issue contents
March 2022 issue
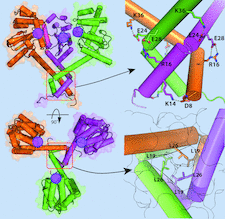
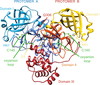
Cover illustration: Trimerization of copper sensitivity protein C from Caulobacter crescentus [Petit et al. (2022), Acta Cryst. D78, 337–352]. A side view (top left image) and top view (bottom left image) of the crystallographic trimer with the three protomer chains represented as surfaces and cartoons coloured orange, magenta and green. On the right are close ups showing the trimerization domain viewed from the side (top image) and the inside of the trimerization domain (bottom image). The surface of each protomer is outlined in black, demonstrating the tight packing of the residues inside the helices and the positions of the active sites on the protomers are indicated with filled magenta circles.
research papers
Open  access
access
 access
accessThe dynamics of proteins can be explored from polymorphs observed by the clustering of multiple data wedges.
Open  access
access
 access
accessAn enzymatic and structural study identifying the factors influencing the transglycosylase activity of RBcel1, a member of glycoside hydrolase family 5, is reported.
Open  access
access
 access
accessUsing the MsCI4 soaking system, the binding of the next-generation thalidomide-derived immunomodulatory drugs avadomide (CC-122) and iberdomide (CC-220) to cereblon was characterized at high resolution, highlighting the utility of the MsCI4 system for studies of the structure–activity relationship of cereblon effectors.
The first structural characterization of long tandem repeats of the d(CGA) triplet motif in the parallel-stranded duplex form is provided and the results show that each strand within the duplex is conformationally distinct and plays a unique role in accommodating duplex asymmetry. The structural uniformity of tandem d(CGA) base-paired triplets obtained from different solution environments further demonstrates that the d(CGA)-based parallel-stranded duplex is a robust and highly predictable structure with applications in DNA nanotechnology.
Open  access
access
 access
accessInlB, an invasion protein from the facultative intracellular pathogen Listeria monocytogenes, is a multi-domain protein that binds and activates the human receptor tyrosine kinase MET. Three new InlB crystal structures reveal the relative orientation of the first two domains (the internalin domain and the B repeat) for the first time. A recurrent packing contact formed by all five crystallographically independent molecules highlights functionally important binding sites in the internalin domain and the B repeat.
PDB references: InlB392, wild type, 7pv9; InlB392_T332E, crystal form I, 7pv8; crystal form II, 7nms
Open  access
access
 access
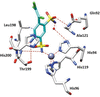
accessThe inhibitory activity of clorsulon and X-ray studies of its complexes with human carbonic anhydrase I and Schistosoma mansoni carbonic anhydrase revealed different modes of binding of this antiparasitic drug, explaining its inhibitory potency against the two enzymes.
Open  access
access
 access
accessThe FFCS platform has recently been developed by the Swiss Light Source macromolecular group. It is an integrated next-generation X-ray crystallography fragment-based drug-discovery pipeline for crystal soaking, handling and data collection.
PDB references: NqrF from Pesudomonas aeruginosa, 7qu3; 7qu5; NqrF from Klebsiella pneumoniae, 7qty; 7qu0
Open  access
access
 access
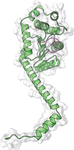
accessThe characterization of the suppressor of copper sensitivity protein C from C. crescentus is reported.
PDB reference: suppressor of copper sensitivity protein C, 7rgv
SASBDB reference: suppressor of copper sensitivity protein C, SASDLE9
Open  access
access
 access
accessThe binding of a series of epibatidine derivatives to acetylcholine-binding protein was investigated using biolayer interferometry. The structures of three complexes inform discussion on the biological implications for interactions with nicotinic acetylcholine receptor subtypes, which are important targets for control of pain.
Open  access
access
 access
accessA new inactive conformation of SARS-CoV-2 main protease that is relevant to comprehension of the catalytic cycle and for structure-based drug-design approaches is reported.
Open  access
access
 access
accessCrystal structures of the UDP glycosyltransferase PaGT3 in complex with various ligands are reported. This study sheds light on how the enzyme accommodates sugar acceptors of different shapes and sizes for successful glycosylation.
Open  access
access
 access
accessX-ray crystal structures of E. coli ribonuclease HI in complex with Mg2+ ions or with a Zn2+ ion reveal the pivotal role of a highly conserved histidine residue.


 journal menu
journal menu