issue contents
September 2024 issue
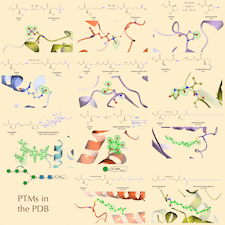
Cover illustration: Post-translational modifications in the Protein Data Bank [Schofield et al. (2024), Acta Cryst. D80, 647–660]. Proteins frequently undergo post-translational modifications, which involve the addition or alteration of chemical groups on specific amino acids. These modifications can have a significant impact on protein function by affecting their activity, location within the cell, interactions with other proteins, and overall stability. PTMs are essential for regulating various cellular processes, but capturing these modifications accurately in protein structural studies is challenging. The review by Schofield et al. focuses on the role of PTMs in influencing protein structure and function, examines the frequency and representation of these modifications in the Protein Data Bank, and highlights examples that can serve as valuable references for researchers in structural biology and protein modeling.
CCP4
 access
accessresearch papers
 access
access access
access access
access access
access
 journal menu
journal menu