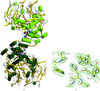issue contents
July 2003 issue
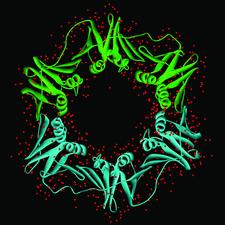
Cover illustration: View of the DNA polymerase III ![[beta]](/logos/entities/beta_rmgif.gif) -clamp dimer. The two monomers are shown as green (monomer A) and cyan (monomer B) ribbons. Water molecules are shown as red spheres (p. 1192).
-clamp dimer. The two monomers are shown as green (monomer A) and cyan (monomer B) ribbons. Water molecules are shown as red spheres (p. 1192).
research papers
The electron-density maps of the protein and the bound waters are found to be better overall in the structures of apocrustacyanin C1 studied from a space-grown crystal compared with those from an earth-grown crystal; both were grown using vapour-diffusion crystal-growth geometry. The improved residues are on the surface of the protein, with two involved in or nearby crystal lattice-forming interactions, thus linking an improved crystal-growth mechanism to the molecular level.
A new approach is presented for analyzing intensity statistics and for diagnosing perfect hemihedral twinning in macromolecular single-crystal X-ray diffraction data. The new statistic compares pairs of reflections that are close in reciprocal space, making it insensitive to phenomena such as anisotropic diffraction and pseudo-centering which can confound traditional intensity statistics.
A readily extensible system interfacing to the main functionality of the CCP4 program suite and providing project-management and file-viewing tools is presented.
An automated crystallographic system (ACrS) was developed for high-throughput structure determination. ACrS uses a web user interface, a relational database and a process distributive interface to drive structure determination with well established crystallographic software.
A new method and software program for the optimal choice of data-collection parameters are presented.
The structure of 1-L-myo-Inositol 1-phosphate synthase in its NAD+-bound, NADH-bound and apo forms is presented.
Crystal structures of the thymus and activation-regulated chemokine (TARC) have been solved by molecular replacement in both the triclinic P1 and tetragonal P41 space groups, and refined to resolutions of 1.72 and 2.1 Å, respectively. Both structural forms and the structure determination are described.
Open  access
access
 access
accessA procedure for iterative model-building, statistical density modification and refinement at moderate resolution (up to about 2.8 Å) is described.
The crystal structure of the oxidized human sulfite oxidase cytochrome b5 domain has been determined at 1.2 Å resolution and has been refined to a crystallographic R factor of 0.107 (Rfree = 0.137).
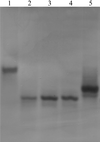
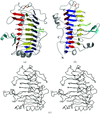
Structural differences between the two subunits in the unit cell are used to comment on the flexibility of parts of the structure of the ring-shaped β sliding clamp.
PDB reference: β subunit of DNA polymerase III, 1mmi, r1mmisf
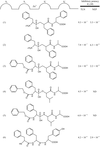
Inhibition of thermolysin by a new α-aminophosphinic compound is discussed.
The high-resolution structure of human DPP-IV as an apo enzyme and in complex with a cyano-pyrrolidine inhibitor is reported. The inhibitor forms a covalent intermediate with the active-site serine.
PDB reference: Apo dipeptidyl peptidase IV/CD26, 1pfq
structural genomics papers
A conserved hypothetical protein, TT1466, from the thermophilic bacterium T. thermophilus HB8 was synthesized by a cell-free system. The crystal structure was highly homologous to that of the CoA-binding domain of succinyl-CoA synthetase and CoA binding of TT1466 was shown by isothermal titration calorimetry.
Hypothetical protein AQ_1354 appeared to have a fold similar to matrix metalloproteinases, but failed to exhibit such an activity under standard assay conditions.
PDB reference: AQ_1354, 1oz9, r1oz9sf
crystallization papers
A soluble mutant of the outer-membrane lipoprotein receptor LolB from E. coli was crystallized in two forms. The monoclinic and hexagonal crystals diffracted X-rays to 1.9 and 2.2 Å resolution, respectively, and MAD phasing of monoclinic crystals of the selenomethionylated protein was successful.
Lychnin and dianthin 30 belong to type 1 RIPs. They have been crystallized by the vapour-diffusion method and the crystals diffracted to 1.7 and 1.3 Å, respectively.
The BRCT domain from Rhp9/Crb2 has been expressed, purified and crystallized, and preliminary data have been collected.
The rVP2H particles, formed by infectious bursal disease virus (IBDV) outer structural and primary immunogenic protein VP2, have been crystallized and the diffraction data (6 Å) showed that the rVP2H crystals belong to the cubic space group P213 with a unit-cell dimension of 323.1 Å. The icosahedral symmetry of the particles is clearly seen in the self-rotation function maps, with dyads and triads in coincidence with the crystallographic axes.
In an attempt to understand how Xis controls the directionality of bacteriophage λ recombination, co-crystals of the DNA-binding domain of Xis in complex with its binding site within the P-arm of the phage have been obtained using the hanging-drop vapor-diffusion method.
Crystals of native lectin-1 from P. aeruginosa have been obtained and diffract to a resolution beyond 1.9 Å on a rotating-anode X-ray source.
A novel strategy is presented that leads to the crystallization of the isolated N-terminal propeptide of human procathepsin S, a proteinase belonging to the cathepsin L-like endopeptidases of the clan CA1 cysteine peptidases.
An efficient overexpression system for the P. falciparum enoyl reductase facilitated its purification and crystallization. A 2.2 Å resolution diffraction data set was collected.
Lectin C from the roots of pokeweed (P. americana) was crystallized in the tetragonal and rhombohedral crystal systems. Both crystals were heavily twinned. Use of an additive yielded rhombohedral single crystals diffracting to 1.8 Å resolution.
Red fluorescent protein eqFP611 from the sea anemone Entacmaea quadricolor has been crystallized in space group P6522, with unit-cell parameters a = 77.26, b = 77.26, and c = 329.49 Å. The crystals diffracted to 2.5 Å.
The catalytic core domain of the psychrophilic cellulase CelG from P. haloplanktis has been crystallized and diffraction data have been collected to 1.8 Å.
The nucleotide-binding domain of Na,K-ATPase was expressed in E. coli, purified and crystallized. Crystals belong to space group F23 and diffract to 3.1 Å.
X-ray diffraction data from a representative of a new class of GST from H. polygyrus were collected to 1.8 Å resolution at CHESS A1.
Quinolinate phosphoribosyltransferase from H. pylori was crystallized in three different complexes with quinolinic acid, phthalic acid and nicotinic acid mononucleotide, respectively. These complex crystals were found to belong to space group P41212 and diffracted to resolutions of 2.3, 2.8 and 3.3 Å, respectively.
β-Alanine synthase catalyzes the last step in the pyrimidine catabolic pathway, the formation of β-alanine or β-aminoisobutyrate from the corresponding N-carbamylated β-amino acids. Crystals of this enzyme diffract to at least 2.7 Å and belong to space group P21 (unit-cell parameters a = 117.2, b = 77.1, c = 225.5 Å, β = 95.0°).
A His31Tyr mutant of β2 microglobulin, the light chain of human major histocompatibily complex MHC class I protein, was expressed, purified and crystallized. The isolated crystals display four independent molecules per asymmetric unit and tight local packing.
The first DH/PH domain of the signal transducer protein Trio has been expressed in E. coli, purified, crystallized and a native data set to 1.7 Å resolution has been collected.
Crystals of formyl-CoA transferase have been grown at room temperature using PEG 4000 as precipitant. The enzyme has been crystallized alone and in presence of CoA: the crystals diffracted to 2.2 and 2.5 Å, respectively.
An α-glucosidase from a Geobacillus strain which was found at the bottom of the Mariana Trench was crystallized in space group C2. The crystal diffracted to 2.5 Å resolution.
This study describes the first crystallization and preliminary X-ray analysis of an unusual thioredoxin (containing an active site of CPDC) from H. pylori. The protein crystallizes in space group P41 and diffracts to 2.4 Å resolution.
The catalytic domain CBH IB from T. emersonii was crystallized in space group P41212, with unit-cell parameters a = b = 74.426, c = 176.920 Å.
Orthorhombic crystals of cytoglobin, a recently discovered haemoprotein widely distributed in human tissues, have been grown and characterized. Two haem Fe sites have been located using SAD data.
Human vascular adhesion protein-1 has been crystallized.
Crystals of the bacterial enterocin A immunity protein diffract to better than 1.7 Å resolution. The crystals belong to space group C2, probably with two molecules in the asymmetric unit.
Gankyrin, an oncoprotein that interacts with the cyclin D-dependent protein kinase CDK4 and the S6b ATPase subunit of the regulator of the proteasome, has been crystallized and native data to 2.1 Å spacing have been collected using synchrotron radiation.
Crystals of the six-helix bundle core structure from Newcastle disease virus F protein were obtained in two different space groups, P21 and C2.
Crystals of the soluble domain of the Sco homolog BsSco have been obtained and diffract to 2.3 Å.
2-Enoyl-CoA hydratase 2 domain of C. tropicalis multifunctional enzyme type 2, involved in the peroxisomal β-oxidation, has been produced, purified and crystallized. Full MAD data and an additional high-resolution data set were collected from a single SeMet crystal to 2.25 and 1.90 Å resolution, respectively.
A Fab fragment of anti-LewisX murine monoclonal antibody 291-2G3-A has been crystallized using the sitting-drop vapour-diffusion method. X-ray data sets have been collected for the native Fab fragment to 2.05 Å resolution and for the Fab fragment soaked with LewisX to 3.2 and 1.8 Å resolution.
The phenylalanine hydroxylase from P. aeruginosa (PaPheOH) was cloned and expressed in E. coli. Crystals grew in space group P61 and diffracted to 2.0 Å. The structure determination is in progress using MAD data collected around the Se K edge.
The 2,3-bisphosphoglycerate-independent phosphoglycerate mutase from L. mexicana has been overexpressed, purified and co-crystallized with substrate and product.
The mechanism by which Hsp40 functions as a molecular chaperone is not understood. To investigate the mechanism, the yeast type I Hsp40 protein Ydj1 C-terminal peptide-binding fragment has been crystallized complexed with its peptide substrate.
The gene for the DNA-binding protein Sso10a from the hyperthermophilic archaeon S. solfataricus was cloned and overexpressed in Escherichia coli. Crystals of the purified protein have been grown that diffract to beyond 2.15 Å resolution.
DFF45 (also known as ICAD) is involved in nuclei DNA fragmentation in apoptosis. Crystals of a key domain in DFF45 were obtained with space group P4n22 and unit-cell parameters a = b = 162.5, c = 113.0 Å.
The crystallization and preliminary crystallographic analysis of the ADP-dependent phosphofructokinase of T. litoralis is reported.
YPM, a superantigen from Y. pseudotuberculosis, has been crystallized. X-ray diffraction data were collected to 1.8 Å using synchrotron radiation.
short communications
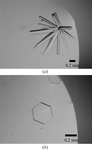
The overall quality and harvest period of adenosine deaminase was improved in two environments: crystallization with agarose gel and crystallization in a high magnetic field.
Binding sites of the monovalent cations in the DNA structure are studied by the detection of the rubidium ions in the Z-DNA crystal. The study indicates most rubidium cation sites located in the Z-DNA structure are cation–water hybrid positions.
Using site-directed mutagenesis techniques, four residues in the T1.5 loop of pectate lyase A were mutated to the homologous sequence in pectate lyase E. Enzymatic characterization and structural analysis of the mutant revealed the significance of the T1.5 loop in the cleavage process of pectic enzymes.
The use of horse hair in protein crystallization has been investigated.


 journal menu
journal menu