issue contents
January 1997 issue
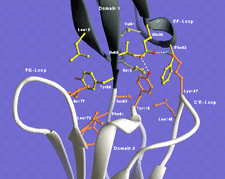
Cover illustration: View of interdomain interface of cell adhesion molecule VCAM-D1D2. The two hydrophobic patches (yellow from domain 1 and orange from domain 2) allow the domains to rock back and forth around the `pivot' residue Tyr89. The long C'E loop and FG loop seem to guide the movement. Courtesy of Jia-huai Wang.
international union of crystallography
Free 

research papers
A report of the experimental and analytical procedures to extract multiwavelength anomalous scattering (MAD) derived phases for the E. coli sulfite reductase hemoprotein from its native iron-containing cofactors. Problems arising from limitations in the experimental system and a small signal-to-noise ratio were overcome by identifying and compensating for aberrant observations and sources of systematic error.
A procedure for refining anomalous-scattering models and calculating protein phases from the Bijvoet and dispersive differences of macromolecular multiwavelength anomalous diffraction (MAD) experiment is described. The protocol, which accounts for more than one type of anomalous scatterer, incorporates stereochemical restraints, approximates maximum likelihood, and partially compensates for correlated errors, produced a MAD-phased electron-density map for the sulfite reductase hemoprotein that was of better quality than the combined MAD/MIR map originally used to determine the structure.
One-line and two-line empirical weighting schemes, as a function of sinθ/λ, are shown to be superior to the unit weights or experimental weights for the refinement of protein structures. Improvements are noted in the figures of merit as well as in the stereochemistry of the resulting model.
A direct method was combined with solvent flattening to resolve the phase ambiguity intrinsic to single isomorphous replacement (SIR) data. A new phase extension procedure was proposed making use of the SIR information enabling phase extension from 6 to 2.5 Å in a test calculation with experimental diffraction data of proteins.
The overlap predicted from B values of paired atoms in two structures to be compared is used to develop a probabilty of difference which is shown to correlate with features in difference maps and to reveal significant differences.
Careful data collection and processing as well as statistically optimal heavy-atom refinement and phasing allowed the determination of a high-quality electron-density map for a 26 kDa protein from a single krypton derivative.
PDB reference: 1lvy
The three-dimensional crystal structure of the single-stranded DNA-containing parvovirus, minute virus of mice, has been determined to 3.5 Å resolution. The structure of minute virus of mice has a large amount of icosahedrally ordered DNA, amounting to 22% of the viral genome, which is significantly more than that found in canine parvovirus.
PDB reference: 1mvm
short communications
A new crystal form of the DNA-binding domain of yeast transcription factor PHO4 complexed with DNA has been obtained.
Rhamnogalaturonase A from Aspergillus aculeatus has been crystallized in space group I212121 or I222; crystals diffract to 2.0 Å resolution. A mass spectrum of the enzyme shows a glycosylation corresponding to 5.9 kDa.
The plasminogen binding protein tetranectin and the C-type lectin CRD of this protein have been crystallized. Diffraction data to 3.0 and 2.0 Å have been obtained of the two proteins, respectively. The structures have been solved by molecular replacement with the CRD of rat mannose-binding protein as a search model.
Salt-mediated killer toxin from Pichia farinosa has been crystallized under both high- and low-salt conditions. Preliminary diffraction studies showed that the crystals from the two conditions have similar but significantly different structures.
The cohesin-2 domain from the cellulosome of Clostridium thermocellum has been crystallized. The crystals diffract to at least 2.3 Å.
The glycosylated functional monoferric C-terminal half (C lobe) (Mr = 40 kDa) of buffalo lactoferrin has been produced by limited proteolysis using proteinase K. The iron-saturated C lobe has been crystallized by microdialysis. The crystals belong to the monoclinic system, space group P21 with unit-cell dimensions a = 44.4, b = 152.3, c = 38.8 Å and β = 105.5°. There is one protein molecule of 40 kDa in the asymmetric unit. A data set at 2.8 Å has been collected on an imaging-plate scanner.
Crystals of psoriasin, a protein related to the skin disease psoriasis, are grown in two different forms. One form contains bound Ca2+ while the other form contains both Ca2+ and Zn2+.
The purification of the DNA recombination protein RuvA and its crystallization are reported. The space-group symmetry implies a fourfold arrangement of the protein and the DNA duplex arms in a Ruv A-Holliday junction complex.
Tobacco ringspot virus, an icosahedral RNA plant virus with putative evolutionary significance, has been purified and crystallized in a new monoclinic crystal form. The particle orientation and packing have been determined with X-ray data collected to 3.3 Å.
Plastocyanin from Silene (SilPc) expressed in E. coli has been crystallized in a form suitable for X-ray diffraction analysis by a macroseeding method using ammonium sulfate as a precipitant in acetate buffer (pH = 5.5).


 journal menu
journal menu


































