issue contents
October 1997 issue
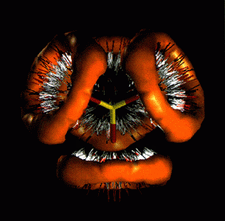
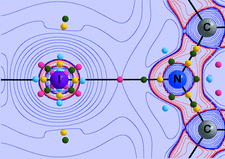
Cover illustration: Contoured distribution of hydrogen bonds from N-H and O-H donors about the oxygen acceptors of the disulfate dianion. Data for the 232 hydrogen bonds (O...H > 2.62 Å) were computed from the Cambridge Structural Database and the required symmetry was applied to generate the final distribution. Courtesy of M. Verdonk and J. Cole
research papers
Download citation


Download citation


Room- and low-temperature experimental electron density studies of YTiO3 provide information on interatomic interactions which may be significant for understanding the origins of ferromagnetic behaviour in that compound.
Depending on the loading procedure of diamond anvil cells, argon under pressure either crystallizes in f.c.c. or h.c.p. configurations. The elastic properties of the h.c.p. crystals are comparable to those with the f.c.c. structure.
Comparison of the observed thermal expansion of bonds with a semi-empirical model reveals the influence of bond strains and structural constraints in thermal expansion.
Download citation


Download citation


From the observation of radial electron distributions in Fe3O4, the charge difference between sites A and B was found to be +0.46e, which is comparable to that between Fe3+ and [Fe2+Fe3+].
Download citation


Download citation


The organometallic derivative of ruthenium TPP is planar with RuII ions forced to be in the plane of the macrocycle by the imposed crystallographic inversion symmetry.
Download citation


Download citation


The salts [MPh4][BPh4], M = P, As and Sb, crystallize in a superlattice of the P-421c, Z' = 1/4, structure known for the molecules MPh4, M = C, Si, Ge, Sn and Pb. The basic P-421c arrangement is retained because the cations and anion are similar in size; the cell is enlarged to accommodate an ionic arrangement that is favorable electrostatically.
MAr4 molecules and ions often retain -4 symmetry in their crystals because the MAr4 units fit together well to form columnar stacks and because the aryl groups fit together well around empty -4 sites.
Download citation


Download citation


This structure may be relevant to the binding of spermine to nucleic acids since phosphate–phosphate distances are similar to those in β-DNA. Neutron diffraction gives details of the H atoms, including those in an ice-like configuration of water molecules.
CCDC reference: 131669
Download citation


Download citation


This diffraction study of the acid rhodo complex chloride salt has shown that the previously assigned tetragonal symmetry is caused by twinning and that the correct space group is P212121. The presence of two independent dimers in the crystal is in agreement with the spectroscopic and magnetic measurements.
Download citation


Download citation


The crystal structures of the optically active meta chloro- and bromo-substituted 3-hydroxy-3-phenylpropionic acids are compared with their racemic counterparts. The observed structural differences are related to differences in fusion enthalpy and entropy.
Download citation


Download citation


Low-temperature X-ray diffraction studies of four new organic non-linear optical materials [tetracyanoquinodimethane (TCNQ) derivatives] are reported and complete a series of five related compounds. The structures demonstrate varying degrees of quinoidal and zwitterionic ground state behaviour.
Download citation


Download citation


Structural properties of the 1,10-dibromodecane–urea and 1,12-dibromododecane–urea inclusion compounds have been determined by single crystal X-ray diffraction for both the high- and low-temperature phases. This represents the first accurate and reliable report of the conventional low-temperature structure of urea inclusion compounds.
The crystal structure of C60 at liquid helium temperature was examined by electron diffraction using an imaging plate and cryo-TEM. The R factor could be reduced a certain amount by assuming a multi-component crystal.
Download citation


Download citation


X-ray structure determination of the racemate and the (S) enantiomer of (1-hydroxy-3-phenyl-2-propenyl) dimethyl- phosphonate provides an example of validating Wallach's rule. The calculated density of the racemate was found to be 6.1% greater than the (S) enantiomer.
Download citation


Download citation


One of the benzyl groups accepts intermolecular hydrogen bonds from a hydroxyl and an ethynyl group, one to each face of the ring. The geometry of these interactions is precisely determined.
CCDC reference: 131679
short communications
Correction to Acta Cryst. (1997). B53 631-638.
books received
Free 



 journal menu
journal menu