issue contents
January 2013 issue
Cover illustration: Cover illustrations from each of the 20 years of Acta Cryst. D.
editorial
Free 

scientific comment
The importance of presenting macromolecular structures in unified, standard ways is discussed.
research papers
Structural snapshots of the pre- and post-aminoacyl-tRNA synthesis stages have been captured by X-ray crystallography of a novel crystal form of pyrrolysyl-tRNA synthetase.
The S. pyogenes ORF SPy1599, classified into CAZy family GH1, has been shown to display 6-phospho-β-glucosidase activity through a series of kinetics studies on aryl glycosides. The three-dimensional structure has been solved at 2.5 Å resolution from both twinned and untwinned crystal forms.
Open  access
access
 access
accessThe crystal structure of the metalloprotease Gentlyase is described and compared with the structures of other related thermolysin-like proteases.
Crystal structures of 2-aminophenol 1,6-dioxygenase, an enzyme that represents a minor group of extradiol dioxygenases and that catalyses the ring opening of 2-aminophenol, in complex with the lactone intermediate (4Z,6Z)-3-iminooxepin-2(3H)-one and the product 2-aminomuconic 6-semialdehyde and in complex with the suicide inhibitor 4-nitrocatechol are reported.
Biofilm is an extracellular matrix created by many microorganisms, and a major problem in clinical and technical settings. The crystal structure reported here helps elucidate the synthesis of the major component of biofilm produced by Y. pestis and E. coli, and may assist in the development of agents to block it.
PDB reference: N-terminal domain of PgaB, 3vus
Two crystal structures of a plant endo-1,3-β-glucanase representing glycoside hydrolase family GH17 in complexes with products of laminarahexose hydrolysis reveal the protein–saccharide interactions at five key binding subsites for the first time. A trisaccharide is bound at subsites −3, −2 and −1 of the nonreducing end (glycon portion) in both structures, and in one structure an extra tetrasaccharide is bound at subsites +1 to +4 of the reducing end (aglycon portion).
The crystal structures of an M. thermophila glucuronoyl esterase (StGE2) and of its S213A mutant in the unliganded form and in complex with methyl 4-O-methyl-β-D-glucopyranuronate determined at 1.55, 1.90 and 2.35 Å resolution, respectively, are presented for the first time, disclosing the role of StGE2 as a potential biocatalyst.
Polarization-resolved second-harmonic generation microscopy is applied to the identification of multiple crystalline domains within protein-crystal samples.
Open  access
access
 access
accessCrystal structures of the AHL-lactonase AidH in complex with substrate and product are reported at high resolution and a catalytic mechanism is proposed for the metal-independent AHL-lactonase.
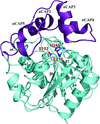
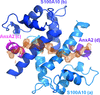
Structure of a 20-amino-acid peptide of AHNAK bound asymmetrically to the AnxA2–S100A10A heterotetramer (1:2:2 symmetry) provides insights into the atomic level interactions that govern this membrane-repair scaffolding complex.
PDB reference: AHNAK peptide bound to AnxA2–S100A10 heterotetramer, 4ftg
Biogenic amine-binding proteins mediate the anti-inflammatory and antihemostatic activities of blood-feeding insect saliva. The structure of the amine-binding protein from R. prolixus reveals the interaction of biogenic amine ligands with the protein.
The crystal structure of SOTI-III in complex with bovine pancreatic trypsin has been solved to 1.7 Å resolution.
The anticancer agents cisplatin and carboplatin bind to a free histidine in a protein. This crystal structure study at data-collection temperatures of 100 and 300 K examines their relative binding affinities to a histidine side chain and the effect of a high radiation dose on the stability of the subsequent protein–Pt adducts. Cisplatin binding is visible and carboplatin binding is not, indicating stronger binding of cisplatin.
The structural basis for differential nucleoside-analogue incorporation by S. mansoni and human adenosine kinase is revealed.
PDB references: SmAK–adenosine, 3vas; 3vaq; SmAK–Ado–AMP, 3uq6; SmAK–tubercidin, 3uq9; SmAK–2-fluoroadenosine, 4dc3
short communications
NO photodissociation in a tetrameric haemoglobin has been detected via Raman microscopy on protein single crystals during X-ray diffraction data collection at different radiation doses.


 journal menu
journal menu