issue contents
January 2015 issue
Crystallography and complementary methods
Proceedings of the CCP4 study weekend

Cover illustration: The crystal packing of plasmodial PLP synthase involved in malarial vitamin B6 biosynthesis (PDB entry 4ads; Tews et al., 2012, Acta Cryst. A68, s32). The study of the enzyme complex consisting of up to 24 protein subunits has used a variety of complementary techniques, such as electron microscopy, analytical ultracentrifugation, static light scattering, isothermal titration calorimetry and spectroscopy, and is thus an example of the use of complementary methods in crystallography.
introduction
Open  access
access
 access
accessbiophysics
Open  access
access
 access
accessGlobal multi-method analysis for protein interactions (GMMA) can increase the precision and complexity of binding studies for the determination of the stoichiometry, affinity and cooperativity of multi-site interactions. The principles and recent developments of biophysical solution methods implemented for GMMA in the software SEDPHAT are reviewed, their complementarity in GMMA is described and a new GMMA simulation tool set in SEDPHAT is presented.
Open  access
access
 access
accessThe current version of the Cryobench in crystallo optical spectroscopy facility of the ESRF is presented. The diverse experiments that can be performed at the Cryobench are also reviewed.

Loading metrics information...
Download citation


Download citation



Loading metrics information...
Open  access
access
 access
accessThe particular challenge of the analysis of optical absorption and Raman spectroscopic data measured from protein crystals and how the SLS-APE software toolbox supports scientists in dealing with such data is described.

Loading metrics information...
Download citation


Download citation



Loading metrics information...
Open  access
access
 access
accessThe biophysical characterization of protein–ligand interactions in solution using techniques such as thermal shift assay, or on surfaces using, for example, dual polarization interferometry, plays an increasingly important role in complementing crystal structure determinations.
SAXS

Loading metrics information...
Download citation


Download citation



Loading metrics information...
Open  access
access
 access
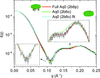
accessA set of quantitative techniques is suggested for assessing SAXS data quality. These are applied in the form of a script, SAXStats, to a test set of 27 proteins, showing that these techniques are more sensitive than manual assessment of data quality.
Open  access
access
 access
accessThe potentialities and limitations of biomacromolecular modelling of small-angle scattering data are reviewed and discussed with a focus on the impact of complementary NMR restraints and a hydration shell.
Open  access
access
 access
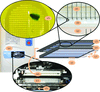
accessA robotic sample changer for solution X-ray scattering experiments optimized for speed and to use the minimum amount of material has been developed. This system is now in routine use at three high-brilliance European synchrotron sites, each capable of several hundred measurements per day.

Loading metrics information...
Download citation


Download citation



Loading metrics information...
Open  access
access
 access
accessThe ISPyB information-management system for crystallography has been adapted to include data from small-angle X-ray scattering of macromolecules in solution experiments.
Open  access
access
 access
accessSystematic SAXS simulations have been analysed over a wide range of parameters in order to better understand the detergent corona around a membrane protein.
new mounts

Loading metrics information...
Download citation


Download citation



Loading metrics information...
Open  access
access
 access
accessAn acoustic high-throughput screening method is described for harvesting protein crystals and combining the protein crystals with chemicals such as a fragment library.
Open  access
access
 access
accessVery little information is available in the literature concerning the experimental heavy-atom phasing of membrane-protein structures where the crystals have been grown using the lipid cubic phase (in meso) method. In this paper, pre-labelling, co-crystallization, soaking, site-specific mercury binding to genetically engineered single-cysteine mutants and selenomethionine labelling as applied to an integral membrane kinase crystallized in meso are described. An assay to assess cysteine accessibility for mercury labelling of membrane proteins is introduced.
EM
Open  access
access
 access
accessThe Collaborative Computational Project for Electron cryo-Microscopy (CCP-EM) is a new initiative for the structural biology community, following the success of CCP4 for macromolecular crystallography. Progress in supporting the users and developers of cryoEM software is reported.
Open  access
access
 access
accessThis review provides a brief update on the use of cryo-electron microscopy for integrated structural biology, along with an overview of the plans for the UK national facility for electron microscopy being built at the Diamond synchrotron.
Open  access
access
 access
accessTools for macromolecular model building and refinement into electron cryo-microscopy reconstructions
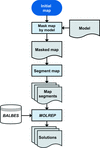
A description is given of new tools to facilitate model building and refinement into electron cryo-microscopy reconstructions.
dynamics

Loading metrics information...
Download citation


Download citation



Loading metrics information...
Open  access
access
 access
accessThis report describes the working of the program CcpNmr Analysis for both NMR chemical shift assignment and structure determination of biological macromolecules.

Loading metrics information...
Download citation


Download citation



Loading metrics information...
Open  access
access
 access
accessThe current computational techniques available for biomolecular simulation are described, and the successes and limitations of each with reference to the experimental biophysical methods that they complement are presented.


 journal menu
journal menu