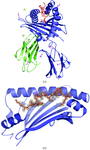issue contents
August 2005 issue
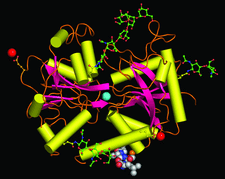
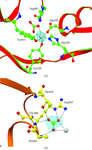
Cover illustration: Structure of zinc-saturated C-terminal lobe of bovine lactoferrin reveals sequestration of zinc ions at acidic pH (p. 1107).
research papers
A novel protein structure was solved by S-SAD using in-house chromium radiation. To reduce the X-ray absorption of the cryo-buffer and cryoloop, a novel free crystal-mounting technique was developed.
A 14 kDa phospholipase, an 18 kDa proteinase inhibitor and a novel 60 kDa glycoside hydrolase were crystallized and their three-dimensional structures were determined using diffraction data collected using a copper rotating-anode X-ray source. Experimental phases were obtained from the anomalous signal provided by S atoms in the case of phospholipase and by I atoms rapidly incorporated into the native structure using the quick cryo-soaking approach in the latter two cases.
This paper describes the 1.45 Å structure of the human MHC-I molecule HLA-A*1101 in complex with a peptide derived from the SARS coronavirus N protein. Implications for vaccine design are discussed.
PDB reference: HLA-A*1101–SNP362-370 complex, 1x7q, r1x7qsf
The shared reservoir solution method, a vapor-diffusion crystallization method where many different experiments share the same reservoir solution, is described.
The three-dimensional structure of uracil-DNA N-glycosylase (UNG) from D. radiodurans has been determined to a resolution of 1.8 Å. An increased number of positively charged residues in the DNA-binding region suggest an explanation of the observed improvement in substrate binding.
PDB reference: drUNG, 2boo, r2boosf
Orthorhombic crystals of the human formylglycine-generating enzyme were grown and phased using SAD based on the anomalous signal of Ca2+ and sulfur using data collected on a rotating-anode generator at the Cu Kα wavelength of 1.54 Å.
PDB reference: FGE, 1z70, r1z70sf
Open  access
access
 access
accessAs an initial characterization of myelin proteins from equine tissue we have purified and crystallized equine P2 from CNS myelin under detergent conditions. The structure was solved to 2.1 Å and shows a molecule of HEPES and the detergent LDAO in the binding cavity.
PDB reference: equine P2 myelin, 1yiv, r1yivsf
The structure of the wild-type TEM-1 at 1.55 Å and the mutant enzyme Ser70Ala at 2.1 Å resolution are presented. The structures suggest the noncovalent mode of catalysis for the mutant enzyme.
The moduli and phases of non-measured reflections are extrapolated to facilitate the ab initio phasing of macromolecules.
A single-crystal neutron diffraction study has been carried out at 1.8 Å resolution on the Z-DNA hexamer d(CGCGCG).
An ab initio method was effectively applied to determine the crystal structures of spherical viruses that are uniquely located at centrosymmetric positions in unit cells.
The C-terminal lobe of bovine lactoferrin was prepared proteolytically using proteinase K. The binding studies with metal ions indicate that a zinc ion remains bound to the C-terminal lobe at pH up to 3.8. The structure of the zinc-saturated C-terminal lobe shows that Zn2+ is bound in the metal-binding cleft with octahedral coordination.
PDB reference: zinc-saturated C-terminal lobe of bovine lactoferrin, 1sdx
The crystal structures at resolutions better than 2.0 Å of native and (S)-lysine-bound dihydrodipicolinate synthase from E. coli are presented and analysed.
The sequence d(TGCGCG)·d(CGCGCA) crystallized in two crystal forms. The structures have been solved in space groups P212121 and P65. The effects of sequence and crystal packing on the structure are reported.
Extraction and visualization of functionally relevant directional motion from the anisotropic atomic displacement parameters in a series of trypsin structures are described.
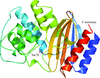
Structural analysis of the single-stranded DNA-binding protein (SSB) from M. smegmatis and related studies confirm the clamp arrangement that stabilizes the dimer and the ellipsoidal shape of the tetramer to be characteristic features of mycobacterial SSBs. Proteins from different sources exhibit considerable variability in quaternary association.
The rate of successful crystallization of prokaryotic proteins can be readily enhanced by utilizing natural intraspecies variants. This is exemplified by the crystallization of six known variants of the lectin domain of the F17G fimbrial adhesin and the analysis of the compatibilities of their sequences with the obtained crystal forms.
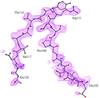
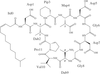
The daptomycin relative tsushimycin is one of the largest structures solved by ab initio direct methods and provides the first X-ray structure for this antibiotic class.
PDB reference: tsushimycin, 1w3m, r1w3msf
The use of a nanovolume asynchronous crystallization robot both to create combinatorial crystallization screens and to set up the crystallization experiments is described.
addenda and errata
Free 



 journal menu
journal menu