issue contents
November 2014 issue
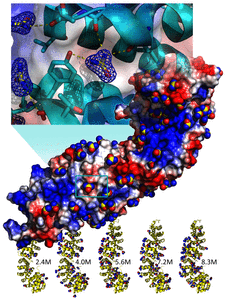
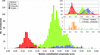
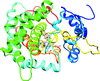
Cover illustration: Hydrogen bonding is the only interaction mode between ![[beta]](/logos/entities/beta_rmgif.gif) -catenin and 105 urea molecules, and the breakdown of hydrogen bonds is the driving force for protein unfolding (p. 2840). Bottom, complex structures of
-catenin and 105 urea molecules, and the breakdown of hydrogen bonds is the driving force for protein unfolding (p. 2840). Bottom, complex structures of ![[beta]](/logos/entities/beta_rmgif.gif) -catenin with urea molecules at five different urea concentrations; middle, distribution of 105 urea molecules on the surface of
-catenin with urea molecules at five different urea concentrations; middle, distribution of 105 urea molecules on the surface of ![[beta]](/logos/entities/beta_rmgif.gif) -catenin; top, detailed interactions of a urea molecule with
-catenin; top, detailed interactions of a urea molecule with ![[beta]](/logos/entities/beta_rmgif.gif) -catenin.
-catenin.
feature articles
Open  access
access
 access
accessThe current and the attainable coverage by X-ray structures of proteins and their functions on the scale of the `protein universe' are estimated. A detailed analysis of the coverage across nearly 2000 proteomes from all superkingdoms of life and functional annotations is performed, with particular focus on the human proteome and the family of GPCR proteins.
research papers
A method to fix protein crystals to mounting loops with hydro-gel using inkjet technology has been developed. The method allows soaking of the mounted crystals in ligand-containing solution and assists in the design of an automated drug-screening pipeline.
Open  access
access
 access
accessH. pylori Csd4 (HP1075), together with other peptidoglycan hydrolases, plays an important role in determining the helical cell shape. Its crystal structure has been determined in three different forms.
Download citation


Download citation


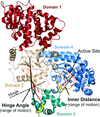
Peptide substrates and small-molecule inhibitors bind anthrax toxin lethal factor in the space between domains 3 and 4 of the hydrolase. Domain 3 is attached on a hinge to domain 2 and can move through an angular arc of more than 35° in response to the binding of different ligands.
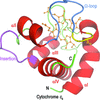
Atomic resolution structures of cytochromes c6 and c6C from Synechococcus sp. PCC 7002 as well as their haem-pocket point mutants are presented. The biophysical and structural properties of these proteins have been characterized with particular focus on the relationship between the structure of the haem-binding pocket and the redox potential.
PDB references: cytochrome c6, 4eic; cytochrome c6 Q57V, 4eid; cytochrome c6C, 4eie; cytochrome c6C L50Q, 4eif
The structures of sperm whale myoglobin variants show that longer distances between the heme axial water and the distal histidine (i.e. weaker hydrogen bonds) correlate with stronger peroxidase activity.
Although urea and guanidine hydrochloride are commonly used to denature proteins, the molecular underpinnings of this process have remained unclear for a century. To address this question, crystal structures of β-catenin were determined at various urea concentrations.
Hydroperoxides are reactive oxygen species (ROS) that are toxic to all cells. This study presents the crystallographic structures of the Escherichia coli alkylhydroperoxides reductase (AhpR) subunits AhpC and AhpF and describes a novel overall mechanistic model of AhpR.
The structure of the discoidin domain from mouse muskelin has been determined and analysis of the interdomain interaction suggests a mechanism for self-association.
PDB reference: discoidin domain from muskelin, 4pqq
Four enzyme–substrate complex structures of CYP154C5 from N. farcinica with the steroids pregnenolone, progesterone, androstenedione and testosterone bound in the active site of the enzyme are reported, revealing structural determinants for the high regioselectivity and stereoselectivity of CYP154C5 in steroid hydroxylation.
The rate of global radiation damage to 70S ribosome crystals has been measured at 100, 180 and 300 K. At 100 and 300 K, damage rates are comparable to and much larger than those of model proteins, respectively. The larger damage rates at 300 K cannot explain the dramatically higher diffraction quality observed at 100 K.
Download citation


Download citation


Open  access
access
 access
accessX-ray and solution structures of the human RyR2 N-terminal region were obtained under near-physiological conditions. The structure exhibits a unique network of interactions between its three domains, revealing an important stabilizing role of the central helix.
PDB reference: N-terminal region of the human ryanodine receptor 2, 4jkq
Open  access
access
 access
accessThe structures of the ascomycetous B. aclada laccase and its L499M T1-site mutant have been solved at 1.7 Å resolution. The mutant enzyme shows a 140 mV lower redox potential of the type 1 copper and altered kinetic behaviour. The wild type and the mutant have very similar structures, which makes it possible to relate the changes in the redox potential to the L499M mutation
The crystal structure of ADC-68 reveals new mechanisms for expanding its substrate spectrum to extended-spectrum cephalosporins and carbapenems.
PDB reference: ADC-68, 4qd4
Open  access
access
 access
accessNuclease A (NucA) is an extracellular nuclease secreted from S. agalactiae and is required for full virulence during infection. Crystal structures and biochemical characterization of NucA mutants reveal possible roles for surface residues in DNA substrate binding and catalysis. These results may serve as a foundation for the design of targeted antibacterial therapeutic compounds.
A GH30 subfamily 8 (GH30-8) xylanase with functional properties different from the canonical GH30-8 glucuronoxylan xylanohydrolases has been biochemically and structurally characterized. The findings highlight the diversity of xylanolytic activities that derive from the GH30 family of glycoside hydrolases.
PDB reference: Clostridium papyrosolvens Xyn30A, 4fmv
Mycobacterium tuberculosis MenB, or 1, 4-dihydroxynaphthoyl coenzyme A synthase, is found to undergo induced-fit conformational changes that play an important role in substrate recognition and catalytic mechanism.
The crystal structures of a first fungal glycoside hydrolase family 5 β-mannosidase from Rhizomucor miehei (RmMan5B) and of its inactive E202A mutant in complex with mannobiose, mannotriose and mannosyl-fructose are presented. Structural analyses reveal the structural basis of substrates binding.
Open  access
access
 access
accessThe present work illustrates that small-angle neutron scattering, deuteration and contrast variation, combined with in vitro particle reconstruction, constitutes a very efficient approach to determine subunit architectures in large, symmetric protein complexes. In the case of the 468 kDa heterododecameric TET peptidase machine, it was demonstrated that the assembly of the 12 subunits is a highly controlled process and represents a way to optimize the catalytic efficiency of the enzyme.
The three-dimensional structure of the GH27 arabinopyranosidase (Abp) from G. stearothermophilus T6 has been determined by molecular replacement, leading to full structural analysis of wild-type Abp (at 2.28 Å resolution) and its catalytic mutant Abp-D197A with (at 2.20 Å resolution) and without (at 2.30 Å resolution) a bound L-arabinose product. The structures demonstrate that Abp is a tetramer built of two pincer-like dimers, as also confirmed by SAXS.
A novel protein fold and the first crystal structure of a gas vesicle protein are described.
PDB reference: GvpF, 4qsg
EhIPPase is probably the inositol polyphosphate 1-phosphatase of E. histolytica.
PDB reference: inositol polyphosphate 1-phosphatase, 4qxd
The structural bases of the inactivation of methionine γ-lyase by a new suicide substrate were determined.
Open  access
access
 access
accessThe enzyme–adenylate structure of a bacterial ATP-dependent DNA ligase (ADL), which does not have any additional DNA-binding domains, is similar to minimal viral ADLs that comprise only the core catalytic domains. The bacterial ADL also lacks the unstructured loops which are involved in DNA binding in the viral ADLs, implying that it must instead use short well structured motifs of the core domains to engage its substrate.
PDB reference: ATP-dependent DNA ligase, 4d05
The first structure of B. anthracis PurK with a bicarbonate ion in the active site is reported. Based on several enzyme structures with different ligands in the active site, a mechanism for the enzymatic reaction is proposed.
Arylamine N-acetyltransferases (NAT) are xenobiotic metabolizing-enzymes that can acetylate aromatic amine antibiotics. The structure of NAT from the pathogen Mycobacterium abscessus is reported and shows differences with other mycobacterial isoforms.
PDB reference: arylamine N-acetyltransferase, 4guz
SERK proteins play a central role in immune and developmental signaling pathways in plants. Structural studies have been performed in order to better understand the role of the OsSERK2 coreceptor in signaling with its partner receptors. Here, crystal structures of the LRR domains of OsSERK2 and a D128N OsSERK2 mutant, expressed as hagfish variable lymphocyte receptor (VLR) fusions, are reported.
addenda and errata
Free 

A correction is made to the article by Wolf et al. [(2014), Acta Cryst. D70, 841–850].


 journal menu
journal menu