issue contents
September 2017 issue
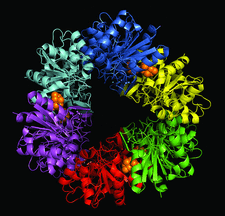
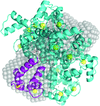
Cover illustration: Structure of the Pfp1 protease from the hyperthermophilic archaeon Thermococcus thioreducens (Larson & McPherson, p. 749).
research papers
Open  access
access
 access
accessUpdated guidelines are presented for publishing biomolecular small-angle scattering (SAS) experiments so that readers can independently assess the quality of the data and models presented. The focus is on solution scattering experiments with either X-rays (SAXS) or neutrons (SANS), where the primary goal is the generation and testing of three-dimensional models, particularly in the context of integrative/hybrid structural modelling.
Open  access
access
 access
accessAUSPEX is a new software tool for the statistical analysis of single-crystal X-ray diffraction data. It can be used to identify problems in the data resulting from the experiment itself, image processing, data scaling or conversion.
Open  access
access
 access
accessProtein structure determination by electron diffraction using a single three-dimensional nanocrystal
A single three-dimensional protein nanocrystal was used for structure determination by electron diffraction. Data were acquired using the rotation method with a Timepix hybrid pixel detector for low-dose data acquisition.
The structure of the Pfp1 intracellular protease from T. thioreducens was solved in two crystal forms: one showing it to be a hexamer and the other a dodecamer. The subunits are disulfide-linked in pairs and the very closely juxtaposed catalytic triads are formed across an interface with amino acids contributed by two separate subunits.
Open  access
access
 access
accessIn order to improve the efficiency of protein crystallization, an alternative approach using the mutation of surface residues was devised based on the results of a statistical analysis of the crystal-packing propensity of amino acids. A systematic crystallization experiment validated the results of the statistical analysis.
High-resolution structures of the Zika virus strain MR766 methyltransferase and helicase proteins are reported.
A pentad mutation of GAPR-1 causes structural changes and shifts the GAPR-1 monomer–dimer equilibrium towards dimerization, which together may prevent its binding to Beclin 1.
PDB reference: pentad mutant of GAPR-1, 5vhg


 journal menu
journal menu