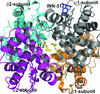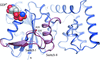issue contents
November 2011 issue
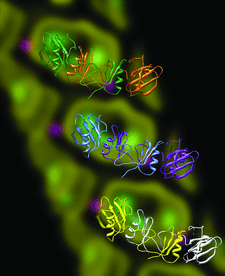
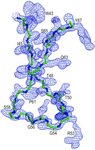
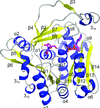
Cover illustration: Asymmetric unit of the galactose-binding Sarcocystis muris lectin SML-2 at 1.95 Å resolution (p. 936). Three dimers are shown in front of the 16 Å resolution ab initio structure discussed by Müller et al. (2006, Acta Cryst. D62, 533-540). Secondary structure plot by CHIMERA [Pettersen, et al. (2004) J. Comput. Chem. 25, 1605-1612].
research papers
Open  access
access
 access
accessThe crystal structure of Mason–Pfizer monkey virus protease folded as a monomer has been solved by molecular replacement using a model generated by players of the online game Foldit. The structure shows at high resolution the details of a retroviral protease folded as a monomer which can guide rational design of protease dimerization inhibitors as retroviral drugs.
PDB reference: monomeric M-PMV retroviral protease, 3sqf
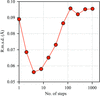
A large-scale study indicates a correlation between thermal stability, as measured using a simplified Thermofluor assay, and crystallization likelihood.
Pyridyl derivatives of vanillin increase the fraction of the more soluble oxygenated sickle hemoglobin and/or directly increase the solubility of deoxygenated sickle hemoglobin. Crystallographic analysis reveals the structural basis of the potent and dual antisickling activity of these derivatives.
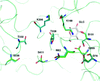
Glucokinase (GK) catalyses the formation of glucose 6-phosphate from glucose and ATP. Here, the first structures of a GK–glucose complex without activator, of GK–glucose–AMP-PNP and of GK–glucose–AMP-PNP with a bound activator are reported.
The PAN-modular structure of the parasite Sarcocystis murismicroneme protein SML-2 and of the complex with 1-thio-β-D-galactose SML-2 was determined by SAD-sulfur phasing. The crystallographic analysis reveals a dimeric lectin structure and the structural basis of the protein's stability in the intestinal tract of intermediate and final hosts, and suggests a role for SML-2 during host-cell invasion.
Download citation


Download citation


Crystal structures of B. thailandensis nucleoside kinase in complex with various ligands are reported.
PDB references: BthNK, 3b1o; BthNK–ADP–MZR, 3b1n; BthNK–ADP–INO, 3b1p; BthNK–INO, 3b1q; BthNK–AMP–Mg2+–AMP, 3b1r
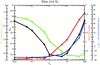
The limitations of weighting between the sum of chemical and data-based targets in macromolecular crystallographic refinement based on comparing the gradients or Hessian diagonal terms of the two potential functions are demonstrated, especially in the context of a maximum-likelihood target that is inherently weighted by the model and data errors
Possible causes of the instability of crystallographic refinement are discussed and a criterion of model convergence is proposed.
Download citation


Download citation


Five structures of NFeoB mutant proteins from S. thermophilus have been determined and reveal how GTP binding induces domain rearrangements in the protein.
A challenging case of crystallization of a human nuclear protein complex consisting of two very similar proteins is described. We highlight the considerations to be made in construct design to obtain diffraction-quality crystals of multi-domain, multi-subunit proteins.
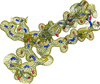
Crystal structures of the human mitochondrial helicase hSuv3 in complex with AMPPNP and with a short strand of RNA are presented.


 journal menu
journal menu