issue contents
May 2018 issue
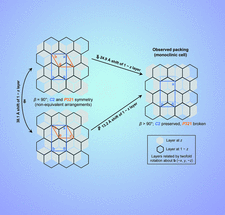
Cover illustration: Staggered packings of trigonal protein layers in crystals of the dynamic protein EutL (Thompson et al., p. 411). Left: two unique, but related, packing arrangements obeying P321 symmetry in a crystal composed of oppositely facing layers of trimers (orange cells). Right: partial offset of the threefold symmetry axes in successive layers of the crystal (perpendicular to the hexagonal axis; the a axis here) leads to breakage of trigonal symmetry while preserving monoclinic symmetry (C2, blue cell).
research papers
High-resolution crystal structures of closed and open conformations of Mycobacterium smegmatis MabA are reported.
Open  access
access
 access
accessAnalysing two sequence-related bacterial glycoside hydrolase family 92 mannosidases with distinct functions, a structural basis for their varied specificities is revealed.
Open  access
access
 access
accessA method is presented to simultaneously resolve the crystal symmetry and indexing ambiguity from sparse data sets.
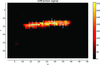
Microfocus X-ray data collection from 18 non-overlapping regions of a single protein crystal revealed unit-cell non-isomorphism and subtle protein dynamics across the crystal specimen.
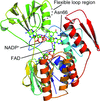
The high-resolution crystal structure of a Zonocerus variegatus flavin-dependent monooxygenase is reported at 1.6 Å resolution together with kinetic studies of structure-based protein variants in order to investigate significant differences in enzyme activity between isoforms.
Open  access
access
 access
accessSignificant improvements to the sample-location, characterization and data-collection algorithms on the autonomous ESRF beamline MASSIF-1 are described. The workflows now include dynamic beam-diameter adjustment and multi-position and multi-crystal data collections.
Open  access
access
 access
accessAn automated data-processing pipeline for protein microcrystals is presented. The processing of multiple small-wedge data sets was made dramatically easier by this pipeline.
Open  access
access
 access
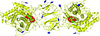
accessA large domain swap encompassing half of the molecule is observed in the crystal structure of the C-terminal domain of human doublecortin. Combined with ultracentrifugation data, the domain swap suggests a mechanism by which doublecortin may cooperatively bind and bundle microtubule fibres.
PDB reference: C-terminal domain of doublecortin, 6fnz
Three industrially relevant glucoamylase structures have been determined, revealing how the starch-binding module can adopt different orientations relative to the catalytic domain.
Open  access
access
 access
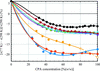
accessThe densities of aqueous solutions of eight common cryoprotectants were measured at T = 77 K and were used to determine electron densities at T = 77 K and thermal contractions on cooling from room temperature. The results provide a quantitative basis for choosing cryoprotectants to optimize outcomes in cryocrystallography, cryo-SAXS, cryogenic temperature X-ray imaging and vitrification-based biological cryopreservation.
Structures of orthorhombic crystals of lysozyme at pH 4.5 show that the binding of phosphate ions produces long-range conformational changes and that low humidity produces a displacement of the fifth α-helix towards the active-site cavity. The interaction of some anions must be considered when analysing experiments with lysozyme at acidic pH values.
PDB references: lysozyme crystallized in the presence of 100 mM ammonium sulfate at pH 4.5, 6fa0; lysozyme crystallized in the presence of 100 mM lithium sulfate at pH 4.5, 6f9x; lysozyme crystallized in the presence of 10 mM lithium sulfate at pH 4.5, 6f9y; lysozyme crystallized in the presence of 5 mM ammonium sulfate at pH 4.5, 6f9z; lysozyme crystallized in the presence of 100 mM sodium phosphate at pH 4.5, 6f1l; lysozyme crystallized in the presence of 100 mM sodium phosphate at pH 4.5, low-humidity form, 6f1m; orthorhombic lysozyme crystallized at 298 K and pH 4.5, 6f1o; tetragonal lysozyme crystallized at 298 K and pH 4.5 with phosphate bound, 6f1p; tetragonal lysozyme crystallized at 298 K and pH 4.5 with phosphate bound, 6f1r


 journal menu
journal menu