issue contents
April 2015 issue

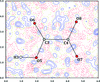
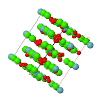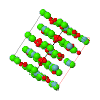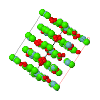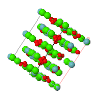
Cover illustration: Plot of the calculated monoclinic structure model of BaTiSi2O7 in the ab plane. See Fig. 8 of Viani et al. [(2015), Acta Cryst. B71, 153-163].
scientific commentaries
Free 

Electron crystallography has made enormous progress over the last decade. It can provide the necessary information that complements powder diffraction data and allows for successful structure analysis of modulated (and unmodulated) structures.
feature articles
Download citation


Download citation


Free 

Using examples of anion-deficient perovskites modulated by periodic crystallographic shear planes, it is demonstrated what kind of local structural information can be obtained using various transmission electron microscopy techniques and how this information can be implemented in the crystal structure refinement against the powder diffraction data.
research papers
Download citation


Download citation


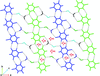
Supramolecular interactions in three salts, namely 2-amino-4,6-dimethylpyrimidin-1-ium thiobarbiturate trihydrate (I), 2-amino-4,6-dimethoxypyrimidin-1-ium thiobarbiturate dihydrate (II) and 2,4-diamino-5-(3′,4′,5′-trimethoxybenzyl)pyrimidin-1-ium thiobarbiturate (III), were analysed. The formation of N—H⋯S hydrogen bonds, a water-mediated base pair and water–water interactions in these crystal systems offer scope for these systems to be considered as a model in the study of hydration of nucleobases and water-mediated nucleobase base pairs in macromolecules.
Download citation


Download citation


The crystal structure of BaTiSi2O7 (BTS2) has been solved from powder diffraction data. It shows the coexistence of monoclinic and triclinic domains within the same crystals. The confirmed presence of TiO5 pyramidal units, with one short Ti—O distance involving the apical oxygen, is in agreement with the theories on the photo-luminescent properties of this compound. Considerations on the stability of BTS2 justifies BaTiSi4O11 (BTS4) as an unavoidable contaminant during synthesis. The reflection list for BTS4 is reported.
Open  access
access
 access
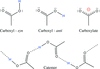
accessThe vast diversity of carboxyl–carboxyl(ate) arrangements is reduced to 17 supramolecular motifs and eight catemers. Examples of each, extracted from the CSD, are presented.
Download citation


Download citation


The synthesis, spectral and structural study of new phosphoramides containing isoxazole, and two lanthanide complexes are reported. Hydrogen bonding and metal–ligand interactions are studied theoretically.
CCDC reference: 989380
Download citation


Download citation


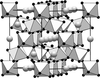
A single crystal of the Nd5Mo3O16 structure type with lead partly substituting for neodymium was studied by precision X-ray diffraction and high-resolution transmission microscopy. Partial substitutions of Mo cations for the Nd position and of Nd for the Mo position in crystals of the Ln5Mo3O16 oxide family are corroborated by X-ray diffraction for the first time.
CCDC reference: 1050401
Download citation


Download citation


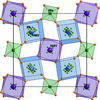
KAgCO3 is polytypic and crystallizes as allotwins (oriented associations of non-equivalent polytypes) with highly ordered domains.
Download citation


Download citation


The symmetry of the magnetic ordering of α-Fe2O3 and α-Cr2O3 is found to be monoclinic and not trigonal, so the symmetry of their crystal structures should also be monoclinic. This conclusion is supported both by experimental X-ray powder diffraction studies as well as by theoretical arguments.
Download citation


Download citation


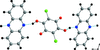
Two chlorine-containing uracil derivatives have been cocrystallized with several triazine and pyrimidine derivatives. Six cocrystals and two (pseudo-) polymorphs were obtained with two-dimensional layers mainly promoted by hydrogen bonds.
Download citation


Download citation


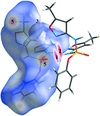
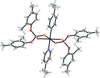
New cobalt(II) complexes of disubstituted diphenyldithiophosphates with donor ligands have been synthesized and structurally characterized by elemental analyses, IR and single-crystal X-ray analyses. Single-crystal X-ray diffraction analysis reveals a six-coordinated distorted octahedral geometry for the CoS4N2 chromophore, defined by two chelating dithio anions and two N-donor ligands.
Download citation


Download citation


Open  access
access
 access
accessCorrelated variations of chemical bonds demonstrate stabilization by the resonance of the chloranilic acid anion. Proton transfer in some of the intermolecular hydrogen bonds is responsible for the ferroelectic properties.
B-IncStrDB reference: 10522E4Avuq
CCDC reference: 1051555
short communications
Statistical models to predict lattice constants of ternary pyrochlores A2B2O6O′ have been derived using multiple linear regression analysis.
addenda and errata
Free 

book reviews
Free 



 journal menu
journal menu