aromatic carbocyclic rings adopt screw-boat conformations. The molecules of (I) are linked into chains of rings by a combination of C-H

N and C-H

![[pi]](/logos/entities/pi_rmgif.gif)
(arene) hydrogen bonds, while in (II) there are no hydrogen bonds of any kind.
Supporting information
CCDC references: 659127; 659128
Equimolar quantities (2.6 mmol of each component) of 2-acetyltetralone and
either 5-amino-3-methyl-1H-pyrazole for (I), or
5-amino-3-tert-butyl-1H-pyrazole for (II), were thoroughly mixed
at room temperature. The solvent-free mixtures were then heated in an oil bath
at 393 K for 1.5–2 min. The resulting melts were stirred briefly, and then
allowed to cool to ambient temperature to solidify. The resulting solids were
extracted with ethanol and, after removal of the solvent under reduced
pressure, the products (I) and (II) were recrystallized from dimethylformamide
to give crystals suitable for single-crystal X-ray diffraction. (I), pale
orange [colourless according to CIF] crystals, yield 86%, m.p. 385–387 K;
MS (70 eV) m/z (%): 249 (100, M+), 234 (8), 221 (9),
127 (16), 39 (22). (II), pale yellow [colourless according to CIF]
crystals, yield 84% m.p. 439–441 K; MS (70 eV) m/z (%): 291
(96, M+), 276 (92), 246 (100), 128 (25), 57 (25), 41 (57), 39 (56).
Crystals of both (I) and (II) are triclinic, and for each the space group
P1 was selected, and confirmed by the structure analysis. All H atoms
were located in difference maps, and then treated as riding atoms in
geometrically idealized position with C—H distances 0.95 Å (aromatic),
0.98 Å (CH3) or 0.99 Å (CH2), and with Uiso(H) =
kUeq(C), where k = 1.5 for the methyl groups and 1.2 for all other H
atoms. A search for possible additional symmetry in (II) revealed none.
For both compounds, data collection: COLLECT (Hooft, 1999); cell refinement: DIRAX/LSQ (Duisenberg et al., 2000); data reduction: EVALCCD (Duisenberg et al., 2003); program(s) used to solve structure: Sir2004 (Burla et al., 2005); program(s) used to refine structure: OSCAIL (McArdle, 2003) and SHELXL97 (Sheldrick, 1997); molecular graphics: PLATON (Spek, 2003); software used to prepare material for publication: SHELXL97 and PRPKAPPA (Ferguson, 1999).
(I) 2,5-Dimethyl-6,7-dihydrobenzo[
h]pyrazolo[1,5-
a]quinazoline
top
Crystal data top
| C16H15N3 | Z = 2 |
| Mr = 249.31 | F(000) = 264 |
| Triclinic, P1 | Dx = 1.328 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 7.9430 (19) Å | Cell parameters from 2867 reflections |
| b = 7.9660 (11) Å | θ = 3.1–27.5° |
| c = 9.943 (3) Å | µ = 0.08 mm−1 |
| α = 87.637 (13)° | T = 120 K |
| β = 82.632 (18)° | Plate, pale orange |
| γ = 89.941 (17)° | 0.49 × 0.44 × 0.07 mm |
| V = 623.4 (3) Å3 | |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 2867 independent reflections |
| Radiation source: Bruker-Nonius FR591 rotating anode | 1600 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.074 |
| ϕ and ω scans | θmax = 27.5°, θmin = 3.1° |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | h = −10→10 |
| Tmin = 0.971, Tmax = 0.994 | k = −10→10 |
| 15496 measured reflections | l = −12→12 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.067 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.220 | H-atom parameters constrained |
| S = 1.04 | w = 1/[σ2(Fo2) + (0.132P)2]
where P = (Fo2 + 2Fc2)/3 |
| 2867 reflections | (Δ/σ)max < 0.001 |
| 174 parameters | Δρmax = 0.42 e Å−3 |
| 0 restraints | Δρmin = −0.29 e Å−3 |
Crystal data top
| C16H15N3 | γ = 89.941 (17)° |
| Mr = 249.31 | V = 623.4 (3) Å3 |
| Triclinic, P1 | Z = 2 |
| a = 7.9430 (19) Å | Mo Kα radiation |
| b = 7.9660 (11) Å | µ = 0.08 mm−1 |
| c = 9.943 (3) Å | T = 120 K |
| α = 87.637 (13)° | 0.49 × 0.44 × 0.07 mm |
| β = 82.632 (18)° | |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 2867 independent reflections |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | 1600 reflections with I > 2σ(I) |
| Tmin = 0.971, Tmax = 0.994 | Rint = 0.074 |
| 15496 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.067 | 0 restraints |
| wR(F2) = 0.220 | H-atom parameters constrained |
| S = 1.04 | Δρmax = 0.42 e Å−3 |
| 2867 reflections | Δρmin = −0.29 e Å−3 |
| 174 parameters | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| N1 | 0.2113 (2) | 0.2787 (2) | 0.7554 (2) | 0.0334 (5) | |
| C2 | 0.1988 (3) | 0.1266 (3) | 0.8210 (2) | 0.0346 (6) | |
| C21 | 0.0519 (3) | 0.0156 (3) | 0.8068 (3) | 0.0441 (7) | |
| C3 | 0.3322 (3) | 0.0946 (3) | 0.8954 (3) | 0.0371 (6) | |
| C3A | 0.4344 (3) | 0.2341 (3) | 0.8756 (2) | 0.0318 (6) | |
| N4 | 0.5821 (2) | 0.2726 (2) | 0.92127 (19) | 0.0334 (5) | |
| C5 | 0.6519 (3) | 0.4197 (3) | 0.8817 (2) | 0.0316 (6) | |
| C51 | 0.8150 (3) | 0.4574 (3) | 0.9349 (2) | 0.0374 (6) | |
| C5A | 0.5806 (3) | 0.5352 (3) | 0.7918 (2) | 0.0295 (6) | |
| C6 | 0.6642 (3) | 0.6992 (3) | 0.7433 (2) | 0.0343 (6) | |
| C7 | 0.6505 (3) | 0.7289 (3) | 0.5925 (2) | 0.0323 (6) | |
| C7A | 0.4709 (3) | 0.7192 (3) | 0.5656 (2) | 0.0301 (6) | |
| C8 | 0.4136 (3) | 0.8146 (3) | 0.4611 (2) | 0.0333 (6) | |
| C9 | 0.2461 (3) | 0.8055 (3) | 0.4363 (3) | 0.0373 (6) | |
| C10 | 0.1328 (3) | 0.7036 (3) | 0.5176 (2) | 0.0369 (6) | |
| C11 | 0.1873 (3) | 0.6047 (3) | 0.6217 (2) | 0.0347 (6) | |
| C11A | 0.3564 (3) | 0.6090 (3) | 0.6450 (2) | 0.0290 (6) | |
| C11B | 0.4299 (3) | 0.4968 (3) | 0.7447 (2) | 0.0285 (6) | |
| N11C | 0.3571 (2) | 0.3450 (2) | 0.78920 (19) | 0.0295 (5) | |
| H21A | 0.0501 | −0.0066 | 0.7108 | 0.066* | |
| H21B | −0.0538 | 0.0711 | 0.8422 | 0.066* | |
| H21C | 0.0626 | −0.0907 | 0.8581 | 0.066* | |
| H3 | 0.3492 | −0.0038 | 0.9491 | 0.044* | |
| H51A | 0.9104 | 0.4399 | 0.8641 | 0.056* | |
| H51B | 0.8266 | 0.3826 | 1.0139 | 0.056* | |
| H51C | 0.8149 | 0.5745 | 0.9617 | 0.056* | |
| H6A | 0.7851 | 0.6967 | 0.7580 | 0.041* | |
| H6B | 0.6081 | 0.7918 | 0.7952 | 0.041* | |
| H7A | 0.6977 | 0.8410 | 0.5619 | 0.039* | |
| H7B | 0.7184 | 0.6436 | 0.5402 | 0.039* | |
| H8 | 0.4903 | 0.8876 | 0.4056 | 0.040* | |
| H9 | 0.2097 | 0.8699 | 0.3629 | 0.045* | |
| H10 | 0.0171 | 0.7008 | 0.5027 | 0.044* | |
| H11 | 0.1088 | 0.5337 | 0.6775 | 0.042* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| N1 | 0.0313 (12) | 0.0351 (12) | 0.0344 (11) | −0.0050 (9) | −0.0074 (9) | 0.0003 (9) |
| C2 | 0.0367 (15) | 0.0342 (14) | 0.0327 (13) | 0.0012 (11) | −0.0048 (11) | 0.0016 (11) |
| C21 | 0.0426 (16) | 0.0424 (16) | 0.0477 (16) | −0.0064 (12) | −0.0096 (13) | 0.0057 (12) |
| C3 | 0.0372 (15) | 0.0367 (14) | 0.0370 (14) | 0.0010 (11) | −0.0056 (12) | 0.0041 (11) |
| C3A | 0.0313 (14) | 0.0379 (14) | 0.0259 (12) | 0.0046 (11) | −0.0040 (10) | 0.0016 (10) |
| N4 | 0.0330 (12) | 0.0399 (12) | 0.0278 (11) | 0.0029 (9) | −0.0057 (9) | 0.0007 (9) |
| C5 | 0.0306 (13) | 0.0380 (14) | 0.0267 (12) | 0.0024 (10) | −0.0054 (10) | −0.0013 (10) |
| C51 | 0.0306 (14) | 0.0501 (16) | 0.0325 (14) | 0.0015 (11) | −0.0075 (11) | −0.0027 (12) |
| C5A | 0.0283 (13) | 0.0340 (13) | 0.0261 (12) | 0.0016 (10) | −0.0028 (10) | −0.0037 (10) |
| C6 | 0.0350 (14) | 0.0332 (13) | 0.0352 (14) | 0.0001 (11) | −0.0057 (11) | −0.0034 (11) |
| C7 | 0.0313 (14) | 0.0307 (13) | 0.0347 (14) | −0.0004 (10) | −0.0043 (11) | 0.0003 (11) |
| C7A | 0.0328 (14) | 0.0280 (13) | 0.0296 (12) | 0.0039 (10) | −0.0043 (10) | −0.0030 (10) |
| C8 | 0.0347 (14) | 0.0308 (13) | 0.0343 (13) | 0.0009 (10) | −0.0054 (11) | −0.0002 (11) |
| C9 | 0.0428 (16) | 0.0342 (14) | 0.0368 (14) | 0.0022 (11) | −0.0122 (12) | −0.0012 (11) |
| C10 | 0.0294 (14) | 0.0378 (14) | 0.0449 (15) | 0.0046 (11) | −0.0090 (12) | −0.0025 (12) |
| C11 | 0.0281 (14) | 0.0351 (14) | 0.0405 (14) | 0.0002 (10) | −0.0039 (11) | 0.0008 (11) |
| C11A | 0.0304 (13) | 0.0296 (13) | 0.0277 (12) | 0.0026 (10) | −0.0049 (10) | −0.0040 (10) |
| C11B | 0.0281 (13) | 0.0303 (13) | 0.0268 (12) | 0.0002 (10) | −0.0023 (10) | −0.0021 (10) |
| N11C | 0.0265 (11) | 0.0341 (11) | 0.0281 (11) | 0.0007 (8) | −0.0052 (8) | 0.0010 (9) |
Geometric parameters (Å, º) top
| N1—C2 | 1.350 (3) | C5A—C6 | 1.499 (3) |
| C2—C3 | 1.385 (4) | C6—C7 | 1.526 (3) |
| C3—C3A | 1.370 (3) | C6—H6A | 0.99 |
| C3A—N4 | 1.351 (3) | C6—H6B | 0.99 |
| N4—C5 | 1.320 (3) | C7—C7A | 1.488 (3) |
| C5—C5A | 1.423 (3) | C7—H7A | 0.99 |
| C5A—C11B | 1.379 (3) | C7—H7B | 0.99 |
| C11B—N11C | 1.372 (3) | C7A—C8 | 1.385 (3) |
| N11C—N1 | 1.360 (3) | C7A—C11A | 1.408 (3) |
| C3A—N11C | 1.402 (3) | C8—C9 | 1.386 (3) |
| C2—C21 | 1.489 (3) | C8—H8 | 0.95 |
| C21—H21A | 0.98 | C9—C10 | 1.372 (3) |
| C21—H21B | 0.98 | C9—H9 | 0.95 |
| C21—H21C | 0.98 | C10—C11 | 1.388 (3) |
| C3—H3 | 0.95 | C10—H10 | 0.95 |
| C5—C51 | 1.497 (3) | C11—C11A | 1.393 (3) |
| C51—H51A | 0.98 | C11—H11 | 0.95 |
| C51—H51B | 0.98 | C11A—C11B | 1.482 (3) |
| C51—H51C | 0.98 | | |
| | | |
| C2—N1—N11C | 104.10 (18) | C7—C6—H6B | 109.8 |
| N1—C2—C3 | 112.6 (2) | H6A—C6—H6B | 108.3 |
| N1—C2—C21 | 119.3 (2) | C7A—C7—C6 | 111.13 (19) |
| C3—C2—C21 | 128.1 (2) | C7A—C7—H7A | 109.4 |
| C2—C21—H21A | 109.5 | C6—C7—H7A | 109.4 |
| C2—C21—H21B | 109.5 | C7A—C7—H7B | 109.4 |
| H21A—C21—H21B | 109.5 | C6—C7—H7B | 109.4 |
| C2—C21—H21C | 109.5 | H7A—C7—H7B | 108.0 |
| H21A—C21—H21C | 109.5 | C8—C7A—C11A | 118.7 (2) |
| H21B—C21—H21C | 109.5 | C8—C7A—C7 | 121.3 (2) |
| C3A—C3—C2 | 105.9 (2) | C11A—C7A—C7 | 120.1 (2) |
| C3A—C3—H3 | 127.1 | C7A—C8—C9 | 121.1 (2) |
| C2—C3—H3 | 127.1 | C7A—C8—H8 | 119.4 |
| N4—C3A—C3 | 132.1 (2) | C9—C8—H8 | 119.4 |
| N4—C3A—N11C | 121.8 (2) | C10—C9—C8 | 120.1 (2) |
| C3—C3A—N11C | 106.1 (2) | C10—C9—H9 | 119.9 |
| C5—N4—C3A | 117.4 (2) | C8—C9—H9 | 119.9 |
| N4—C5—C5A | 123.0 (2) | C9—C10—C11 | 120.0 (2) |
| N4—C5—C51 | 115.7 (2) | C9—C10—H10 | 120.0 |
| C5A—C5—C51 | 121.3 (2) | C11—C10—H10 | 120.0 |
| C5—C51—H51A | 109.5 | C10—C11—C11A | 120.4 (2) |
| C5—C51—H51B | 109.5 | C10—C11—H11 | 119.8 |
| H51A—C51—H51B | 109.5 | C11A—C11—H11 | 119.8 |
| C5—C51—H51C | 109.5 | C11—C11A—C7A | 119.6 (2) |
| H51A—C51—H51C | 109.5 | C11—C11A—C11B | 124.3 (2) |
| H51B—C51—H51C | 109.5 | C7A—C11A—C11B | 116.0 (2) |
| C11B—C5A—C5 | 119.9 (2) | N11C—C11B—C5A | 116.4 (2) |
| C11B—C5A—C6 | 117.5 (2) | N11C—C11B—C11A | 121.9 (2) |
| C5—C5A—C6 | 122.6 (2) | C5A—C11B—C11A | 121.6 (2) |
| C5A—C6—C7 | 109.26 (19) | N1—N11C—C11B | 127.18 (19) |
| C5A—C6—H6A | 109.8 | N1—N11C—C3A | 111.30 (19) |
| C7—C6—H6A | 109.8 | C11B—N11C—C3A | 121.5 (2) |
| C5A—C6—H6B | 109.8 | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| C3—H3···N4i | 0.95 | 2.55 | 3.495 (3) | 175 |
| C7—H7B···Cgii | 0.99 | 2.97 | 3.789 (3) | 141 |
| Symmetry codes: (i) −x+1, −y, −z+2; (ii) −x+1, −y+1, −z+1. |
(II) 2-
tert-Butyl-5-methyl-6,7-dihydrobenzo[
h]pyrazolo[1,5-
a]quinazoline
top
Crystal data top
| C19H21N3 | Z = 4 |
| Mr = 291.39 | F(000) = 624 |
| Triclinic, P1 | Dx = 1.270 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 11.2632 (5) Å | Cell parameters from 6991 reflections |
| b = 11.4574 (7) Å | θ = 3.3–27.5° |
| c = 12.1317 (6) Å | µ = 0.08 mm−1 |
| α = 93.449 (5)° | T = 120 K |
| β = 90.313 (4)° | Plate, pale yellow |
| γ = 102.791 (6)° | 0.52 × 0.35 × 0.15 mm |
| V = 1523.66 (14) Å3 | |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 6991 independent reflections |
| Radiation source: Bruker-Nonius FR591 rotating anode | 4144 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.061 |
| ϕ and ω scans | θmax = 27.5°, θmin = 3.3° |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | h = −14→14 |
| Tmin = 0.972, Tmax = 0.989 | k = −14→14 |
| 37817 measured reflections | l = −15→15 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.065 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.213 | H-atom parameters constrained |
| S = 1.06 | w = 1/[σ2(Fo2) + (0.112P)2 + 0.7885P]
where P = (Fo2 + 2Fc2)/3 |
| 6991 reflections | (Δ/σ)max < 0.001 |
| 405 parameters | Δρmax = 0.42 e Å−3 |
| 0 restraints | Δρmin = −0.39 e Å−3 |
Crystal data top
| C19H21N3 | γ = 102.791 (6)° |
| Mr = 291.39 | V = 1523.66 (14) Å3 |
| Triclinic, P1 | Z = 4 |
| a = 11.2632 (5) Å | Mo Kα radiation |
| b = 11.4574 (7) Å | µ = 0.08 mm−1 |
| c = 12.1317 (6) Å | T = 120 K |
| α = 93.449 (5)° | 0.52 × 0.35 × 0.15 mm |
| β = 90.313 (4)° | |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 6991 independent reflections |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | 4144 reflections with I > 2σ(I) |
| Tmin = 0.972, Tmax = 0.989 | Rint = 0.061 |
| 37817 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.065 | 0 restraints |
| wR(F2) = 0.213 | H-atom parameters constrained |
| S = 1.06 | Δρmax = 0.42 e Å−3 |
| 6991 reflections | Δρmin = −0.39 e Å−3 |
| 405 parameters | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| N11 | −0.08028 (18) | 0.23561 (18) | 0.22469 (16) | 0.0251 (5) | |
| C12 | −0.1426 (2) | 0.2742 (2) | 0.3086 (2) | 0.0256 (5) | |
| C121 | −0.2776 (2) | 0.2234 (2) | 0.3140 (2) | 0.0262 (5) | |
| C122 | −0.2987 (2) | 0.1016 (2) | 0.3654 (3) | 0.0394 (7) | |
| C123 | −0.3360 (2) | 0.3096 (3) | 0.3846 (2) | 0.0361 (6) | |
| C124 | −0.3344 (2) | 0.2068 (3) | 0.1978 (2) | 0.0367 (7) | |
| C13 | −0.0666 (2) | 0.3564 (2) | 0.3832 (2) | 0.0263 (5) | |
| C13A | 0.0496 (2) | 0.3677 (2) | 0.34248 (19) | 0.0244 (5) | |
| N14 | 0.15925 (18) | 0.42942 (18) | 0.38278 (16) | 0.0262 (5) | |
| C15 | 0.2563 (2) | 0.4153 (2) | 0.3269 (2) | 0.0247 (5) | |
| C151 | 0.3777 (2) | 0.4774 (2) | 0.3765 (2) | 0.0309 (6) | |
| C15A | 0.2485 (2) | 0.3441 (2) | 0.22562 (19) | 0.0235 (5) | |
| C16 | 0.3596 (2) | 0.3320 (2) | 0.1612 (2) | 0.0281 (6) | |
| C17 | 0.3425 (2) | 0.2065 (2) | 0.1057 (2) | 0.0270 (5) | |
| C17A | 0.2270 (2) | 0.1742 (2) | 0.0378 (2) | 0.0247 (5) | |
| C18 | 0.2199 (2) | 0.1075 (2) | −0.0621 (2) | 0.0277 (6) | |
| C19 | 0.1128 (2) | 0.0790 (2) | −0.1247 (2) | 0.0290 (6) | |
| C110 | 0.0127 (2) | 0.1194 (2) | −0.0881 (2) | 0.0281 (6) | |
| C111 | 0.0166 (2) | 0.1848 (2) | 0.0122 (2) | 0.0258 (5) | |
| C11A | 0.1235 (2) | 0.2122 (2) | 0.07662 (19) | 0.0226 (5) | |
| C11B | 0.1367 (2) | 0.2840 (2) | 0.18268 (19) | 0.0221 (5) | |
| N11C | 0.03751 (17) | 0.29473 (17) | 0.24558 (16) | 0.0225 (4) | |
| N21 | 0.54131 (18) | 0.76318 (18) | 0.27332 (16) | 0.0251 (5) | |
| C22 | 0.4613 (2) | 0.7232 (2) | 0.1894 (2) | 0.0243 (5) | |
| C221 | 0.3478 (2) | 0.7725 (2) | 0.1843 (2) | 0.0271 (5) | |
| C222 | 0.3788 (3) | 0.8926 (2) | 0.1309 (3) | 0.0388 (7) | |
| C223 | 0.2499 (2) | 0.6853 (2) | 0.1147 (2) | 0.0345 (6) | |
| C224 | 0.2998 (2) | 0.7903 (3) | 0.3005 (2) | 0.0391 (7) | |
| C23 | 0.5003 (2) | 0.6421 (2) | 0.1150 (2) | 0.0245 (5) | |
| C23A | 0.6119 (2) | 0.6321 (2) | 0.15577 (19) | 0.0222 (5) | |
| N24 | 0.69424 (18) | 0.57077 (18) | 0.11621 (16) | 0.0255 (5) | |
| C25 | 0.7983 (2) | 0.5853 (2) | 0.17177 (19) | 0.0240 (5) | |
| C252 | 0.8909 (2) | 0.5232 (2) | 0.1234 (2) | 0.0297 (6) | |
| C25A | 0.8231 (2) | 0.6578 (2) | 0.27291 (19) | 0.0238 (5) | |
| C26 | 0.9404 (2) | 0.6700 (2) | 0.3370 (2) | 0.0284 (6) | |
| C27 | 0.9783 (2) | 0.7949 (2) | 0.3942 (2) | 0.0259 (5) | |
| C27A | 0.8775 (2) | 0.8241 (2) | 0.4631 (2) | 0.0239 (5) | |
| C28 | 0.8997 (2) | 0.8861 (2) | 0.5658 (2) | 0.0258 (5) | |
| C29 | 0.8056 (2) | 0.9120 (2) | 0.6284 (2) | 0.0285 (6) | |
| C210 | 0.6872 (2) | 0.8770 (2) | 0.5880 (2) | 0.0271 (5) | |
| C211 | 0.6624 (2) | 0.8151 (2) | 0.4860 (2) | 0.0250 (5) | |
| C21A | 0.7567 (2) | 0.7878 (2) | 0.42219 (19) | 0.0231 (5) | |
| C21B | 0.7382 (2) | 0.7173 (2) | 0.31500 (19) | 0.0218 (5) | |
| N21C | 0.63371 (17) | 0.70587 (17) | 0.25265 (16) | 0.0225 (4) | |
| H12A | −0.2625 | 0.0464 | 0.3187 | 0.059* | |
| H12B | −0.3864 | 0.0688 | 0.3708 | 0.059* | |
| H12C | −0.2607 | 0.1119 | 0.4391 | 0.059* | |
| H12D | −0.3018 | 0.3184 | 0.4598 | 0.054* | |
| H12E | −0.4242 | 0.2775 | 0.3865 | 0.054* | |
| H12F | −0.3198 | 0.3879 | 0.3526 | 0.054* | |
| H12G | −0.3242 | 0.2849 | 0.1660 | 0.055* | |
| H12H | −0.4214 | 0.1697 | 0.2016 | 0.055* | |
| H12I | −0.2942 | 0.1547 | 0.1512 | 0.055* | |
| H13 | −0.0900 | 0.3958 | 0.4480 | 0.032* | |
| H51A | 0.3658 | 0.5246 | 0.4440 | 0.046* | |
| H51B | 0.4231 | 0.5308 | 0.3235 | 0.046* | |
| H51C | 0.4238 | 0.4176 | 0.3944 | 0.046* | |
| H16A | 0.4317 | 0.3481 | 0.2116 | 0.034* | |
| H16B | 0.3745 | 0.3918 | 0.1044 | 0.034* | |
| H17A | 0.4126 | 0.2027 | 0.0577 | 0.032* | |
| H17B | 0.3401 | 0.1478 | 0.1628 | 0.032* | |
| H18 | 0.2894 | 0.0807 | −0.0881 | 0.033* | |
| H19 | 0.1086 | 0.0318 | −0.1923 | 0.035* | |
| H110 | −0.0599 | 0.1019 | −0.1320 | 0.034* | |
| H111 | −0.0535 | 0.2111 | 0.0371 | 0.031* | |
| H22A | 0.4122 | 0.8814 | 0.0574 | 0.058* | |
| H22B | 0.4392 | 0.9500 | 0.1768 | 0.058* | |
| H22C | 0.3049 | 0.9237 | 0.1234 | 0.058* | |
| H22D | 0.2322 | 0.6071 | 0.1472 | 0.052* | |
| H22E | 0.2789 | 0.6760 | 0.0393 | 0.052* | |
| H22F | 0.1756 | 0.7164 | 0.1128 | 0.052* | |
| H22G | 0.3636 | 0.8433 | 0.3466 | 0.059* | |
| H22H | 0.2760 | 0.7124 | 0.3331 | 0.059* | |
| H22I | 0.2288 | 0.8263 | 0.2962 | 0.059* | |
| H23 | 0.4588 | 0.6023 | 0.0501 | 0.029* | |
| H52A | 0.8573 | 0.4760 | 0.0558 | 0.045* | |
| H52B | 0.9123 | 0.4700 | 0.1767 | 0.045* | |
| H52C | 0.9639 | 0.5830 | 0.1057 | 0.045* | |
| H26A | 1.0051 | 0.6559 | 0.2863 | 0.034* | |
| H26B | 0.9298 | 0.6091 | 0.3928 | 0.034* | |
| H27A | 1.0511 | 0.7992 | 0.4418 | 0.031* | |
| H27B | 1.0004 | 0.8548 | 0.3380 | 0.031* | |
| H28 | 0.9811 | 0.9111 | 0.5934 | 0.031* | |
| H29 | 0.8221 | 0.9534 | 0.6991 | 0.034* | |
| H210 | 0.6224 | 0.8955 | 0.6306 | 0.033* | |
| H211 | 0.5807 | 0.7910 | 0.4592 | 0.030* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| N11 | 0.0255 (11) | 0.0243 (10) | 0.0247 (11) | 0.0042 (8) | 0.0011 (8) | 0.0006 (8) |
| C12 | 0.0298 (13) | 0.0226 (12) | 0.0255 (13) | 0.0075 (10) | 0.0019 (10) | 0.0042 (10) |
| C121 | 0.0243 (12) | 0.0286 (13) | 0.0255 (13) | 0.0054 (10) | 0.0022 (10) | 0.0021 (10) |
| C122 | 0.0330 (15) | 0.0356 (15) | 0.0490 (18) | 0.0043 (12) | 0.0038 (13) | 0.0101 (13) |
| C123 | 0.0309 (14) | 0.0390 (16) | 0.0376 (15) | 0.0070 (12) | 0.0065 (12) | −0.0024 (12) |
| C124 | 0.0283 (14) | 0.0483 (17) | 0.0310 (15) | 0.0038 (12) | −0.0027 (11) | −0.0003 (13) |
| C13 | 0.0330 (14) | 0.0259 (13) | 0.0217 (12) | 0.0105 (10) | 0.0023 (10) | 0.0008 (10) |
| C13A | 0.0312 (13) | 0.0217 (12) | 0.0203 (12) | 0.0058 (10) | 0.0005 (10) | 0.0009 (10) |
| N14 | 0.0292 (11) | 0.0228 (10) | 0.0255 (11) | 0.0041 (9) | −0.0009 (9) | 0.0005 (8) |
| C15 | 0.0298 (13) | 0.0212 (12) | 0.0234 (12) | 0.0053 (10) | −0.0004 (10) | 0.0048 (10) |
| C151 | 0.0291 (13) | 0.0301 (14) | 0.0310 (14) | 0.0027 (11) | −0.0010 (11) | −0.0038 (11) |
| C15A | 0.0280 (13) | 0.0213 (12) | 0.0210 (12) | 0.0048 (10) | 0.0010 (10) | 0.0025 (9) |
| C16 | 0.0255 (13) | 0.0290 (13) | 0.0290 (13) | 0.0045 (10) | 0.0035 (10) | 0.0011 (11) |
| C17 | 0.0264 (13) | 0.0303 (13) | 0.0256 (13) | 0.0084 (10) | 0.0034 (10) | 0.0039 (10) |
| C17A | 0.0278 (13) | 0.0216 (12) | 0.0250 (13) | 0.0055 (10) | 0.0038 (10) | 0.0034 (10) |
| C18 | 0.0324 (14) | 0.0250 (13) | 0.0269 (13) | 0.0085 (10) | 0.0052 (11) | 0.0037 (10) |
| C19 | 0.0382 (14) | 0.0258 (13) | 0.0217 (12) | 0.0047 (11) | 0.0021 (11) | 0.0001 (10) |
| C110 | 0.0318 (14) | 0.0276 (13) | 0.0240 (13) | 0.0041 (11) | −0.0008 (10) | 0.0020 (10) |
| C111 | 0.0283 (13) | 0.0256 (13) | 0.0233 (12) | 0.0053 (10) | 0.0021 (10) | 0.0022 (10) |
| C11A | 0.0278 (13) | 0.0190 (11) | 0.0210 (12) | 0.0043 (9) | 0.0013 (10) | 0.0038 (9) |
| C11B | 0.0246 (12) | 0.0215 (12) | 0.0210 (12) | 0.0059 (9) | 0.0019 (9) | 0.0045 (9) |
| N11C | 0.0229 (10) | 0.0238 (10) | 0.0201 (10) | 0.0034 (8) | −0.0002 (8) | 0.0010 (8) |
| N21 | 0.0253 (11) | 0.0239 (10) | 0.0267 (11) | 0.0070 (8) | 0.0001 (8) | 0.0001 (9) |
| C22 | 0.0245 (12) | 0.0228 (12) | 0.0242 (12) | 0.0016 (9) | −0.0013 (10) | 0.0037 (10) |
| C221 | 0.0272 (13) | 0.0279 (13) | 0.0267 (13) | 0.0074 (10) | −0.0024 (10) | 0.0014 (10) |
| C222 | 0.0369 (15) | 0.0302 (14) | 0.0507 (18) | 0.0083 (12) | −0.0003 (13) | 0.0102 (13) |
| C223 | 0.0305 (14) | 0.0341 (14) | 0.0381 (15) | 0.0073 (11) | −0.0068 (11) | −0.0034 (12) |
| C224 | 0.0345 (15) | 0.0534 (18) | 0.0326 (15) | 0.0175 (13) | 0.0018 (12) | −0.0021 (13) |
| C23 | 0.02457 (12) | 0.0259 (12) | 0.0217 (12) | 0.0021 (10) | −0.0012 (10) | 0.0025 (10) |
| C23A | 0.0246 (12) | 0.0209 (12) | 0.0203 (12) | 0.0036 (9) | 0.0013 (9) | 0.0007 (9) |
| N24 | 0.0271 (11) | 0.0243 (11) | 0.0252 (11) | 0.0063 (8) | 0.0008 (8) | 0.0009 (8) |
| C25 | 0.0264 (12) | 0.0220 (12) | 0.0221 (12) | 0.0022 (10) | 0.0010 (10) | 0.0026 (10) |
| C252 | 0.0299 (14) | 0.0285 (13) | 0.0308 (14) | 0.0080 (11) | −0.0008 (11) | −0.0026 (11) |
| C25A | 0.0252 (12) | 0.0220 (12) | 0.0238 (12) | 0.0040 (10) | 0.0012 (10) | 0.0028 (10) |
| C26 | 0.0284 (13) | 0.0302 (13) | 0.0271 (13) | 0.0073 (10) | 0.0008 (10) | 0.0009 (11) |
| C27 | 0.0258 (12) | 0.0278 (13) | 0.0226 (12) | 0.0030 (10) | −0.0016 (10) | 0.0016 (10) |
| C27A | 0.0275 (13) | 0.0196 (12) | 0.0240 (12) | 0.0032 (10) | 0.0004 (10) | 0.0035 (10) |
| C28 | 0.0268 (13) | 0.0240 (12) | 0.0242 (12) | 0.0002 (10) | −0.0032 (10) | 0.0031 (10) |
| C29 | 0.0368 (14) | 0.0247 (13) | 0.0223 (12) | 0.0035 (11) | −0.0002 (10) | −0.0007 (10) |
| C210 | 0.0305 (13) | 0.0281 (13) | 0.0229 (12) | 0.0067 (10) | 0.0025 (10) | 0.0018 (10) |
| C211 | 0.0256 (12) | 0.0246 (12) | 0.0233 (12) | 0.0020 (10) | 0.0008 (10) | 0.0033 (10) |
| C21A | 0.0296 (13) | 0.0192 (11) | 0.0194 (12) | 0.0024 (9) | −0.0018 (10) | 0.0040 (9) |
| C21B | 0.0216 (12) | 0.0212 (12) | 0.0213 (12) | 0.0014 (9) | 0.0000 (9) | 0.0041 (9) |
| N21C | 0.0223 (10) | 0.0238 (10) | 0.0212 (10) | 0.0052 (8) | −0.0007 (8) | −0.0001 (8) |
Geometric parameters (Å, º) top
| N11—C12 | 1.347 (3) | N21—C22 | 1.345 (3) |
| C12—C13 | 1.404 (3) | C22—C23 | 1.399 (3) |
| C13—C13A | 1.383 (3) | C23—C23A | 1.379 (3) |
| C13A—N14 | 1.353 (3) | C23A—N24 | 1.355 (3) |
| N14—C15 | 1.324 (3) | N24—C25 | 1.323 (3) |
| C15—C15A | 1.424 (3) | C25—C25A | 1.430 (3) |
| C15A—C11B | 1.377 (3) | C25A—C21B | 1.375 (3) |
| C11B—N11C | 1.381 (3) | C21B—N21C | 1.373 (3) |
| N11C—N11 | 1.364 (3) | N21C—N21 | 1.365 (3) |
| C13A—N11C | 1.389 (3) | C23A—N21C | 1.393 (3) |
| C12—C121 | 1.506 (3) | C22—C221 | 1.511 (3) |
| C121—C123 | 1.527 (4) | C221—C223 | 1.527 (4) |
| C121—C124 | 1.530 (3) | C221—C222 | 1.530 (4) |
| C121—C122 | 1.533 (4) | C221—C224 | 1.531 (4) |
| C122—H12A | 0.98 | C222—H22A | 0.98 |
| C122—H12B | 0.98 | C222—H22B | 0.98 |
| C122—H12C | 0.98 | C222—H22C | 0.98 |
| C123—H12D | 0.98 | C223—H22D | 0.98 |
| C123—H12E | 0.98 | C223—H22E | 0.98 |
| C123—H12F | 0.98 | C223—H22F | 0.98 |
| C124—H12G | 0.98 | C224—H22G | 0.98 |
| C124—H12H | 0.98 | C224—H22H | 0.98 |
| C124—H12I | 0.98 | C224—H22I | 0.98 |
| C13—H13 | 0.95 | C23—H23 | 0.95 |
| C15—C151 | 1.500 (3) | C25—C252 | 1.491 (3) |
| C151—H51A | 0.98 | C252—H52A | 0.98 |
| C151—H51B | 0.98 | C252—H52B | 0.98 |
| C151—H51C | 0.98 | C252—H52C | 0.98 |
| C15A—C16 | 1.508 (3) | C25A—C26 | 1.504 (3) |
| C16—C17 | 1.523 (3) | C26—C27 | 1.522 (3) |
| C16—H16A | 0.99 | C26—H26A | 0.99 |
| C16—H16B | 0.99 | C26—H26B | 0.99 |
| C17—C17A | 1.499 (3) | C27—C27A | 1.498 (3) |
| C17—H17A | 0.99 | C27—H27A | 0.99 |
| C17—H17B | 0.99 | C27—H27B | 0.99 |
| C17A—C18 | 1.387 (3) | C27A—C28 | 1.389 (3) |
| C17A—C11A | 1.405 (3) | C27A—C21A | 1.410 (3) |
| C18—C19 | 1.386 (3) | C28—C29 | 1.384 (3) |
| C18—H18 | 0.95 | C28—H28 | 0.95 |
| C19—C110 | 1.377 (4) | C29—C210 | 1.383 (3) |
| C19—H19 | 0.95 | C29—H29 | 0.95 |
| C110—C111 | 1.385 (3) | C210—C211 | 1.384 (3) |
| C110—H110 | 0.95 | C210—H210 | 0.95 |
| C111—C11A | 1.398 (3) | C211—C21A | 1.397 (3) |
| C111—H111 | 0.95 | C211—H211 | 0.95 |
| C11A—C11B | 1.474 (3) | C21A—C21B | 1.478 (3) |
| | | |
| C12—N11—N11C | 104.26 (19) | C22—N21—N21C | 104.16 (18) |
| N11—C12—C13 | 112.3 (2) | N21—C22—C23 | 112.7 (2) |
| N11—C12—C121 | 118.8 (2) | N21—C22—C221 | 118.3 (2) |
| C13—C12—C121 | 128.9 (2) | C23—C22—C221 | 128.9 (2) |
| C12—C121—C123 | 109.4 (2) | C22—C221—C223 | 109.5 (2) |
| C12—C121—C124 | 110.0 (2) | C22—C221—C222 | 108.7 (2) |
| C123—C121—C124 | 109.3 (2) | C223—C221—C222 | 109.3 (2) |
| C12—C121—C122 | 108.9 (2) | C22—C221—C224 | 110.5 (2) |
| C123—C121—C122 | 110.0 (2) | C223—C221—C224 | 109.1 (2) |
| C124—C121—C122 | 109.3 (2) | C222—C221—C224 | 109.8 (2) |
| C121—C122—H12A | 109.5 | C221—C222—H22A | 109.5 |
| C121—C122—H12B | 109.5 | C221—C222—H22B | 109.5 |
| H12A—C122—H12B | 109.5 | H22A—C222—H22B | 109.5 |
| C121—C122—H12C | 109.5 | C221—C222—H22C | 109.5 |
| H12A—C122—H12C | 109.5 | H22A—C222—H22C | 109.5 |
| H12B—C122—H12C | 109.5 | H22B—C222—H22C | 109.5 |
| C121—C123—H12D | 109.5 | C221—C223—H22D | 109.5 |
| C121—C123—H12E | 109.5 | C221—C223—H22E | 109.5 |
| H12D—C123—H12E | 109.5 | H22D—C223—H22E | 109.5 |
| C121—C123—H12F | 109.5 | C221—C223—H22F | 109.5 |
| H12D—C123—H12F | 109.5 | H22D—C223—H22F | 109.5 |
| H12E—C123—H12F | 109.5 | H22E—C223—H22F | 109.5 |
| C121—C124—H12G | 109.5 | C221—C224—H22G | 109.5 |
| C121—C124—H12H | 109.5 | C221—C224—H22H | 109.5 |
| H12G—C124—H12H | 109.5 | H22G—C224—H22H | 109.5 |
| C121—C124—H12I | 109.5 | C221—C224—H22I | 109.5 |
| H12G—C124—H12I | 109.5 | H22G—C224—H22I | 109.5 |
| H12H—C124—H12I | 109.5 | H22H—C224—H22I | 109.5 |
| C13A—C13—C12 | 105.3 (2) | C23A—C23—C22 | 105.2 (2) |
| C13A—C13—H13 | 127.3 | C23A—C23—H23 | 127.4 |
| C12—C13—H13 | 127.3 | C22—C23—H23 | 127.4 |
| N14—C13A—C13 | 131.6 (2) | N24—C23A—C23 | 131.7 (2) |
| N14—C13A—N11C | 122.2 (2) | N24—C23A—N21C | 121.9 (2) |
| C13—C13A—N11C | 106.1 (2) | C23—C23A—N21C | 106.3 (2) |
| C15—N14—C13A | 117.0 (2) | C25—N24—C23A | 117.5 (2) |
| N14—C15—C15A | 122.9 (2) | N24—C25—C25A | 122.4 (2) |
| N14—C15—C151 | 116.4 (2) | N24—C25—C252 | 116.8 (2) |
| C15A—C15—C151 | 120.6 (2) | C25A—C25—C252 | 120.8 (2) |
| C15—C151—H51A | 109.5 | C25—C252—H52A | 109.5 |
| C15—C151—H51B | 109.5 | C25—C252—H52B | 109.5 |
| H51A—C151—H51B | 109.5 | H52A—C252—H52B | 109.5 |
| C15—C151—H51C | 109.5 | C25—C252—H52C | 109.5 |
| H51A—C151—H51C | 109.5 | H52A—C252—H52C | 109.5 |
| H51B—C151—H51C | 109.5 | H52B—C252—H52C | 109.5 |
| C11B—C15A—C15 | 120.2 (2) | C21B—C25A—C25 | 120.2 (2) |
| C11B—C15A—C16 | 117.5 (2) | C21B—C25A—C26 | 118.2 (2) |
| C15—C15A—C16 | 122.3 (2) | C25—C25A—C26 | 121.7 (2) |
| C15A—C16—C17 | 110.6 (2) | C25A—C26—C27 | 110.3 (2) |
| C15A—C16—H16A | 109.5 | C25A—C26—H26A | 109.6 |
| C17—C16—H16A | 109.5 | C27—C26—H26A | 109.6 |
| C15A—C16—H16B | 109.5 | C25A—C26—H26B | 109.6 |
| C17—C16—H16B | 109.5 | C27—C26—H26B | 109.6 |
| H16A—C16—H16B | 108.1 | H26A—C26—H26B | 108.1 |
| C17A—C17—C16 | 110.9 (2) | C27A—C27—C26 | 111.0 (2) |
| C17A—C17—H17A | 109.4 | C27A—C27—H27A | 109.4 |
| C16—C17—H17A | 109.4 | C26—C27—H27A | 109.4 |
| C17A—C17—H17B | 109.4 | C27A—C27—H27B | 109.4 |
| C16—C17—H17B | 109.4 | C26—C27—H27B | 109.4 |
| H17A—C17—H17B | 108.0 | H27A—C27—H27B | 108.0 |
| C18—C17A—C11A | 119.3 (2) | C28—C27A—C21A | 119.2 (2) |
| C18—C17A—C17 | 121.5 (2) | C28—C27A—C27 | 121.9 (2) |
| C11A—C17A—C17 | 119.2 (2) | C21A—C27A—C27 | 118.9 (2) |
| C19—C18—C17A | 121.0 (2) | C29—C28—C27A | 121.2 (2) |
| C19—C18—H18 | 119.5 | C29—C28—H28 | 119.4 |
| C17A—C18—H18 | 119.5 | C27A—C28—H28 | 119.4 |
| C110—C19—C18 | 119.6 (2) | C210—C29—C28 | 119.6 (2) |
| C110—C19—H19 | 120.2 | C210—C29—H29 | 120.2 |
| C18—C19—H19 | 120.2 | C28—C29—H29 | 120.2 |
| C19—C110—C111 | 120.7 (2) | C29—C210—C211 | 120.4 (2) |
| C19—C110—H110 | 119.6 | C29—C210—H210 | 119.8 |
| C111—C110—H110 | 119.6 | C211—C210—H210 | 119.8 |
| C110—C111—C11A | 120.0 (2) | C210—C211—C21A | 120.5 (2) |
| C110—C111—H111 | 120.0 | C210—C211—H211 | 119.7 |
| C11A—C111—H111 | 120.0 | C21A—C211—H211 | 119.7 |
| C111—C11A—C17A | 119.4 (2) | C211—C21A—C27A | 119.1 (2) |
| C111—C11A—C11B | 123.6 (2) | C211—C21A—C21B | 124.2 (2) |
| C17A—C11A—C11B | 116.9 (2) | C27A—C21A—C21B | 116.7 (2) |
| C15A—C11B—N11C | 115.7 (2) | N21C—C21B—C25A | 116.3 (2) |
| C15A—C11B—C11A | 122.3 (2) | N21C—C21B—C21A | 121.5 (2) |
| N11C—C11B—C11A | 121.9 (2) | C25A—C21B—C21A | 122.2 (2) |
| N11—N11C—C11B | 126.17 (19) | N21—N21C—C21B | 126.72 (19) |
| N11—N11C—C13A | 111.99 (18) | N21—N21C—C23A | 111.59 (18) |
| C11B—N11C—C13A | 121.81 (19) | C21B—N21C—C23A | 121.68 (19) |
| | | |
| N11C—N11—C12—C13 | −0.4 (3) | N21C—N21—C22—C23 | −0.3 (3) |
| N11C—N11—C12—C121 | −178.1 (2) | N21C—N21—C22—C221 | −178.0 (2) |
| N11—C12—C121—C123 | −158.5 (2) | N21—C22—C221—C223 | −158.1 (2) |
| C13—C12—C121—C123 | 24.3 (3) | C23—C22—C221—C223 | 24.7 (3) |
| N11—C12—C121—C124 | −38.5 (3) | N21—C22—C221—C222 | 82.6 (3) |
| C13—C12—C121—C124 | 144.3 (3) | C23—C22—C221—C222 | −94.6 (3) |
| N11—C12—C121—C122 | 81.3 (3) | N21—C22—C221—C224 | −37.9 (3) |
| C13—C12—C121—C122 | −95.9 (3) | C23—C22—C221—C224 | 144.9 (3) |
| N11—C12—C13—C13A | −0.5 (3) | N21—C22—C23—C23A | −0.2 (3) |
| C121—C12—C13—C13A | 176.9 (2) | C221—C22—C23—C23A | 177.0 (2) |
| C12—C13—C13A—N14 | −176.2 (2) | C22—C23—C23A—N24 | −176.8 (2) |
| C12—C13—C13A—N11C | 1.2 (3) | C22—C23—C23A—N21C | 0.7 (2) |
| C13—C13A—N14—C15 | 175.4 (2) | C23—C23A—N24—C25 | 175.8 (2) |
| N11C—C13A—N14—C15 | −1.6 (3) | N21C—C23A—N24—C25 | −1.5 (3) |
| C13A—N14—C15—C15A | 3.3 (3) | C23A—N24—C25—C25A | 3.1 (3) |
| C13A—N14—C15—C151 | −175.9 (2) | C23A—N24—C25—C252 | −176.3 (2) |
| N14—C15—C15A—C11B | −1.5 (4) | N24—C25—C25A—C21B | −1.2 (4) |
| C151—C15—C15A—C11B | 177.7 (2) | C252—C25—C25A—C21B | 178.1 (2) |
| N14—C15—C15A—C16 | 178.1 (2) | N24—C25—C25A—C26 | 177.9 (2) |
| C151—C15—C15A—C16 | −2.8 (3) | C252—C25—C25A—C26 | −2.7 (3) |
| C11B—C15A—C16—C17 | −35.5 (3) | C21B—C25A—C26—C27 | −34.2 (3) |
| C15—C15A—C16—C17 | 145.0 (2) | C25—C25A—C26—C27 | 146.6 (2) |
| C15A—C16—C17—C17A | 53.3 (3) | C25A—C26—C27—C27A | 53.6 (3) |
| C16—C17—C17A—C18 | 143.0 (2) | C26—C27—C27A—C28 | 140.8 (2) |
| C16—C17—C17A—C11A | −37.2 (3) | C26—C27—C27A—C21A | −39.3 (3) |
| C11A—C17A—C18—C19 | 0.8 (4) | C21A—C27A—C28—C29 | 0.4 (4) |
| C17—C17A—C18—C19 | −179.4 (2) | C27—C27A—C28—C29 | −179.7 (2) |
| C17A—C18—C19—C110 | 1.2 (4) | C27A—C28—C29—C210 | −0.8 (4) |
| C18—C19—C110—C111 | −2.0 (4) | C28—C29—C210—C211 | 0.8 (4) |
| C19—C110—C111—C11A | 1.0 (4) | C29—C210—C211—C21A | −0.4 (4) |
| C110—C111—C11A—C17A | 1.0 (4) | C210—C211—C21A—C27A | 0.0 (3) |
| C110—C111—C11A—C11B | 178.3 (2) | C210—C211—C21A—C21B | 176.9 (2) |
| C18—C17A—C11A—C111 | −1.8 (3) | C28—C27A—C21A—C211 | 0.0 (3) |
| C17—C17A—C11A—C111 | 178.4 (2) | C27—C27A—C21A—C211 | −179.9 (2) |
| C18—C17A—C11A—C11B | −179.4 (2) | C28—C27A—C21A—C21B | −177.2 (2) |
| C17—C17A—C11A—C11B | 0.8 (3) | C27—C27A—C21A—C21B | 3.0 (3) |
| C15—C15A—C11B—N11C | −2.1 (3) | C25—C25A—C21B—N21C | −2.3 (3) |
| C16—C15A—C11B—N11C | 178.3 (2) | C26—C25A—C21B—N21C | 178.5 (2) |
| C15—C15A—C11B—C11A | 178.1 (2) | C25—C25A—C21B—C21A | 176.6 (2) |
| C16—C15A—C11B—C11A | −1.4 (3) | C26—C25A—C21B—C21A | −2.5 (3) |
| C111—C11A—C11B—C15A | −157.1 (2) | C211—C21A—C21B—N21C | 21.6 (3) |
| C17A—C11A—C11B—C15A | 20.3 (3) | C27A—C21A—C21B—N21C | −161.4 (2) |
| C111—C11A—C11B—N11C | 23.1 (3) | C211—C21A—C21B—C25A | −157.3 (2) |
| C17A—C11A—C11B—N11C | −159.4 (2) | C27A—C21A—C21B—C25A | 19.7 (3) |
| C12—N11—N11C—C11B | 179.3 (2) | C22—N21—N21C—C21B | 179.7 (2) |
| C12—N11—N11C—C13A | 1.2 (2) | C22—N21—N21C—C23A | 0.8 (2) |
| C15A—C11B—N11C—N11 | −174.1 (2) | C25A—C21B—N21C—N21 | −174.9 (2) |
| C11A—C11B—N11C—N11 | 5.7 (3) | C21A—C21B—N21C—N21 | 6.1 (3) |
| C15A—C11B—N11C—C13A | 3.8 (3) | C25A—C21B—N21C—C23A | 3.9 (3) |
| C11A—C11B—N11C—C13A | −176.4 (2) | C21A—C21B—N21C—C23A | −175.0 (2) |
| N14—C13A—N11C—N11 | 176.1 (2) | N24—C23A—N21C—N21 | 176.9 (2) |
| C13—C13A—N11C—N11 | −1.6 (3) | C23—C23A—N21C—N21 | −1.0 (3) |
| N14—C13A—N11C—C11B | −2.1 (3) | N24—C23A—N21C—C21B | −2.1 (3) |
| C13—C13A—N11C—C11B | −179.7 (2) | C23—C23A—N21C—C21B | −180.0 (2) |
Experimental details
| | (I) | (II) |
| Crystal data |
| Chemical formula | C16H15N3 | C19H21N3 |
| Mr | 249.31 | 291.39 |
| Crystal system, space group | Triclinic, P1 | Triclinic, P1 |
| Temperature (K) | 120 | 120 |
| a, b, c (Å) | 7.9430 (19), 7.9660 (11), 9.943 (3) | 11.2632 (5), 11.4574 (7), 12.1317 (6) |
| α, β, γ (°) | 87.637 (13), 82.632 (18), 89.941 (17) | 93.449 (5), 90.313 (4), 102.791 (6) |
| V (Å3) | 623.4 (3) | 1523.66 (14) |
| Z | 2 | 4 |
| Radiation type | Mo Kα | Mo Kα |
| µ (mm−1) | 0.08 | 0.08 |
| Crystal size (mm) | 0.49 × 0.44 × 0.07 | 0.52 × 0.35 × 0.15 |
| |
| Data collection |
| Diffractometer | Bruker–Nonius KappaCCD
diffractometer | Bruker–Nonius KappaCCD
diffractometer |
| Absorption correction | Multi-scan
(SADABS; Sheldrick, 2003) | Multi-scan
(SADABS; Sheldrick, 2003) |
| Tmin, Tmax | 0.971, 0.994 | 0.972, 0.989 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 15496, 2867, 1600 | 37817, 6991, 4144 |
| Rint | 0.074 | 0.061 |
| (sin θ/λ)max (Å−1) | 0.650 | 0.650 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.067, 0.220, 1.04 | 0.065, 0.213, 1.06 |
| No. of reflections | 2867 | 6991 |
| No. of parameters | 174 | 405 |
| H-atom treatment | H-atom parameters constrained | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 0.42, −0.29 | 0.42, −0.39 |
Selected bond lengths (Å) for (I) top| N1—C2 | 1.350 (3) | C5—C5A | 1.423 (3) |
| C2—C3 | 1.385 (4) | C5A—C11B | 1.379 (3) |
| C3—C3A | 1.370 (3) | C11B—N11C | 1.372 (3) |
| C3A—N4 | 1.351 (3) | N11C—N1 | 1.360 (3) |
| N4—C5 | 1.320 (3) | C3A—N11C | 1.402 (3) |
Hydrogen-bond geometry (Å, º) for (I) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| C3—H3···N4i | 0.95 | 2.55 | 3.495 (3) | 175 |
| C7—H7B···Cgii | 0.99 | 2.97 | 3.789 (3) | 141 |
| Symmetry codes: (i) −x+1, −y, −z+2; (ii) −x+1, −y+1, −z+1. |
Selected bond lengths (Å) for (II) top| N11—C12 | 1.347 (3) | N21—C22 | 1.345 (3) |
| C12—C13 | 1.404 (3) | C22—C23 | 1.399 (3) |
| C13—C13A | 1.383 (3) | C23—C23A | 1.379 (3) |
| C13A—N14 | 1.353 (3) | C23A—N24 | 1.355 (3) |
| N14—C15 | 1.324 (3) | N24—C25 | 1.323 (3) |
| C15—C15A | 1.424 (3) | C25—C25A | 1.430 (3) |
| C15A—C11B | 1.377 (3) | C25A—C21B | 1.375 (3) |
| C11B—N11C | 1.381 (3) | C21B—N21C | 1.373 (3) |
| N11C—N11 | 1.364 (3) | N21C—N21 | 1.365 (3) |
| C13A—N11C | 1.389 (3) | C23A—N21C | 1.393 (3) |
 N and C-H
N and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds, while in (II) there are no hydrogen bonds of any kind.
(arene) hydrogen bonds, while in (II) there are no hydrogen bonds of any kind.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2007/08/00/sk3137/sk3137fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2007/08/00/sk3137/sk3137fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/c/issues/2007/08/00/sk3137/sk3137fig3thm.gif)




We report here the structures of two closely related 6,7-dihydrobenzo[h]pyrazolo[1,5-a]quinazolines, (I) and (II) (Figs. 1 and 2), both prepared using solvent-free cyclocondensation reactions between 2-acetyltetralone and the appropriate 3-alkyl-5-amino-1H-pyrazole. We compare the molecular and supramolecular structures of compounds (I) and (II) with those of the related aryl compounds (III) and (IV) (Portilla et al., 2005) and with that of compound (V) (Low et al., 2004).
In (I) and in each of the independent molecules of (II), the non-aromatic carbocyclic rings adopt a screw-boat conformation. The ring-puckering amplitudes (Cremer & Pople 1975) are 0.506 (2) Å in (I), and 0.481 (2) and 0.482 (3) Å, respectively, for the molecules of types 1 and 2 (containing atoms N11 and N21, see Fig. 2) in (II). In (I), the corresponding ring-puckering angles are θ = 69.0 (2)° and ϕ = 82.6 (3)° for the atom sequence C5A—C6—C7—C7A—C11A—C11B; in (II), for the atom sequences Cx1A—Cx1B—Cx5A—Cx6—Cx7—Cx7A (where x = 1 for molecule 1 and 2 for molecule 2), θ = 67.5 (2)° and ϕ = 210.6 (3)° when x = 1, and θ = 67.0 (2)° and ϕ = 213.4 (3)° when x = 2. For an idealized screw-boat conformation, the values are θ = 67.5° and ϕ = (60n + 30)°, where n represents zero or an integer (Evans & Boeyens, 1989). Similar conformation have been found for the corresponding rings in compounds (III)–(V) (Low et al., 2004; Portilla et al., 2005). Because of the puckering of the non-aromatic carbocyclic rings, the molecules exhibit no internal symmetry and hence they are chiral; however, the centrosymmetric space group in each case accommodates equal numbers of the two enantiomeric forms.
Within the heterobicyclic portions of the molecules, the patterns of the bond distances (Tables 1 and 3) are very similar for all three molecules. Hence we discuss in detail only the distances in (I), as similar remarks apply to (II). In (I), N1—C2 and C3A—N4 the bonds, which are formally double and single bonds respectively, are effectively of the same length; on the other hand, the C5—C5A bond, which is formally a single bond, is significantly longer than the C2—C3 and C5A—C11B bonds, which are formally double bonds. These observations are consistent with bond-fixation of the type characteristic of naphthalene rather than complete peripheral delocalization of the ten π electrons. By contrast, the distances in the carbocylic ring (C7A/C8–C11/C11A) are indicative of typical aromatic delocalization.
The geometries and conformations of the two independent molecules in (II) are very similar. Although the ADDSYM routine in PLATON (Spek, 2003) indicated that no additional crystallographic symmetry is present in (II), examination of the atom coordinates showed that the two independent molecules are approximately related by a pseudo-c-glide plane at x = 0.25. Consistent with this approximate relationship, the reflections of type (0kl) are, in general, very much weaker when l is odd than when l is even, although some exhibit significant intensity.
Two hydrogen bonds (Table 2) link the molecules of (I) into chains of rings. Atom C3 in the molecule at (x, y, z) acts as a hydrogen-bond donor to the ring atom N4 in the molecule at (1 - x, -y, 2 - z), so generating by inversion a cyclic centrosymmetric R22(8) (Bernstein et al., 1995) dimer centred at (1/2, 0, 1). At the same time, atom C7 in the molecule at (x, y, z), acts as a hydrogen-bond donor, via the axial atom H7B, to the C7A/C8–C11/C11A ring in the molecule at (1 - x, 1 - y, 1 - z), thus generating by inversion a second cyclic motif centred at (1/2, 1/2, 1/2). The propagation by inversion of these two hydrogen bonds therefore leads to the formation of a chain of centrosymmetric hydrogen-bonded rings running parallel to the [011] direction, with the rings formed by pairs of C—H···N hydrogen bonds centred at (1/2, n, 1 - n) (where n represents zero or an integer) alternating with the rings formed by pairs of C—H···π(arene) hydrogen bonds centred at (1/2, 1/2 + n, 1/2 - n) (where n represents zero or an integer) (Fig. 3). There is a very weak π–π stacking interaction between pairs of pyrimidine rings. These rings in the molecules at (x, y, z) and (1 - x, 1 - y, 2 - z) are strictly parallel with an interplanar spacing of 3.354 (2) Å; the ring-centroid separation is 3.840 (2) Å, corresponding to a ring-centroid offset of 1.870 (2) Å. If this interaction is regarded as significant, its action is to link the hydrogen-bonded chains into a sheet parallel to (100).
The supramolecular aggregation in (II), by contrast, is extremely simple. There are no hydrogen bonds of any kind in the structure, but the molecules of type 1 are linked in pairs by a single weak π–π stacking interaction between the phenyl rings of the molecules at (x, y, z) and (-x, -y, -z). The interplanar spacing is 3.395 (2) Å, with a ring-centroid separation of 3.974 (2) Å, which corresponds to a ring-centroid offset of 1.867 (2) Å. There are no direction-specific interactions of any kind involving the molecules of type 2.
The stacking interaction in (II) closely resembles that in the 4-methylphenyl analogue (IV) (Portilla et al.,2005), where pairs of molecules are again linked into isolated dimers. In the unsubstituted phenyl compound (III), the molecules are linked into chains by a combination of a C—H···π(arene) hydrogen bond and a π–π stacking interaction. The 4-chlorophenyl derivative (V) crystallizes with Z' = 2 (Low et al., 2004), and the molecules are linked by two independent π–π stacking interactions into chains in which the two types of molecules alternate.