issue contents
August 2018 issue
5th International Symposium on Diffraction Structural Biology (ISDSB2016)

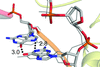
Cover illustration: The city of Knoxville, Tennessee, USA where the 5th International Symposium on Diffraction Structural Biology was held on 7-10 August 2016. In the background, the neutron structures of the active sites of a 5'-methylthioadenosine nucleosidase and of a lytic polysaccharide monooxygenase (LPMO) are superposed onto a Laue neutron diffraction pattern from an LPMO crystal (Schröder et al., p. 778). Photo credit: iStock.com/Sean Pavone. Design: ORNL/Jill Hemman.
introduction
An introductory overview to the articles on diffraction structural biology in this special issue of the journal.
research papers
Small-angle neutron scattering (SANS) provides structural information on biological macromolecules in solution that is perfectly complementary to the information obtained using other structural biology techniques, including crystallography, nuclear magnetic resonance and electron microscopy. Compared with its sister technique small-angle X-ray scattering, SANS allows specific and individual information to be obtained on subunits within important heterogeneous biological assemblies such as multiprotein or protein–RNA/DNA complexes as well as solubilized membrane proteins.
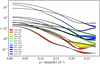
First-principles signal-to-noise calculations for BioSAXS have been developed and applied to evaluate the impact of energy, source brightness, window scattering and other variables on the ability to detect and distinguish protein conformational changes.
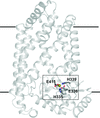
The interaction between the integral membrane protein zinc metalloprotease ZMPSTE24 and the natural product broad-specificity zinc metalloprotease peptidic inhibitor phosphoramidon has been characterized functionally and structurally. The functional results demonstrate the sensitivity of ZMPSTE24 to phosphoramidon in a manner consistent with competitive inhibition, as in soluble zinc metalloproteases, and that the overall mode of binding of phosphoramidon to ZMPSTE24 and soluble zinc metalloproteases, especially gluzincins, is conserved.
PDB reference: ZMPSTE24 in complex with phosphoramidon, 6bh8
The structure of tryptophan indole-lyase with a bound inhibitor, oxindolyl-L-alanine, was determined. The structure provides new insights into the reaction mechanism of the enzyme.
Mismatch conformations are dynamic and vary depending on the environment, including restraints imposed by DNA-binding proteins such as apurinic/apyrimidinic endonuclease 1 (APE1), a key DNA-repair enzyme. Here, both key insights revealed by X-ray crystallography of APE1 bound to mismatch-containing substrates and the specific challenges associated with elucidating base-pairing properties based on implied protonation states and X-ray crystallographic data alone are highlighted.
PDB references: human APE1, substrate complex with an A·A mismatch adjacent to THF, 6boq; substrate complex with a G·G mismatch adjacent to THF, 6bor; substrate complex with an A·C mismatch adjacent to THF, 6bos; substrate complex with a C·C mismatch adjacent to THF, 6bot; substrate complex with a T·C mismatch adjacent to THF, 6bou; substrate complex with an A·G mismatch adjacent to THF, 6bov; substrate complex with a T·T mismatch adjacent to THF, 6bow
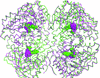
A structure of a copper-containing nitrite reductase from Geobacillus thermodenitrificans (GtNIR) was determined using an anaerobically manipulated crystal. Furthermore, a peroxide-bound structure of GtNIR was determined.
The capabilities of the IMAGINE neutron protein diffractometer at the Oak Ridge National Laboratory High Flux Isotope Reactor and highlights of the first five years of the scientific program are reviewed.
Open  access
access
 access
accessTo improve the sensitivity of hydrogen detection using neutrons, high-pressure cooling of a relatively large protein single crystal and proton polarization of a protein polycrystalline state have been demonstrated as preliminary experiments.
Open  access
access
 access
accessThe application of the cryogenic data-collection environments used in protein X-ray crystallography to neutron protein crystallography is discussed.
Open  access
access
 access
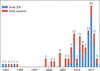
accessModels of crystal structures determined by neutron diffraction and deposited in the Protein Data Bank to date were analysed. The lessons learned from this data-mining effort are summarized and suggestions for improvements to the deposition and validation of neutron models are outlined.


 journal menu
journal menu