

metal-organic compounds
Dibromido[bis(η5-cyclopentadienyl)dimethylsilane]zirconium(IV)
aDepartment of General and Inorganic Chemistry, Faculty of Chemical Technology, University of Pardubice, Nám. Čs. legií 565, Pardubice 532 10, Czech Republic
*Correspondence e-mail: [email protected]
The title molecule, [ZrBr2(C12H14Si)], possesses a crystallographically imposed twofold rotational symmetry with the rotation axis passing through the Zr and Si atoms. The ZrIV centre is in a distorted tetrahedral environment defined by two Cp rings of chelating organic ligands and two Br anions. Two five-membered rings form a dihedral angle of 59.7 (2)°. Unequal Zr—C bonds [2.471 (3)–2.556 (3) Å] in the molecule indicate that the interaction of the central metal with the [(C5H4)2SiMe2]2− ligand contains noticeable η3-allyl and η2-olefin contributions.
Related literature
For related ansa-zirconocenes, see, for example: Bajgur et al. (1985![Bajgur, C. S., Tikkanen, W. R. & Petersen, J. L. (1985). Inorg. Chem. 24, 2539-2546. [Bajgur, C. S., Tikkanen, W. R. & Petersen, J. L. (1985). Inorg. Chem. 24, 2539-2546.]](../../../../../../logos/arrows/e_arr.gif) ), Borrelli et al. (2002
), Borrelli et al. (2002![Borrelli, M., Busico, V., Cipulo, R. & Ronca, S. (2002). Macromolecules, 35, 2835-2844. [Borrelli, M., Busico, V., Cipulo, R. & Ronca, S. (2002). Macromolecules, 35, 2835-2844.]](../../../../../../logos/arrows/e_arr.gif) ).
).
Experimental
Crystal data
|
|
Data collection: COLLECT (Hooft, 1998![Hooft, R. W. (1998). COLLECT. Nonius BV, Delft, The Netherlands. [Hooft, R. W. (1998). COLLECT. Nonius BV, Delft, The Netherlands.]](../../../../../../logos/arrows/e_arr.gif) ) and DENZO (Otwinowski & Minor, 1997
) and DENZO (Otwinowski & Minor, 1997![Otwinowski, Z. & Minor, W. (1997). Methods in Enzymology, Vol. 276, Macromolecular Crystallography, Part A, edited by C. W. Carter Jr & R. M. Sweet, pp. 307-326. New York: Academic Press. [Otwinowski, Z. & Minor, W. (1997). Methods in Enzymology, Vol. 276, Macromolecular Crystallography, Part A, edited by C. W. Carter Jr & R. M. Sweet, pp. 307-326. New York: Academic Press.]](../../../../../../logos/arrows/e_arr.gif) ); cell refinement: COLLECT and DENZO; data reduction: COLLECT and DENZO; program(s) used to solve structure: SIR92 (Altomare et al., 1994
); cell refinement: COLLECT and DENZO; data reduction: COLLECT and DENZO; program(s) used to solve structure: SIR92 (Altomare et al., 1994![Altomare, A., Cascarano, G., Giacovazzo, C., Guagliardi, A., Burla, M. C., Polidori, G. & Camalli, M. (1994). J. Appl. Cryst. 27, 435. [Altomare, A., Cascarano, G., Giacovazzo, C., Guagliardi, A., Burla, M. C., Polidori, G. & Camalli, M. (1994). J. Appl. Cryst. 27, 435.]](../../../../../../logos/arrows/e_arr.gif) ); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008
); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008![Sheldrick, G. M. (2008). Acta Cryst. A64, 112-122. [Sheldrick, G. M. (2008). Acta Cryst. A64, 112-122.]](../../../../../../logos/arrows/e_arr.gif) ); molecular graphics: PLATON (Spek, 2003
); molecular graphics: PLATON (Spek, 2003![Spek, A. L. (2003). J. Appl. Cryst. 36, 7-13. [Spek, A. L. (2003). J. Appl. Cryst. 36, 7-13.]](../../../../../../logos/arrows/e_arr.gif) ); software used to prepare material for publication: enCIFer (Allen et al., 2004
); software used to prepare material for publication: enCIFer (Allen et al., 2004![Allen, F. H., Johnson, O., Shields, G. P., Smith, B. R. & Towler, M. (2004). J. Appl. Cryst. 37, 335-338. [Allen, F. H., Johnson, O., Shields, G. P., Smith, B. R. & Towler, M. (2004). J. Appl. Cryst. 37, 335-338.]](../../../../../../logos/arrows/e_arr.gif) ).
).
Supporting information
contains datablocks I, global. DOI: 10.1107/S1600536808040713/cv2494sup1.cif
Structure factors: contains datablock I. DOI: 10.1107/S1600536808040713/cv2494Isup2.hkl
Compound (I) was prepared by bromination of analogous chloride derivative using boron tribromide. To the starting complex [ZrCl2(η5-C5H4)2SiMe2] (0.2 g; 0.57 mol) in 20 ml of dichloromethane 0.04 ml (0.42 mmol) of BBr3 was added. The color of the reaction mixture immediatelly turned to green and it was stirred for additional 2 h at 293 K. The solvent was evaporated in vacuum, solid residue was washed with hexane (2x5 ml) and vacuum-dried. of crude product at 10 -3 Pa and 475 K gave 0.105 g (42%) of (I). Crystals of (I) suitable for X-ray diffraction measurements were grown during slow evaporation of chloroform solution at 273 K.
All H atoms were positioned geometrically and refined as riding on their parent C atoms, with C—H = 0.93 Å, Uiso(H) = 1.2Ue.g(C) and C—H = 0.96 Å, Uiso(H) = 1.5Ue.g(C) for cyclopentadienyl and methyl H atoms, respectively.
Data collection: COLLECT (Hooft, 1998) and DENZO (Otwinowski & Minor, 1997); cell COLLECT (Hooft, 1998) and DENZO (Otwinowski & Minor, 1997); data reduction: COLLECT (Hooft, 1998) and DENZO (Otwinowski & Minor, 1997); program(s) used to solve structure: SIR92 (Altomare et al., 1994); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008); molecular graphics: PLATON (Spek, 2003); software used to prepare material for publication: enCIFer (Allen et al., 2004).
![[Figure 1]](./cv2494fig1thm.gif) | Fig. 1. Perspective view of (I), shown with 30% probability displacement ellipsoids (arbitrary spheres for H atoms) [symmetry code: (a) -x, y, 1/2-z.] |
| [ZrBr2(C12H14Si)] | F(000) = 840 |
| Mr = 437.36 | Dx = 2.079 Mg m−3 |
| Monoclinic, C2/c | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: -C 2yc | Cell parameters from 6471 reflections |
| a = 13.6160 (4) Å | θ = 1–27.5° |
| b = 10.0990 (2) Å | µ = 6.57 mm−1 |
| c = 10.9770 (3) Å | T = 120 K |
| β = 112.2540 (12)° | Prism, yellow |
| V = 1396.99 (6) Å3 | 0.4 × 0.3 × 0.22 mm |
| Z = 4 |
| Nonius KappaCCD area-detector diffractometer | 1602 independent reflections |
| Radiation source: fine-focus sealed tube | 1560 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.047 |
| Detector resolution: 9.091 pixels mm-1 | θmax = 27.5°, θmin = 2.6° |
| ϕ and ω scans to fill the Ewald sphere | h = −17→17 |
| Absorption correction: gaussian (Coppens et al., 1970) | k = −13→13 |
| Tmin = 0.137, Tmax = 0.271 | l = −14→14 |
| 9698 measured reflections |
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.024 | H-atom parameters constrained |
| wR(F2) = 0.063 | w = 1/[σ2(Fo2) + (0.0323P)2 + 3.2435P] where P = (Fo2 + 2Fc2)/3 |
| S = 1.14 | (Δ/σ)max < 0.001 |
| 1602 reflections | Δρmax = 0.69 e Å−3 |
| 76 parameters | Δρmin = −1.07 e Å−3 |
| 0 restraints | Extinction correction: SHELXL97 (Sheldrick, 1997), Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| Primary atom site location: structure-invariant direct methods | Extinction coefficient: 0.0044 (3) |
| [ZrBr2(C12H14Si)] | V = 1396.99 (6) Å3 |
| Mr = 437.36 | Z = 4 |
| Monoclinic, C2/c | Mo Kα radiation |
| a = 13.6160 (4) Å | µ = 6.57 mm−1 |
| b = 10.0990 (2) Å | T = 120 K |
| c = 10.9770 (3) Å | 0.4 × 0.3 × 0.22 mm |
| β = 112.2540 (12)° |
| Nonius KappaCCD area-detector diffractometer | 1602 independent reflections |
| Absorption correction: gaussian (Coppens et al., 1970) | 1560 reflections with I > 2σ(I) |
| Tmin = 0.137, Tmax = 0.271 | Rint = 0.047 |
| 9698 measured reflections |
| R[F2 > 2σ(F2)] = 0.024 | 0 restraints |
| wR(F2) = 0.063 | H-atom parameters constrained |
| S = 1.14 | Δρmax = 0.69 e Å−3 |
| 1602 reflections | Δρmin = −1.07 e Å−3 |
| 76 parameters |
Experimental. Spectroscopic analysis: 1H NMR (CDCl3, δ, p.p.m.): 7.15 (m, 4H), 5.97 (m, 4H), 0.73 (s, 6H). 13C NMR (CDCl3, δ, p.p.m.): -5.2, 1, 114.3, 115.8, 128.9. IR (KBr disc, cm-1): 3115 (m), 3099 (m), 3074 (m), 2964 (m), 2958 (m), 1448 (m), 1409 (w), 1400 (s), 1370 (s), 1362 (m), 1324 (m), 1314 (w), 1260 (s), 1200 (w), 1179 (s), 1168 (s), 1069 (s), 1048 (s), 1020 (s), 942 (m), 902 (s), 870 (s), 870 (s), 813 (s), 680 (s), 633 (s), 555 (m), 502 (m), 454 (s), 420 (s), 384 (m), 347 (s); UV-Vis (CH2Cl2, maxima at nm): 470, 377, 313, 233. Elemental analysis, calculated for C12H14Br2SiZr: C 32.96, H 3.23; found: C 32.61, H 3.14%. |
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
| x | y | z | Uiso*/Ueq | ||
| Zr1 | 0.0000 | 0.31648 (3) | 0.2500 | 0.01164 (12) | |
| Br1 | 0.155827 (19) | 0.48477 (3) | 0.33572 (2) | 0.02090 (12) | |
| Si1 | 0.0000 | −0.01625 (9) | 0.2500 | 0.0160 (2) | |
| C1 | 0.0077 (2) | 0.1116 (2) | 0.1295 (2) | 0.0170 (5) | |
| C2 | −0.0792 (2) | 0.1870 (3) | 0.0443 (3) | 0.0206 (5) | |
| H2 | −0.1504 | 0.1647 | 0.0193 | 0.025* | |
| C3 | −0.0400 (3) | 0.3017 (3) | 0.0033 (3) | 0.0250 (6) | |
| H3 | −0.0807 | 0.3658 | −0.0547 | 0.030* | |
| C4 | 0.0707 (3) | 0.3014 (3) | 0.0658 (3) | 0.0247 (6) | |
| H4 | 0.1166 | 0.3654 | 0.0567 | 0.030* | |
| C5 | 0.1003 (2) | 0.1861 (3) | 0.1454 (3) | 0.0207 (5) | |
| H5 | 0.1693 | 0.1630 | 0.1993 | 0.025* | |
| C6 | 0.1245 (2) | −0.1135 (3) | 0.3168 (3) | 0.0247 (6) | |
| H6A | 0.1191 | −0.1775 | 0.3787 | 0.037* | |
| H6B | 0.1830 | −0.0551 | 0.3603 | 0.037* | |
| H6C | 0.1359 | −0.1583 | 0.2462 | 0.037* |
| U11 | U22 | U33 | U12 | U13 | U23 | |
| Zr1 | 0.01466 (17) | 0.01152 (18) | 0.01051 (17) | 0.000 | 0.00678 (12) | 0.000 |
| Br1 | 0.01687 (16) | 0.02362 (18) | 0.02273 (17) | −0.00544 (9) | 0.00811 (11) | −0.00413 (9) |
| Si1 | 0.0237 (5) | 0.0120 (4) | 0.0156 (5) | 0.000 | 0.0112 (4) | 0.000 |
| C1 | 0.0261 (12) | 0.0145 (11) | 0.0147 (11) | 0.0005 (10) | 0.0125 (9) | −0.0021 (9) |
| C2 | 0.0282 (13) | 0.0187 (13) | 0.0134 (11) | −0.0003 (10) | 0.0062 (10) | −0.0028 (9) |
| C3 | 0.0467 (17) | 0.0181 (13) | 0.0122 (11) | 0.0029 (12) | 0.0134 (11) | 0.0003 (10) |
| C4 | 0.0435 (16) | 0.0189 (13) | 0.0224 (13) | −0.0035 (12) | 0.0245 (12) | −0.0017 (10) |
| C5 | 0.0272 (13) | 0.0193 (13) | 0.0227 (13) | 0.0015 (10) | 0.0176 (11) | −0.0012 (10) |
| C6 | 0.0321 (14) | 0.0228 (14) | 0.0220 (13) | 0.0080 (12) | 0.0135 (11) | 0.0048 (11) |
| Zr1—C5i | 2.471 (3) | Si1—C1i | 1.880 (3) |
| Zr1—C5 | 2.471 (3) | Si1—C1 | 1.880 (3) |
| Zr1—C2i | 2.476 (3) | C1—C2 | 1.420 (4) |
| Zr1—C2 | 2.476 (3) | C1—C5 | 1.422 (4) |
| Zr1—C1 | 2.480 (2) | C2—C3 | 1.418 (4) |
| Zr1—C1i | 2.480 (2) | C2—H2 | 0.9300 |
| Zr1—C4i | 2.552 (3) | C3—C4 | 1.400 (4) |
| Zr1—C4 | 2.552 (3) | C3—H3 | 0.9300 |
| Zr1—C3 | 2.556 (3) | C4—C5 | 1.420 (4) |
| Zr1—C3i | 2.556 (3) | C4—H4 | 0.9300 |
| Zr1—Br1i | 2.6007 (3) | C5—H5 | 0.9300 |
| Zr1—Br1 | 2.6007 (3) | C6—H6A | 0.9600 |
| Si1—C6i | 1.853 (3) | C6—H6B | 0.9600 |
| Si1—C6 | 1.853 (3) | C6—H6C | 0.9600 |
| C5i—Zr1—C5 | 115.60 (12) | C3i—Zr1—Br1i | 103.82 (7) |
| C5i—Zr1—C2i | 54.64 (9) | C5i—Zr1—Br1 | 134.35 (7) |
| C5—Zr1—C2i | 90.90 (9) | C5—Zr1—Br1 | 89.86 (6) |
| C5i—Zr1—C2 | 90.90 (9) | C2i—Zr1—Br1 | 90.07 (6) |
| C5—Zr1—C2 | 54.64 (9) | C2—Zr1—Br1 | 133.57 (7) |
| C2i—Zr1—C2 | 116.22 (12) | C1—Zr1—Br1 | 123.01 (6) |
| C5i—Zr1—C1 | 86.89 (8) | C1i—Zr1—Br1 | 123.08 (6) |
| C5—Zr1—C1 | 33.37 (8) | C4i—Zr1—Br1 | 104.65 (7) |
| C2i—Zr1—C1 | 87.36 (9) | C4—Zr1—Br1 | 79.92 (7) |
| C2—Zr1—C1 | 33.31 (9) | C3—Zr1—Br1 | 103.82 (7) |
| C5i—Zr1—C1i | 33.37 (8) | C3i—Zr1—Br1 | 80.64 (7) |
| C5—Zr1—C1i | 86.89 (8) | Br1i—Zr1—Br1 | 98.387 (17) |
| C2i—Zr1—C1i | 33.31 (9) | C6i—Si1—C6 | 116.0 (2) |
| C2—Zr1—C1i | 87.36 (9) | C6i—Si1—C1i | 110.88 (12) |
| C1—Zr1—C1i | 66.87 (11) | C6—Si1—C1i | 111.83 (12) |
| C5i—Zr1—C4i | 32.79 (9) | C6i—Si1—C1 | 111.83 (12) |
| C5—Zr1—C4i | 141.00 (9) | C6—Si1—C1 | 110.87 (12) |
| C2i—Zr1—C4i | 53.88 (9) | C1i—Si1—C1 | 93.26 (15) |
| C2—Zr1—C4i | 121.77 (9) | C2—C1—C5 | 106.1 (2) |
| C1—Zr1—C4i | 118.55 (9) | C2—C1—Si1 | 125.24 (19) |
| C1i—Zr1—C4i | 54.73 (8) | C5—C1—Si1 | 124.1 (2) |
| C5i—Zr1—C4 | 141.00 (9) | C2—C1—Zr1 | 73.19 (14) |
| C5—Zr1—C4 | 32.80 (9) | C5—C1—Zr1 | 72.99 (14) |
| C2i—Zr1—C4 | 121.77 (9) | Si1—C1—Zr1 | 99.94 (10) |
| C2—Zr1—C4 | 53.88 (9) | C3—C2—C1 | 109.1 (2) |
| C1—Zr1—C4 | 54.73 (8) | C3—C2—Zr1 | 76.77 (15) |
| C1i—Zr1—C4 | 118.55 (9) | C1—C2—Zr1 | 73.51 (14) |
| C4i—Zr1—C4 | 173.17 (13) | C3—C2—H2 | 125.5 |
| C5i—Zr1—C3 | 121.83 (10) | C1—C2—H2 | 125.5 |
| C5—Zr1—C3 | 53.87 (9) | Zr1—C2—H2 | 116.2 |
| C2i—Zr1—C3 | 141.26 (9) | C4—C3—C2 | 107.9 (2) |
| C2—Zr1—C3 | 32.69 (9) | C4—C3—Zr1 | 73.94 (15) |
| C1—Zr1—C3 | 54.63 (8) | C2—C3—Zr1 | 70.54 (15) |
| C1i—Zr1—C3 | 118.78 (9) | C4—C3—H3 | 126.0 |
| C4i—Zr1—C3 | 147.45 (10) | C2—C3—H3 | 126.0 |
| C4—Zr1—C3 | 31.81 (10) | Zr1—C3—H3 | 121.2 |
| C5i—Zr1—C3i | 53.87 (9) | C3—C4—C5 | 107.8 (2) |
| C5—Zr1—C3i | 121.83 (10) | C3—C4—Zr1 | 74.25 (15) |
| C2i—Zr1—C3i | 32.69 (9) | C5—C4—Zr1 | 70.48 (14) |
| C2—Zr1—C3i | 141.25 (9) | C3—C4—H4 | 126.1 |
| C1—Zr1—C3i | 118.78 (9) | C5—C4—H4 | 126.1 |
| C1i—Zr1—C3i | 54.63 (8) | Zr1—C4—H4 | 120.9 |
| C4i—Zr1—C3i | 31.81 (10) | C4—C5—C1 | 109.0 (2) |
| C4—Zr1—C3i | 147.45 (10) | C4—C5—Zr1 | 76.72 (15) |
| C3—Zr1—C3i | 173.31 (13) | C1—C5—Zr1 | 73.64 (14) |
| C5i—Zr1—Br1i | 89.86 (6) | C4—C5—H5 | 125.5 |
| C5—Zr1—Br1i | 134.35 (7) | C1—C5—H5 | 125.5 |
| C2i—Zr1—Br1i | 133.57 (7) | Zr1—C5—H5 | 116.1 |
| C2—Zr1—Br1i | 90.07 (6) | Si1—C6—H6A | 109.5 |
| C1—Zr1—Br1i | 123.08 (6) | Si1—C6—H6B | 109.5 |
| C1i—Zr1—Br1i | 123.01 (6) | H6A—C6—H6B | 109.5 |
| C4i—Zr1—Br1i | 79.92 (7) | Si1—C6—H6C | 109.5 |
| C4—Zr1—Br1i | 104.65 (7) | H6A—C6—H6C | 109.5 |
| C3—Zr1—Br1i | 80.64 (7) | H6B—C6—H6C | 109.5 |
| Symmetry code: (i) −x, y, −z+1/2. |
Experimental details
| Crystal data | |
| Chemical formula | [ZrBr2(C12H14Si)] |
| Mr | 437.36 |
| Crystal system, space group | Monoclinic, C2/c |
| Temperature (K) | 120 |
| a, b, c (Å) | 13.6160 (4), 10.0990 (2), 10.9770 (3) |
| β (°) | 112.2540 (12) |
| V (Å3) | 1396.99 (6) |
| Z | 4 |
| Radiation type | Mo Kα |
| µ (mm−1) | 6.57 |
| Crystal size (mm) | 0.4 × 0.3 × 0.22 |
| Data collection | |
| Diffractometer | Nonius KappaCCD area-detector diffractometer |
| Absorption correction | Gaussian (Coppens et al., 1970) |
| Tmin, Tmax | 0.137, 0.271 |
| No. of measured, independent and observed [I > 2σ(I)] reflections | 9698, 1602, 1560 |
| Rint | 0.047 |
| (sin θ/λ)max (Å−1) | 0.649 |
| Refinement | |
| R[F2 > 2σ(F2)], wR(F2), S | 0.024, 0.063, 1.14 |
| No. of reflections | 1602 |
| No. of parameters | 76 |
| H-atom treatment | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 0.69, −1.07 |
Computer programs: COLLECT (Hooft, 1998) and DENZO (Otwinowski & Minor, 1997), SIR92 (Altomare et al., 1994), SHELXL97 (Sheldrick, 2008), PLATON (Spek, 2003), enCIFer (Allen et al., 2004).
| Zr1—Cg1 | 2.199 (1) | C6—Si1—C6i | 116.0 (1) |
| Zr1—Br1 | 2.6007 (4) | C1—Si1—C1i | 93.2 (1) |
| Cg1—Zr1—Cg1i | 125.96 (5) | Br1—Zr1—Br1i | 98.39 (1) |
| Symmetry code: (i) -x, y, 1/2-z. Cg1 is the centroid of atoms C1–C5. |
Acknowledgements
The authors thank the Ministry of Education, Youth and Sports of the Czech Republic for financial support of this work within the framework of research project MSM 0021627501.
References
 Allen, F. H., Johnson, O., Shields, G. P., Smith, B. R. & Towler, M. (2004). J. Appl. Cryst. 37, 335–338. Web of Science CrossRef CAS IUCr Journals Google Scholar
Allen, F. H., Johnson, O., Shields, G. P., Smith, B. R. & Towler, M. (2004). J. Appl. Cryst. 37, 335–338. Web of Science CrossRef CAS IUCr Journals Google Scholar
 Altomare, A., Cascarano, G., Giacovazzo, C., Guagliardi, A., Burla, M. C., Polidori, G. & Camalli, M. (1994). J. Appl. Cryst. 27, 435. CrossRef Web of Science IUCr Journals Google Scholar
Altomare, A., Cascarano, G., Giacovazzo, C., Guagliardi, A., Burla, M. C., Polidori, G. & Camalli, M. (1994). J. Appl. Cryst. 27, 435. CrossRef Web of Science IUCr Journals Google Scholar
 Bajgur, C. S., Tikkanen, W. R. & Petersen, J. L. (1985). Inorg. Chem. 24, 2539–2546. CSD CrossRef CAS Web of Science Google Scholar
Bajgur, C. S., Tikkanen, W. R. & Petersen, J. L. (1985). Inorg. Chem. 24, 2539–2546. CSD CrossRef CAS Web of Science Google Scholar
 Borrelli, M., Busico, V., Cipulo, R. & Ronca, S. (2002). Macromolecules, 35, 2835–2844. Web of Science CrossRef CAS Google Scholar
Borrelli, M., Busico, V., Cipulo, R. & Ronca, S. (2002). Macromolecules, 35, 2835–2844. Web of Science CrossRef CAS Google Scholar
 Coppens, P. (1970). Crystallographic Computing, edited by F. R. Ahmed, S. R. Hall & C. P. Huber, pp. 255–270. Copenhagen: Munksgaard. Google Scholar
Coppens, P. (1970). Crystallographic Computing, edited by F. R. Ahmed, S. R. Hall & C. P. Huber, pp. 255–270. Copenhagen: Munksgaard. Google Scholar
 Hooft, R. W. (1998). COLLECT. Nonius BV, Delft, The Netherlands. Google Scholar
Hooft, R. W. (1998). COLLECT. Nonius BV, Delft, The Netherlands. Google Scholar
 Otwinowski, Z. & Minor, W. (1997). Methods in Enzymology, Vol. 276, Macromolecular Crystallography, Part A, edited by C. W. Carter Jr & R. M. Sweet, pp. 307–326. New York: Academic Press. Google Scholar
Otwinowski, Z. & Minor, W. (1997). Methods in Enzymology, Vol. 276, Macromolecular Crystallography, Part A, edited by C. W. Carter Jr & R. M. Sweet, pp. 307–326. New York: Academic Press. Google Scholar
 Sheldrick, G. M. (2008). Acta Cryst. A64, 112–122. Web of Science CrossRef CAS IUCr Journals Google Scholar
Sheldrick, G. M. (2008). Acta Cryst. A64, 112–122. Web of Science CrossRef CAS IUCr Journals Google Scholar
 Spek, A. L. (2003). J. Appl. Cryst. 36, 7–13. Web of Science CrossRef CAS IUCr Journals Google Scholar
Spek, A. L. (2003). J. Appl. Cryst. 36, 7–13. Web of Science CrossRef CAS IUCr Journals Google Scholar
This is an open-access article distributed under the terms of the Creative Commons Attribution (CC-BY) Licence, which permits unrestricted use, distribution, and reproduction in any medium, provided the original authors and source are cited.


 journal menu
journal menu











 access
access![[Scheme 1]](cv2494scheme1.gif)
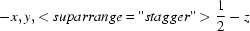 {\script{1\over 2}}-z]" class="img_align_bottom" style="max-width: 100%; height: auto; width: 250px;" />.
{\script{1\over 2}}-z]" class="img_align_bottom" style="max-width: 100%; height: auto; width: 250px;" />. 



Metallocene complexes of Group 4 are intensively investigated as very efficient and selective olefin polymerization and co-polymerization catalysts (Borrelli et al., 2002). The most used strategy for the modification of catalytic properties of these metallocenes is the substitution at the cyclopentadienyl rings. Incorporation of an interannular bridge connecting both cyclopentadienyl rings in the molecule of metallocene leads to a broad class of complexes known as ansa-metallocenes. As part of an investigation of the influence of ring substitution on the properties of cyclopentadienyl complexes, the title compound, (I), was prepared, spectroscopically characterized and its structure determined
A perspective view of molecular structure of (I) is shown in Figure 1 with appropriate atom labeling scheme, selected bond distances and angles are summarized in Table 1. In the molecule of (I) the Zr atom is pseudotetrahedrally coordinated by two η5-bonded Cp rings and two Br atoms with geometry constrained by crystallographic twofold rotation axis, which bisects Br1—Zr—Br1a angle and passes through the metal and Si atom. The molecular parameters are comparable to those reported for [ZrCl2(η5-C5H4)2SiMe2] (Bajgur et al., 1985). On the inspection of parameters associated with Cp rings, the deviation from the ideal η5-bonding fashion could be observed. The C3—C4 bond distance of 1.400 (6) Å is shorter than the remaining C—C bonds (average of 1.42 Å). Similarly Zr1—C3 and Zr1—C4 bonds are longer [2.556 (3) Å and 2.552 (4) Å, respectively] than remaining metal-carbon bonds (average of 2.475 Å). These facts indicate the presence of η3-allyl and η2-olefin bonding fashion of Cp ring rather than η5-cyclopentadienyl bonding pattern.