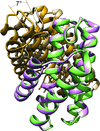issue contents
July 2019 issue

Cover illustration: The closed form of Mycobacterium tuberculosis dihydrofolate reductase in complex with dihydrofolate and antifolates [Ribeiro et al. (2019), Acta Cryst. D75, 682-693]. Structures of MtDHFR in complex with different ligands: pyrimethamine (PYR), cycloguanil (CYC), pemetrexed (PMX), diaverdine (DIA) and dihydrofolate (DHF).
research papers
A crystal structure of an asymmetric dimer of the N-terminal domain of the Euprosthenops australis dragline silk protein is reported. The structure is proposed to represent an intermediate state in spider silk formation.
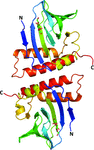
Ncb5or is a multi-domain redox enzyme involved in disease pathways. Structures of the naturally fused CS and cytochrome b5 reductase domains are reported, which provide a window into the function of Ncb5or and reveal distinct differences relative to previously reported CS and cytochrome b5 reductase structures.
Open  access
access
 access
accessThe crystal structure of the transcriptional regulator BgaR with no sugar added is presented, along with those of its complexes with lactose and lactulose. The structure is related to that of the transcriptional regulator AraC, but shows differences in the effector-binding pocket.
To test the theory of `gatekeeper' residues, the nucleotide-binding domains of Blastocystis hominis succinyl-CoA synthetase and mutant proteins were produced, purified and crystallized. The structures of the complexes with Mg2+-ADP show why the enzyme is ATP-specific.
Open  access
access
 access
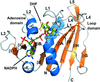
accessInsight is provided into the dynamic interactions of LAT1 and CD98hc and their role in the transport cycle of the complex.
Open  access
access
 access
accessAn efficient optimization screen was devised using random microseed matrix screening to quickly derivatize crystals using an iodinated phasing ligand.
Structures of dihydrofolate reductase from Mycobacterium tuberculosis in complex with dihydrofolate and antifolates show key conformational changes and different binding modes in the protein structure and ligands, respectively, that reflect the plasticity of this enzyme. Several of the movements associated with the dihydrofolate-binding site may be involved in the catalytic cycle of the enzyme, while the different binding modes of the ligands are associated with their distinct functional groups that have to adopt alternative poses to fit into the active site of the protein.
obituaries
Free 

Obituary for Barbara Wharton Low, an important figure in the history of structure determination and our understanding of chemistry and physics today.


 journal menu
journal menu