Buy article online - an online subscription or single-article purchase is required to access this article.

Three photoluminescent complexes containing either Zn
II or Cd
II have been synthesized and their structures determined. Bis[4-amino-3,5-bis(pyridin-2-yl)-1,2,4-triazole-κ
2N1,
N5]bis(dicyanamido-κ
N1)zinc(II), [Zn(C
12H
10N
6)
2(C
2N
3)
2], (I), bis[4-amino-3,5-bis(pyridin-2-yl)-1,2,4-triazole-κ
2N1,
N5]bis(dicyanamido-κ
N1)cadmium(II), [Cd(C
12H
10N
6)
2(C
2N
3)
2], (II), and bis[4-amino-3,5-bis(pyridin-2-yl)-1,2,4-triazole-κ
2N1,
N5]bis(tricyanomethanido-κ
N1)cadmium(II), [Cd(C
12H
10N
6)
2(C
4N
3)
2], (III), all crystallize in the space group
P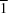
, with the metal centres lying on centres of inversion, but neither analogues (I) and (II) nor Cd
II complexes (II) and (III) are isomorphous. A combination of N—H

N and C—H

N hydrogen bonds and π–π stacking interactions generates three-dimensional framework structures in (I) and (II), and a sheet structure in (III). The photoluminescence spectra of (I)–(III) indicate that the energies of the π–π* transitions in the coordinated triazole ligand are modified by minor changes of the ligand geometry associated with coordination to the metal centres.
Supporting information
CCDC references: 1039282; 1039280; 1039281
For all structures, data collection: APEX2 (Bruker, 2009); cell refinement: APEX2 and SAINT (Bruker, 2009); data reduction: SAINT (Bruker, 2009); program(s) used to solve structure: SHELXS97 (Sheldrick, 2008); program(s) used to refine structure: SHELXL2014 (Sheldrick, 2015); molecular graphics: PLATON (Spek, 2009); software used to prepare material for publication: SHELXL2014 (Sheldrick, 2015) and PLATON (Spek, 2009).
Bis[4-amino-3,5-bis(pyridin-2-yl)-1,2,4-triazole-
κ2N1,
N5]bis(dicyanamido-
κN1)zinc(II) (I)
top
Crystal data top
| [Zn(C12H10N6)2(C2N3)2] | Z = 1 |
| Mr = 674.01 | F(000) = 344 |
| Triclinic, P1 | Dx = 1.575 Mg m−3 |
| a = 8.4211 (11) Å | Mo Kα radiation, λ = 0.71073 Å |
| b = 9.5379 (12) Å | Cell parameters from 3720 reflections |
| c = 9.7491 (12) Å | θ = 2.2–29.0° |
| α = 83.531 (6)° | µ = 0.92 mm−1 |
| β = 66.048 (4)° | T = 296 K |
| γ = 85.922 (6)° | Block, colourless |
| V = 710.78 (16) Å3 | 0.25 × 0.20 × 0.12 mm |
Data collection top
Bruker APEXII CCD
diffractometer | 3716 independent reflections |
| Radiation source: fine-focus sealed tube | 2731 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.033 |
| Detector resolution: 9.091 pixels mm-1 | θmax = 29.0°, θmin = 3.0° |
| φ & ω scans | h = −11→11 |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | k = −12→8 |
| Tmin = 0.791, Tmax = 0.895 | l = −12→13 |
| 13438 measured reflections | |
Refinement top
| Refinement on F2 | 0 restraints |
| Least-squares matrix: full | Hydrogen site location: mixed |
| R[F2 > 2σ(F2)] = 0.045 | H-atom parameters constrained |
| wR(F2) = 0.117 | w = 1/[σ2(Fo2) + (0.0583P)2 + 0.1896P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.02 | (Δ/σ)max < 0.001 |
| 3716 reflections | Δρmax = 0.72 e Å−3 |
| 214 parameters | Δρmin = −0.33 e Å−3 |
Special details top
Geometry. All esds (except the esd in the dihedral angle between two l.s. planes)
are estimated using the full covariance matrix. The cell esds are taken
into account individually in the estimation of esds in distances, angles
and torsion angles; correlations between esds in cell parameters are only
used when they are defined by crystal symmetry. An approximate (isotropic)
treatment of cell esds is used for estimating esds involving l.s. planes. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Zn1 | 0.5000 | 0.5000 | 0.5000 | 0.04045 (15) | |
| N1 | 0.4959 (2) | 0.7099 (2) | 0.4169 (2) | 0.0399 (4) | |
| N2 | 0.4040 (2) | 0.7887 (2) | 0.3461 (2) | 0.0404 (4) | |
| C3 | 0.4821 (3) | 0.9105 (2) | 0.2965 (2) | 0.0371 (5) | |
| N4 | 0.6213 (2) | 0.91108 (19) | 0.3346 (2) | 0.0365 (4) | |
| C5 | 0.6253 (3) | 0.7839 (2) | 0.4110 (2) | 0.0361 (5) | |
| N31 | 0.5021 (3) | 1.1478 (2) | 0.1787 (2) | 0.0462 (5) | |
| C32 | 0.4236 (3) | 1.0238 (2) | 0.2106 (3) | 0.0390 (5) | |
| C33 | 0.2939 (3) | 0.9958 (3) | 0.1648 (3) | 0.0433 (5) | |
| H33 | 0.2419 | 0.9084 | 0.1910 | 0.052* | |
| C34 | 0.2442 (3) | 1.1006 (3) | 0.0798 (3) | 0.0505 (6) | |
| H34 | 0.1582 | 1.0853 | 0.0467 | 0.061* | |
| C35 | 0.3246 (4) | 1.2285 (3) | 0.0446 (3) | 0.0554 (7) | |
| H35 | 0.2939 | 1.3010 | −0.0129 | 0.067* | |
| C36 | 0.4508 (4) | 1.2475 (3) | 0.0958 (3) | 0.0550 (7) | |
| H36 | 0.5036 | 1.3346 | 0.0716 | 0.066* | |
| N41 | 0.7445 (3) | 1.0182 (2) | 0.3006 (2) | 0.0481 (5) | |
| H41A | 0.6988 | 1.0972 | 0.2605 | 0.058* | |
| H41B | 0.8403 | 0.9862 | 0.2187 | 0.058* | |
| N51 | 0.6971 (2) | 0.5930 (2) | 0.5503 (2) | 0.0381 (4) | |
| C52 | 0.7390 (3) | 0.7255 (2) | 0.4858 (2) | 0.0372 (5) | |
| C53 | 0.8705 (3) | 0.7967 (3) | 0.4976 (3) | 0.0460 (6) | |
| H53 | 0.8995 | 0.8871 | 0.4502 | 0.055* | |
| C54 | 0.9575 (3) | 0.7296 (3) | 0.5815 (3) | 0.0511 (6) | |
| H54 | 1.0469 | 0.7744 | 0.5905 | 0.061* | |
| C55 | 0.9111 (3) | 0.5969 (3) | 0.6514 (3) | 0.0510 (6) | |
| H55 | 0.9658 | 0.5518 | 0.7108 | 0.061* | |
| C56 | 0.7813 (3) | 0.5313 (3) | 0.6319 (3) | 0.0459 (6) | |
| H56 | 0.7516 | 0.4404 | 0.6775 | 0.055* | |
| N11 | 0.1643 (4) | 0.5921 (3) | 0.9902 (3) | 0.0752 (8) | |
| C12 | 0.2311 (3) | 0.5779 (3) | 0.8451 (3) | 0.0460 (6) | |
| N13 | 0.3017 (3) | 0.5464 (2) | 0.7253 (2) | 0.0506 (5) | |
| C14 | 0.0802 (3) | 0.7104 (3) | 1.0423 (3) | 0.0478 (6) | |
| N15 | 0.0100 (3) | 0.8017 (3) | 1.1071 (3) | 0.0663 (7) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Zn1 | 0.0410 (2) | 0.0411 (2) | 0.0436 (2) | −0.00772 (15) | −0.02016 (18) | −0.00485 (16) |
| N1 | 0.0385 (10) | 0.0442 (11) | 0.0415 (11) | −0.0072 (8) | −0.0192 (9) | −0.0055 (8) |
| N2 | 0.0374 (10) | 0.0429 (10) | 0.0435 (11) | −0.0057 (8) | −0.0181 (9) | −0.0038 (8) |
| C3 | 0.0333 (11) | 0.0412 (12) | 0.0351 (11) | −0.0029 (9) | −0.0097 (9) | −0.0112 (9) |
| N4 | 0.0297 (9) | 0.0394 (10) | 0.0393 (10) | −0.0047 (7) | −0.0102 (8) | −0.0114 (8) |
| C5 | 0.0320 (11) | 0.0409 (12) | 0.0342 (11) | −0.0047 (9) | −0.0097 (9) | −0.0110 (9) |
| N31 | 0.0428 (11) | 0.0402 (11) | 0.0521 (12) | −0.0003 (8) | −0.0136 (10) | −0.0121 (9) |
| C32 | 0.0353 (12) | 0.0411 (12) | 0.0357 (12) | 0.0015 (9) | −0.0074 (10) | −0.0123 (9) |
| C33 | 0.0410 (13) | 0.0436 (13) | 0.0442 (13) | −0.0001 (10) | −0.0148 (11) | −0.0092 (10) |
| C34 | 0.0510 (15) | 0.0540 (15) | 0.0480 (14) | 0.0081 (12) | −0.0208 (12) | −0.0129 (12) |
| C35 | 0.0576 (16) | 0.0480 (15) | 0.0553 (16) | 0.0134 (12) | −0.0193 (14) | −0.0060 (12) |
| C36 | 0.0538 (16) | 0.0365 (12) | 0.0654 (17) | 0.0011 (11) | −0.0143 (14) | −0.0065 (12) |
| N41 | 0.0411 (11) | 0.0428 (11) | 0.0605 (13) | −0.0130 (9) | −0.0185 (10) | −0.0052 (10) |
| N51 | 0.0349 (10) | 0.0461 (10) | 0.0359 (10) | 0.0006 (8) | −0.0148 (8) | −0.0139 (8) |
| C52 | 0.0318 (11) | 0.0467 (12) | 0.0328 (11) | −0.0020 (9) | −0.0096 (9) | −0.0152 (9) |
| C53 | 0.0382 (13) | 0.0567 (15) | 0.0439 (13) | −0.0097 (11) | −0.0130 (11) | −0.0161 (11) |
| C54 | 0.0362 (13) | 0.0755 (18) | 0.0482 (15) | −0.0050 (12) | −0.0197 (11) | −0.0199 (13) |
| C55 | 0.0429 (14) | 0.0699 (18) | 0.0470 (14) | 0.0055 (12) | −0.0233 (12) | −0.0162 (13) |
| C56 | 0.0424 (13) | 0.0555 (15) | 0.0425 (13) | 0.0031 (11) | −0.0184 (11) | −0.0130 (11) |
| N11 | 0.102 (2) | 0.0582 (15) | 0.0492 (14) | 0.0068 (14) | −0.0162 (14) | −0.0025 (12) |
| C12 | 0.0380 (13) | 0.0445 (13) | 0.0511 (16) | −0.0067 (10) | −0.0112 (12) | −0.0100 (11) |
| N13 | 0.0452 (12) | 0.0592 (13) | 0.0454 (13) | −0.0056 (10) | −0.0132 (10) | −0.0132 (10) |
| C14 | 0.0424 (13) | 0.0546 (15) | 0.0434 (14) | −0.0074 (11) | −0.0121 (11) | −0.0085 (12) |
| N15 | 0.0599 (15) | 0.0770 (17) | 0.0645 (16) | 0.0124 (13) | −0.0240 (13) | −0.0296 (14) |
Geometric parameters (Å, º) top
| Zn1—N1i | 2.0771 (19) | C34—H34 | 0.9300 |
| Zn1—N1 | 2.0772 (19) | C35—C36 | 1.375 (4) |
| Zn1—N51 | 2.1800 (19) | C35—H35 | 0.9300 |
| Zn1—N51i | 2.1800 (19) | C36—H36 | 0.9300 |
| Zn1—N13i | 2.223 (2) | N41—H41A | 0.9395 |
| Zn1—N13 | 2.223 (2) | N41—H41B | 0.9371 |
| N1—C5 | 1.318 (3) | N51—C56 | 1.332 (3) |
| N1—N2 | 1.369 (3) | N51—C52 | 1.349 (3) |
| N2—C3 | 1.319 (3) | C52—C53 | 1.389 (3) |
| C3—N4 | 1.366 (3) | C53—C54 | 1.385 (4) |
| C3—C32 | 1.472 (3) | C53—H53 | 0.9300 |
| N4—C5 | 1.356 (3) | C54—C55 | 1.371 (4) |
| N4—N41 | 1.423 (3) | C54—H54 | 0.9300 |
| C5—C52 | 1.469 (3) | C55—C56 | 1.384 (4) |
| N31—C32 | 1.340 (3) | C55—H55 | 0.9300 |
| N31—C36 | 1.340 (3) | C56—H56 | 0.9300 |
| C32—C33 | 1.388 (3) | N11—C12 | 1.313 (4) |
| C33—C34 | 1.377 (4) | N11—C14 | 1.325 (4) |
| C33—H33 | 0.9300 | C12—N13 | 1.139 (3) |
| C34—C35 | 1.378 (4) | C14—N15 | 1.120 (3) |
| | | |
| N1i—Zn1—N1 | 180.00 (12) | C33—C34—C35 | 118.6 (3) |
| N1i—Zn1—N51 | 103.20 (7) | C33—C34—H34 | 120.7 |
| N1—Zn1—N51 | 76.80 (7) | C35—C34—H34 | 120.7 |
| N1i—Zn1—N51i | 76.80 (7) | C36—C35—C34 | 119.1 (3) |
| N1—Zn1—N51i | 103.20 (7) | C36—C35—H35 | 120.5 |
| N51—Zn1—N51i | 180.0 | C34—C35—H35 | 120.5 |
| N1i—Zn1—N13i | 91.26 (8) | N31—C36—C35 | 123.7 (2) |
| N1—Zn1—N13i | 88.74 (8) | N31—C36—H36 | 118.1 |
| N51—Zn1—N13i | 90.42 (7) | C35—C36—H36 | 118.1 |
| N51i—Zn1—N13i | 89.58 (7) | N4—N41—H41A | 104.8 |
| N1i—Zn1—N13 | 88.74 (8) | N4—N41—H41B | 103.0 |
| N1—Zn1—N13 | 91.26 (8) | H41A—N41—H41B | 105.5 |
| N51—Zn1—N13 | 89.58 (7) | C56—N51—C52 | 118.5 (2) |
| N51i—Zn1—N13 | 90.42 (7) | C56—N51—Zn1 | 126.87 (17) |
| N13i—Zn1—N13 | 180.0 | C52—N51—Zn1 | 114.59 (15) |
| C5—N1—N2 | 109.48 (19) | N51—C52—C53 | 122.1 (2) |
| C5—N1—Zn1 | 114.61 (16) | N51—C52—C5 | 112.19 (19) |
| N2—N1—Zn1 | 135.19 (15) | C53—C52—C5 | 125.7 (2) |
| C3—N2—N1 | 106.26 (18) | C54—C53—C52 | 118.3 (2) |
| N2—C3—N4 | 109.8 (2) | C54—C53—H53 | 120.9 |
| N2—C3—C32 | 122.8 (2) | C52—C53—H53 | 120.9 |
| N4—C3—C32 | 127.4 (2) | C55—C54—C53 | 119.7 (2) |
| C5—N4—C3 | 106.25 (18) | C55—C54—H54 | 120.2 |
| C5—N4—N41 | 124.68 (19) | C53—C54—H54 | 120.2 |
| C3—N4—N41 | 129.1 (2) | C54—C55—C56 | 118.8 (2) |
| N1—C5—N4 | 108.2 (2) | C54—C55—H55 | 120.6 |
| N1—C5—C52 | 120.3 (2) | C56—C55—H55 | 120.6 |
| N4—C5—C52 | 131.5 (2) | N51—C56—C55 | 122.6 (2) |
| C32—N31—C36 | 116.3 (2) | N51—C56—H56 | 118.7 |
| N31—C32—C33 | 123.8 (2) | C55—C56—H56 | 118.7 |
| N31—C32—C3 | 117.5 (2) | C12—N11—C14 | 120.5 (3) |
| C33—C32—C3 | 118.7 (2) | N13—C12—N11 | 168.9 (3) |
| C34—C33—C32 | 118.4 (2) | C12—N13—Zn1 | 165.0 (2) |
| C34—C33—H33 | 120.8 | N15—C14—N11 | 169.1 (3) |
| C32—C33—H33 | 120.8 | | |
| | | |
| C5—N1—N2—C3 | 0.6 (2) | C3—C32—C33—C34 | −177.6 (2) |
| Zn1—N1—N2—C3 | −168.76 (16) | C32—C33—C34—C35 | −0.3 (4) |
| N1—N2—C3—N4 | −0.2 (2) | C33—C34—C35—C36 | −0.2 (4) |
| N1—N2—C3—C32 | 178.66 (19) | C32—N31—C36—C35 | 0.3 (4) |
| N2—C3—N4—C5 | −0.3 (2) | C34—C35—C36—N31 | 0.3 (4) |
| C32—C3—N4—C5 | −179.1 (2) | C56—N51—C52—C53 | 2.4 (3) |
| N2—C3—N4—N41 | 178.0 (2) | Zn1—N51—C52—C53 | −175.45 (16) |
| C32—C3—N4—N41 | −0.7 (4) | C56—N51—C52—C5 | −174.99 (19) |
| N2—N1—C5—N4 | −0.8 (2) | Zn1—N51—C52—C5 | 7.2 (2) |
| Zn1—N1—C5—N4 | 170.96 (13) | N1—C5—C52—N51 | 2.5 (3) |
| N2—N1—C5—C52 | 176.91 (18) | N4—C5—C52—N51 | 179.6 (2) |
| Zn1—N1—C5—C52 | −11.3 (3) | N1—C5—C52—C53 | −174.8 (2) |
| C3—N4—C5—N1 | 0.7 (2) | N4—C5—C52—C53 | 2.4 (4) |
| N41—N4—C5—N1 | −177.74 (19) | N51—C52—C53—C54 | −1.6 (3) |
| C3—N4—C5—C52 | −176.7 (2) | C5—C52—C53—C54 | 175.4 (2) |
| N41—N4—C5—C52 | 4.9 (4) | C52—C53—C54—C55 | −0.6 (4) |
| C36—N31—C32—C33 | −0.9 (3) | C53—C54—C55—C56 | 2.1 (4) |
| C36—N31—C32—C3 | 177.7 (2) | C52—N51—C56—C55 | −0.8 (3) |
| N2—C3—C32—N31 | 173.7 (2) | Zn1—N51—C56—C55 | 176.68 (17) |
| N4—C3—C32—N31 | −7.7 (3) | C54—C55—C56—N51 | −1.4 (4) |
| N2—C3—C32—C33 | −7.7 (3) | C14—N11—C12—N13 | 169.7 (13) |
| N4—C3—C32—C33 | 171.0 (2) | N11—C12—N13—Zn1 | −69.3 (18) |
| N31—C32—C33—C34 | 0.9 (3) | C12—N11—C14—N15 | −169.9 (15) |
| Symmetry code: (i) −x+1, −y+1, −z+1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N41—H41A···N31 | 0.94 | 2.12 | 2.896 (2) | 139 |
| N41—H41B···N15ii | 0.94 | 2.27 | 3.101 (3) | 148 |
| C34—H34···N15iii | 0.93 | 2.56 | 3.357 (4) | 144 |
| Symmetry codes: (ii) x+1, y, z−1; (iii) −x, −y+2, −z+1. |
Bis[4-amino-3,5-bis(pyridin-2-yl)-1,2,4-triazole-
κ2N1,
N5]bis(dicyanamido-
κN1)cadmium(II) (II)
top
Crystal data top
| [Cd(C12H10N6)2(C2N3)2] | Z = 1 |
| Mr = 721.03 | F(000) = 362 |
| Triclinic, P1 | Dx = 1.645 Mg m−3 |
| a = 8.7209 (4) Å | Mo Kα radiation, λ = 0.71073 Å |
| b = 8.7949 (4) Å | Cell parameters from 7093 reflections |
| c = 10.3490 (4) Å | θ = 2.0–30.5° |
| α = 91.966 (2)° | µ = 0.81 mm−1 |
| β = 94.945 (2)° | T = 296 K |
| γ = 112.682 (2)° | Block, colourless |
| V = 727.67 (6) Å3 | 0.18 × 0.16 × 0.12 mm |
Data collection top
Bruker APEXII CCD
diffractometer | 4392 independent reflections |
| Radiation source: fine-focus sealed tube | 4028 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.026 |
| Detector resolution: 9.091 pixels mm-1 | θmax = 30.5°, θmin = 2.5° |
| φ & ω scans | h = −12→8 |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | k = −12→12 |
| Tmin = 0.835, Tmax = 0.908 | l = −14→14 |
| 16382 measured reflections | |
Refinement top
| Refinement on F2 | 0 restraints |
| Least-squares matrix: full | Hydrogen site location: mixed |
| R[F2 > 2σ(F2)] = 0.030 | H-atom parameters constrained |
| wR(F2) = 0.068 | w = 1/[σ2(Fo2) + (0.0298P)2 + 0.1878P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.10 | (Δ/σ)max < 0.001 |
| 4392 reflections | Δρmax = 0.74 e Å−3 |
| 214 parameters | Δρmin = −0.26 e Å−3 |
Special details top
Geometry. All esds (except the esd in the dihedral angle between two l.s. planes)
are estimated using the full covariance matrix. The cell esds are taken
into account individually in the estimation of esds in distances, angles
and torsion angles; correlations between esds in cell parameters are only
used when they are defined by crystal symmetry. An approximate (isotropic)
treatment of cell esds is used for estimating esds involving l.s. planes. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Cd1 | 0.5000 | 0.5000 | 0.5000 | 0.03552 (7) | |
| N1 | 0.42177 (18) | 0.49212 (16) | 0.28558 (13) | 0.0329 (3) | |
| N2 | 0.35782 (18) | 0.58440 (16) | 0.21291 (13) | 0.0335 (3) | |
| C3 | 0.30610 (19) | 0.50560 (18) | 0.09715 (15) | 0.0288 (3) | |
| N4 | 0.33633 (16) | 0.36337 (15) | 0.09455 (12) | 0.0272 (2) | |
| C5 | 0.41039 (19) | 0.36019 (18) | 0.21448 (15) | 0.0281 (3) | |
| N31 | 0.19861 (18) | 0.48614 (18) | −0.12728 (14) | 0.0363 (3) | |
| C32 | 0.22708 (19) | 0.56498 (19) | −0.00901 (15) | 0.0297 (3) | |
| C33 | 0.1834 (2) | 0.6981 (2) | 0.01743 (17) | 0.0357 (3) | |
| H33 | 0.2069 | 0.7508 | 0.1005 | 0.043* | |
| C34 | 0.1048 (2) | 0.7505 (2) | −0.08193 (19) | 0.0404 (4) | |
| H34 | 0.0753 | 0.8401 | −0.0673 | 0.049* | |
| C35 | 0.0705 (2) | 0.6683 (2) | −0.20334 (19) | 0.0417 (4) | |
| H35 | 0.0152 | 0.6996 | −0.2716 | 0.050* | |
| C36 | 0.1201 (2) | 0.5384 (2) | −0.22148 (18) | 0.0424 (4) | |
| H36 | 0.0977 | 0.4842 | −0.3039 | 0.051* | |
| N41 | 0.29499 (19) | 0.23950 (18) | −0.00863 (14) | 0.0369 (3) | |
| H41A | 0.2625 | 0.2834 | −0.0753 | 0.044* | |
| H41B | 0.2124 | 0.1495 | 0.0142 | 0.044* | |
| N51 | 0.54952 (18) | 0.28263 (17) | 0.38956 (14) | 0.0340 (3) | |
| C52 | 0.48039 (19) | 0.24287 (19) | 0.26500 (16) | 0.0299 (3) | |
| C53 | 0.4835 (2) | 0.1090 (2) | 0.19269 (18) | 0.0374 (4) | |
| H53 | 0.4335 | 0.0826 | 0.1074 | 0.045* | |
| C54 | 0.5630 (2) | 0.0149 (2) | 0.2505 (2) | 0.0451 (4) | |
| H54 | 0.5659 | −0.0765 | 0.2045 | 0.054* | |
| C55 | 0.6371 (2) | 0.0579 (2) | 0.3759 (2) | 0.0463 (4) | |
| H55 | 0.6933 | −0.0021 | 0.4154 | 0.056* | |
| C56 | 0.6270 (2) | 0.1914 (2) | 0.44211 (18) | 0.0422 (4) | |
| H56 | 0.6764 | 0.2194 | 0.5275 | 0.051* | |
| N11 | 1.0291 (2) | 0.8653 (3) | 0.38882 (19) | 0.0593 (5) | |
| C12 | 0.8859 (2) | 0.7785 (2) | 0.42838 (18) | 0.0415 (4) | |
| N13 | 0.7688 (2) | 0.6972 (2) | 0.47342 (19) | 0.0563 (5) | |
| C14 | 1.0396 (2) | 0.9320 (2) | 0.27615 (18) | 0.0364 (4) | |
| N15 | 1.0655 (2) | 0.9913 (2) | 0.17991 (19) | 0.0539 (4) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Cd1 | 0.04493 (11) | 0.03405 (10) | 0.02380 (9) | 0.01100 (7) | 0.00564 (7) | 0.00052 (6) |
| N1 | 0.0429 (8) | 0.0274 (6) | 0.0278 (6) | 0.0134 (6) | 0.0024 (6) | 0.0007 (5) |
| N2 | 0.0434 (8) | 0.0273 (6) | 0.0296 (6) | 0.0138 (6) | 0.0031 (6) | 0.0011 (5) |
| C3 | 0.0303 (7) | 0.0236 (7) | 0.0305 (7) | 0.0077 (6) | 0.0061 (6) | 0.0022 (6) |
| N4 | 0.0293 (6) | 0.0238 (6) | 0.0266 (6) | 0.0083 (5) | 0.0034 (5) | −0.0010 (5) |
| C5 | 0.0295 (7) | 0.0250 (7) | 0.0270 (7) | 0.0073 (6) | 0.0051 (6) | 0.0005 (5) |
| N31 | 0.0423 (8) | 0.0358 (7) | 0.0301 (7) | 0.0141 (6) | 0.0061 (6) | 0.0036 (6) |
| C32 | 0.0276 (7) | 0.0268 (7) | 0.0310 (7) | 0.0061 (6) | 0.0059 (6) | 0.0041 (6) |
| C33 | 0.0369 (8) | 0.0307 (8) | 0.0383 (9) | 0.0117 (7) | 0.0058 (7) | 0.0021 (7) |
| C34 | 0.0371 (9) | 0.0350 (9) | 0.0515 (10) | 0.0154 (7) | 0.0080 (8) | 0.0109 (8) |
| C35 | 0.0366 (9) | 0.0455 (10) | 0.0411 (9) | 0.0132 (8) | 0.0029 (7) | 0.0162 (8) |
| C36 | 0.0478 (10) | 0.0455 (10) | 0.0299 (8) | 0.0138 (8) | 0.0033 (7) | 0.0056 (7) |
| N41 | 0.0470 (8) | 0.0316 (7) | 0.0309 (7) | 0.0158 (6) | −0.0011 (6) | −0.0073 (5) |
| N51 | 0.0385 (7) | 0.0319 (7) | 0.0306 (7) | 0.0119 (6) | 0.0057 (6) | 0.0073 (5) |
| C52 | 0.0282 (7) | 0.0262 (7) | 0.0330 (8) | 0.0071 (6) | 0.0065 (6) | 0.0044 (6) |
| C53 | 0.0372 (8) | 0.0295 (8) | 0.0435 (9) | 0.0113 (7) | 0.0030 (7) | −0.0014 (7) |
| C54 | 0.0455 (10) | 0.0300 (8) | 0.0624 (12) | 0.0166 (8) | 0.0107 (9) | 0.0045 (8) |
| C55 | 0.0464 (10) | 0.0406 (10) | 0.0582 (12) | 0.0216 (8) | 0.0102 (9) | 0.0196 (9) |
| C56 | 0.0460 (10) | 0.0438 (10) | 0.0378 (9) | 0.0176 (8) | 0.0055 (8) | 0.0151 (8) |
| N11 | 0.0406 (9) | 0.0737 (13) | 0.0500 (10) | 0.0072 (9) | 0.0008 (8) | 0.0183 (9) |
| C12 | 0.0435 (10) | 0.0379 (9) | 0.0371 (9) | 0.0101 (8) | 0.0019 (7) | −0.0003 (7) |
| N13 | 0.0519 (10) | 0.0500 (10) | 0.0525 (10) | 0.0025 (8) | 0.0141 (8) | 0.0015 (8) |
| C14 | 0.0302 (8) | 0.0344 (8) | 0.0421 (9) | 0.0101 (7) | 0.0028 (7) | 0.0009 (7) |
| N15 | 0.0544 (10) | 0.0531 (10) | 0.0552 (10) | 0.0203 (8) | 0.0093 (8) | 0.0160 (8) |
Geometric parameters (Å, º) top
| Cd1—N1 | 2.2553 (13) | C34—H34 | 0.9300 |
| Cd1—N1i | 2.2553 (13) | C35—C36 | 1.382 (3) |
| Cd1—N13i | 2.3638 (17) | C35—H35 | 0.9300 |
| Cd1—N13 | 2.3638 (17) | C36—H36 | 0.9300 |
| Cd1—N51i | 2.3915 (14) | N41—H41A | 0.8802 |
| Cd1—N51 | 2.3916 (14) | N41—H41B | 0.8962 |
| N1—C5 | 1.3182 (19) | N51—C56 | 1.334 (2) |
| N1—N2 | 1.362 (2) | N51—C52 | 1.349 (2) |
| N2—C3 | 1.314 (2) | C52—C53 | 1.384 (2) |
| C3—N4 | 1.3741 (19) | C53—C54 | 1.388 (3) |
| C3—C32 | 1.463 (2) | C53—H53 | 0.9300 |
| N4—C5 | 1.355 (2) | C54—C55 | 1.370 (3) |
| N4—N41 | 1.4170 (17) | C54—H54 | 0.9300 |
| C5—C52 | 1.476 (2) | C55—C56 | 1.375 (3) |
| N31—C36 | 1.337 (2) | C55—H55 | 0.9300 |
| N31—C32 | 1.341 (2) | C56—H56 | 0.9300 |
| C32—C33 | 1.390 (2) | N11—C12 | 1.298 (3) |
| C33—C34 | 1.376 (3) | N11—C14 | 1.318 (3) |
| C33—H33 | 0.9300 | C12—N13 | 1.145 (2) |
| C34—C35 | 1.377 (3) | C14—N15 | 1.140 (2) |
| | | |
| N1—Cd1—N1i | 180.00 (8) | C33—C34—C35 | 119.02 (17) |
| N1—Cd1—N13i | 89.71 (6) | C33—C34—H34 | 120.5 |
| N1i—Cd1—N13i | 90.28 (6) | C35—C34—H34 | 120.5 |
| N1—Cd1—N13 | 90.29 (6) | C34—C35—C36 | 118.55 (17) |
| N1i—Cd1—N13 | 89.72 (6) | C34—C35—H35 | 120.7 |
| N13i—Cd1—N13 | 180.0 | C36—C35—H35 | 120.7 |
| N1—Cd1—N51i | 108.93 (5) | N31—C36—C35 | 123.75 (17) |
| N1i—Cd1—N51i | 71.07 (5) | N31—C36—H36 | 118.1 |
| N13i—Cd1—N51i | 92.62 (6) | C35—C36—H36 | 118.1 |
| N13—Cd1—N51i | 87.38 (6) | N4—N41—H41A | 103.6 |
| N1—Cd1—N51 | 71.07 (5) | N4—N41—H41B | 107.3 |
| N1i—Cd1—N51 | 108.93 (5) | H41A—N41—H41B | 112.6 |
| N13i—Cd1—N51 | 87.38 (6) | C56—N51—C52 | 118.14 (15) |
| N13—Cd1—N51 | 92.62 (6) | C56—N51—Cd1 | 126.23 (12) |
| N51i—Cd1—N51 | 180.00 (6) | C52—N51—Cd1 | 115.43 (11) |
| C5—N1—N2 | 109.62 (13) | N51—C52—C53 | 122.14 (15) |
| C5—N1—Cd1 | 116.37 (10) | N51—C52—C5 | 113.16 (14) |
| N2—N1—Cd1 | 133.17 (10) | C53—C52—C5 | 124.64 (15) |
| C3—N2—N1 | 106.59 (13) | C52—C53—C54 | 118.43 (17) |
| N2—C3—N4 | 109.64 (14) | C52—C53—H53 | 120.8 |
| N2—C3—C32 | 123.20 (14) | C54—C53—H53 | 120.8 |
| N4—C3—C32 | 127.15 (14) | C55—C54—C53 | 119.45 (18) |
| C5—N4—C3 | 105.99 (12) | C55—C54—H54 | 120.3 |
| C5—N4—N41 | 125.56 (13) | C53—C54—H54 | 120.3 |
| C3—N4—N41 | 128.42 (13) | C54—C55—C56 | 118.76 (17) |
| N1—C5—N4 | 108.16 (13) | C54—C55—H55 | 120.6 |
| N1—C5—C52 | 121.74 (14) | C56—C55—H55 | 120.6 |
| N4—C5—C52 | 130.00 (14) | N51—C56—C55 | 123.05 (18) |
| C36—N31—C32 | 116.81 (15) | N51—C56—H56 | 118.5 |
| N31—C32—C33 | 123.22 (15) | C55—C56—H56 | 118.5 |
| N31—C32—C3 | 117.94 (14) | C12—N11—C14 | 121.65 (18) |
| C33—C32—C3 | 118.83 (14) | N13—C12—N11 | 172.8 (2) |
| C34—C33—C32 | 118.61 (16) | C12—N13—Cd1 | 162.67 (17) |
| C34—C33—H33 | 120.7 | N15—C14—N11 | 172.9 (2) |
| C32—C33—H33 | 120.7 | | |
| | | |
| C5—N1—N2—C3 | 0.58 (18) | N31—C32—C33—C34 | 1.0 (2) |
| Cd1—N1—N2—C3 | −168.22 (11) | C3—C32—C33—C34 | −178.17 (15) |
| N1—N2—C3—N4 | −0.06 (17) | C32—C33—C34—C35 | 0.7 (2) |
| N1—N2—C3—C32 | 179.23 (14) | C33—C34—C35—C36 | −1.5 (3) |
| N2—C3—N4—C5 | −0.47 (17) | C32—N31—C36—C35 | 1.0 (3) |
| C32—C3—N4—C5 | −179.72 (14) | C34—C35—C36—N31 | 0.7 (3) |
| N2—C3—N4—N41 | 177.40 (14) | C56—N51—C52—C53 | 1.9 (2) |
| C32—C3—N4—N41 | −1.9 (2) | Cd1—N51—C52—C53 | −173.28 (12) |
| N2—N1—C5—N4 | −0.88 (17) | C56—N51—C52—C5 | −175.39 (14) |
| Cd1—N1—C5—N4 | 170.02 (9) | Cd1—N51—C52—C5 | 9.43 (16) |
| N2—N1—C5—C52 | 175.78 (13) | N1—C5—C52—N51 | 2.1 (2) |
| Cd1—N1—C5—C52 | −13.31 (19) | N4—C5—C52—N51 | 177.98 (15) |
| C3—N4—C5—N1 | 0.82 (16) | N1—C5—C52—C53 | −175.09 (15) |
| N41—N4—C5—N1 | −177.13 (14) | N4—C5—C52—C53 | 0.8 (3) |
| C3—N4—C5—C52 | −175.47 (15) | N51—C52—C53—C54 | −1.1 (2) |
| N41—N4—C5—C52 | 6.6 (2) | C5—C52—C53—C54 | 175.92 (15) |
| C36—N31—C32—C33 | −1.8 (2) | C52—C53—C54—C55 | −0.7 (3) |
| C36—N31—C32—C3 | 177.36 (14) | C53—C54—C55—C56 | 1.6 (3) |
| N2—C3—C32—N31 | 171.72 (15) | C52—N51—C56—C55 | −1.0 (3) |
| N4—C3—C32—N31 | −9.1 (2) | Cd1—N51—C56—C55 | 173.60 (13) |
| N2—C3—C32—C33 | −9.1 (2) | C54—C55—C56—N51 | −0.7 (3) |
| N4—C3—C32—C33 | 170.10 (15) | | |
| Symmetry code: (i) −x+1, −y+1, −z+1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N41—H41A···N31 | 0.88 | 2.14 | 2.886 (2) | 143 |
| N41—H41B···N15ii | 0.90 | 2.38 | 3.190 (2) | 150 |
| C34—H34···N15iii | 0.93 | 2.52 | 3.300 (3) | 142 |
| Symmetry codes: (ii) x−1, y−1, z; (iii) −x+1, −y+2, −z. |
Bis[4-amino-3,5-bis(pyridin-2-yl)-1,2,4-triazole-
κ2N1,
N5]bis(tricyanomethanido-
κN1)cadmium(II) (III)
top
Crystal data top
| [Cd(C12H10N6)2(C4N3)2] | Z = 1 |
| Mr = 769.07 | F(000) = 386 |
| Triclinic, P1 | Dx = 1.525 Mg m−3 |
| a = 9.271 (5) Å | Mo Kα radiation, λ = 0.71073 Å |
| b = 9.571 (5) Å | Cell parameters from 5042 reflections |
| c = 10.156 (5) Å | θ = 2.1–30.7° |
| α = 87.833 (5)° | µ = 0.71 mm−1 |
| β = 68.383 (5)° | T = 296 K |
| γ = 88.803 (5)° | Plate, colourless |
| V = 837.2 (8) Å3 | 0.42 × 0.34 × 0.16 mm |
Data collection top
Bruker APEXII CCD
diffractometer | 5040 independent reflections |
| Radiation source: fine-focus sealed tube | 4682 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.027 |
| Detector resolution: 9.091 pixels mm-1 | θmax = 30.7°, θmin = 2.6° |
| φ & ω scans | h = −13→13 |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | k = −13→13 |
| Tmin = 0.789, Tmax = 0.893 | l = −14→14 |
| 17569 measured reflections | |
Refinement top
| Refinement on F2 | 0 restraints |
| Least-squares matrix: full | Hydrogen site location: mixed |
| R[F2 > 2σ(F2)] = 0.026 | H-atom parameters constrained |
| wR(F2) = 0.064 | w = 1/[σ2(Fo2) + (0.0292P)2 + 0.1701P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.06 | (Δ/σ)max < 0.001 |
| 5040 reflections | Δρmax = 0.35 e Å−3 |
| 232 parameters | Δρmin = −0.27 e Å−3 |
Special details top
Geometry. All esds (except the esd in the dihedral angle between two l.s. planes)
are estimated using the full covariance matrix. The cell esds are taken
into account individually in the estimation of esds in distances, angles
and torsion angles; correlations between esds in cell parameters are only
used when they are defined by crystal symmetry. An approximate (isotropic)
treatment of cell esds is used for estimating esds involving l.s. planes. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Cd1 | 0.5000 | 0.5000 | 0.5000 | 0.03593 (5) | |
| N1 | 0.40227 (14) | 0.28256 (12) | 0.58927 (13) | 0.0370 (3) | |
| N2 | 0.25756 (15) | 0.22441 (13) | 0.64461 (14) | 0.0397 (3) | |
| C3 | 0.27462 (16) | 0.10046 (14) | 0.69908 (14) | 0.0329 (3) | |
| N4 | 0.42789 (13) | 0.07716 (11) | 0.67750 (12) | 0.0311 (2) | |
| C5 | 0.50407 (16) | 0.19435 (13) | 0.60928 (13) | 0.0309 (3) | |
| N31 | 0.18341 (16) | −0.13123 (13) | 0.78425 (14) | 0.0394 (3) | |
| C32 | 0.14578 (17) | 0.00371 (14) | 0.77116 (14) | 0.0341 (3) | |
| C33 | −0.00545 (19) | 0.05402 (17) | 0.81991 (16) | 0.0418 (3) | |
| H33 | −0.0266 | 0.1483 | 0.8092 | 0.050* | |
| C34 | −0.1244 (2) | −0.0401 (2) | 0.88523 (18) | 0.0494 (4) | |
| H34 | −0.2273 | −0.0102 | 0.9189 | 0.059* | |
| C35 | −0.0876 (2) | −0.17915 (19) | 0.89937 (19) | 0.0512 (4) | |
| H35 | −0.1652 | −0.2444 | 0.9423 | 0.061* | |
| C36 | 0.0661 (2) | −0.21934 (17) | 0.84879 (18) | 0.0462 (4) | |
| H36 | 0.0898 | −0.3128 | 0.8600 | 0.055* | |
| N41 | 0.49708 (15) | −0.03943 (12) | 0.72259 (15) | 0.0418 (3) | |
| H41A | 0.4338 | −0.1068 | 0.7292 | 0.050* | |
| H41B | 0.4943 | −0.0171 | 0.8089 | 0.050* | |
| N51 | 0.70511 (14) | 0.35659 (12) | 0.50000 (12) | 0.0345 (2) | |
| C52 | 0.67068 (16) | 0.22782 (13) | 0.56142 (13) | 0.0315 (3) | |
| C53 | 0.78406 (18) | 0.13841 (16) | 0.57522 (17) | 0.0421 (3) | |
| H53 | 0.7584 | 0.0506 | 0.6192 | 0.051* | |
| C54 | 0.93701 (19) | 0.18322 (19) | 0.52168 (18) | 0.0477 (4) | |
| H54 | 1.0152 | 0.1257 | 0.5306 | 0.057* | |
| C55 | 0.97249 (19) | 0.31293 (19) | 0.45552 (18) | 0.0468 (4) | |
| H55 | 1.0747 | 0.3433 | 0.4172 | 0.056* | |
| C56 | 0.85348 (19) | 0.39696 (17) | 0.44715 (17) | 0.0430 (3) | |
| H56 | 0.8771 | 0.4850 | 0.4032 | 0.052* | |
| C11 | 0.63172 (19) | 0.36077 (16) | 0.01038 (15) | 0.0408 (3) | |
| C12 | 0.5809 (2) | 0.40430 (16) | 0.14978 (16) | 0.0426 (3) | |
| N13 | 0.5399 (2) | 0.43872 (18) | 0.26415 (16) | 0.0622 (4) | |
| C14 | 0.5511 (2) | 0.25687 (18) | −0.02763 (17) | 0.0472 (4) | |
| N15 | 0.4882 (2) | 0.1709 (2) | −0.0598 (2) | 0.0783 (6) | |
| C16 | 0.7657 (3) | 0.4184 (2) | −0.0923 (2) | 0.0644 (5) | |
| N17 | 0.8755 (3) | 0.4637 (3) | −0.1762 (3) | 0.1235 (11) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Cd1 | 0.04851 (10) | 0.02405 (7) | 0.03407 (8) | 0.00161 (5) | −0.01430 (6) | 0.00277 (5) |
| N1 | 0.0365 (6) | 0.0297 (6) | 0.0452 (7) | −0.0007 (4) | −0.0160 (5) | 0.0068 (5) |
| N2 | 0.0369 (6) | 0.0337 (6) | 0.0489 (7) | −0.0009 (5) | −0.0169 (5) | 0.0071 (5) |
| C3 | 0.0363 (7) | 0.0304 (6) | 0.0335 (6) | −0.0011 (5) | −0.0146 (5) | 0.0005 (5) |
| N4 | 0.0367 (6) | 0.0244 (5) | 0.0340 (5) | 0.0002 (4) | −0.0156 (5) | 0.0024 (4) |
| C5 | 0.0379 (7) | 0.0243 (5) | 0.0314 (6) | −0.0005 (5) | −0.0141 (5) | 0.0017 (4) |
| N31 | 0.0465 (7) | 0.0311 (6) | 0.0426 (6) | −0.0060 (5) | −0.0186 (6) | 0.0000 (5) |
| C32 | 0.0406 (7) | 0.0333 (6) | 0.0309 (6) | −0.0051 (5) | −0.0161 (5) | 0.0002 (5) |
| C33 | 0.0435 (8) | 0.0412 (8) | 0.0407 (7) | −0.0009 (6) | −0.0155 (6) | −0.0006 (6) |
| C34 | 0.0393 (8) | 0.0595 (10) | 0.0469 (9) | −0.0059 (7) | −0.0128 (7) | −0.0021 (7) |
| C35 | 0.0509 (10) | 0.0517 (9) | 0.0491 (9) | −0.0197 (8) | −0.0158 (8) | 0.0032 (7) |
| C36 | 0.0563 (10) | 0.0350 (7) | 0.0494 (9) | −0.0117 (7) | −0.0216 (8) | 0.0022 (6) |
| N41 | 0.0471 (7) | 0.0269 (6) | 0.0553 (8) | −0.0006 (5) | −0.0244 (6) | 0.0106 (5) |
| N51 | 0.0390 (6) | 0.0283 (5) | 0.0346 (6) | −0.0027 (4) | −0.0115 (5) | 0.0000 (4) |
| C52 | 0.0363 (7) | 0.0288 (6) | 0.0289 (6) | −0.0005 (5) | −0.0115 (5) | −0.0010 (4) |
| C53 | 0.0423 (8) | 0.0374 (7) | 0.0469 (8) | 0.0028 (6) | −0.0175 (7) | 0.0057 (6) |
| C54 | 0.0386 (8) | 0.0539 (9) | 0.0523 (9) | 0.0059 (7) | −0.0193 (7) | 0.0001 (7) |
| C55 | 0.0359 (8) | 0.0561 (10) | 0.0470 (8) | −0.0075 (7) | −0.0129 (7) | −0.0038 (7) |
| C56 | 0.0446 (8) | 0.0378 (7) | 0.0431 (8) | −0.0084 (6) | −0.0116 (7) | 0.0002 (6) |
| C11 | 0.0489 (8) | 0.0375 (7) | 0.0334 (7) | −0.0022 (6) | −0.0118 (6) | −0.0010 (5) |
| C12 | 0.0537 (9) | 0.0364 (7) | 0.0391 (8) | 0.0011 (6) | −0.0188 (7) | 0.0003 (6) |
| N13 | 0.0859 (12) | 0.0604 (10) | 0.0409 (8) | −0.0005 (9) | −0.0234 (8) | −0.0084 (7) |
| C14 | 0.0524 (9) | 0.0510 (9) | 0.0350 (7) | −0.0049 (7) | −0.0122 (7) | −0.0018 (6) |
| N15 | 0.0819 (13) | 0.0873 (14) | 0.0624 (11) | −0.0315 (11) | −0.0198 (10) | −0.0154 (10) |
| C16 | 0.0743 (13) | 0.0511 (10) | 0.0521 (10) | −0.0179 (9) | −0.0037 (9) | −0.0044 (8) |
| N17 | 0.118 (2) | 0.1047 (19) | 0.0944 (17) | −0.0563 (16) | 0.0275 (15) | −0.0083 (14) |
Geometric parameters (Å, º) top
| Cd1—N1i | 2.3032 (15) | C35—C36 | 1.376 (3) |
| Cd1—N1 | 2.3032 (15) | C35—H35 | 0.9300 |
| Cd1—N51 | 2.3219 (15) | C36—H36 | 0.9300 |
| Cd1—N51i | 2.3219 (15) | N41—H41A | 0.8649 |
| Cd1—N13i | 2.3797 (19) | N41—H41B | 0.9012 |
| Cd1—N13 | 2.3797 (19) | N51—C56 | 1.339 (2) |
| N1—C5 | 1.3191 (18) | N51—C52 | 1.3496 (18) |
| N1—N2 | 1.3702 (19) | C52—C53 | 1.385 (2) |
| N2—C3 | 1.3211 (18) | C53—C54 | 1.389 (2) |
| C3—N4 | 1.3703 (19) | C53—H53 | 0.9300 |
| C3—C32 | 1.473 (2) | C54—C55 | 1.376 (3) |
| N4—C5 | 1.3604 (17) | C54—H54 | 0.9300 |
| N4—N41 | 1.4193 (16) | C55—C56 | 1.378 (2) |
| C5—C52 | 1.477 (2) | C55—H55 | 0.9300 |
| N31—C36 | 1.340 (2) | C56—H56 | 0.9300 |
| N31—C32 | 1.3453 (19) | C11—C12 | 1.395 (2) |
| C32—C33 | 1.386 (2) | C11—C16 | 1.402 (2) |
| C33—C34 | 1.388 (2) | C11—C14 | 1.403 (2) |
| C33—H33 | 0.9300 | C12—N13 | 1.140 (2) |
| C34—C35 | 1.382 (3) | C14—N15 | 1.142 (2) |
| C34—H34 | 0.9300 | C16—N17 | 1.141 (3) |
| | | |
| N1i—Cd1—N1 | 180.00 (6) | C35—C34—H34 | 120.5 |
| N1i—Cd1—N51 | 108.14 (5) | C33—C34—H34 | 120.5 |
| N1—Cd1—N51 | 71.86 (5) | C36—C35—C34 | 118.82 (15) |
| N1i—Cd1—N51i | 71.86 (5) | C36—C35—H35 | 120.6 |
| N1—Cd1—N51i | 108.14 (5) | C34—C35—H35 | 120.6 |
| N51—Cd1—N51i | 180.0 | N31—C36—C35 | 123.65 (16) |
| N1i—Cd1—N13i | 92.09 (6) | N31—C36—H36 | 118.2 |
| N1—Cd1—N13i | 87.91 (6) | C35—C36—H36 | 118.2 |
| N51—Cd1—N13i | 89.17 (5) | N4—N41—H41A | 103.3 |
| N51i—Cd1—N13i | 90.83 (5) | N4—N41—H41B | 104.4 |
| N1i—Cd1—N13 | 87.91 (6) | H41A—N41—H41B | 110.8 |
| N1—Cd1—N13 | 92.09 (6) | C56—N51—C52 | 118.96 (13) |
| N51—Cd1—N13 | 90.83 (5) | C56—N51—Cd1 | 123.45 (10) |
| N51i—Cd1—N13 | 89.17 (5) | C52—N51—Cd1 | 117.55 (10) |
| N13i—Cd1—N13 | 180.0 | N51—C52—C53 | 121.74 (13) |
| C5—N1—N2 | 109.27 (12) | N51—C52—C5 | 113.38 (11) |
| C5—N1—Cd1 | 114.58 (10) | C53—C52—C5 | 124.88 (13) |
| N2—N1—Cd1 | 135.56 (9) | C52—C53—C54 | 118.35 (15) |
| C3—N2—N1 | 106.46 (12) | C52—C53—H53 | 120.8 |
| N2—C3—N4 | 109.83 (12) | C54—C53—H53 | 120.8 |
| N2—C3—C32 | 123.99 (13) | C55—C54—C53 | 119.83 (15) |
| N4—C3—C32 | 126.18 (12) | C55—C54—H54 | 120.1 |
| C5—N4—C3 | 105.95 (11) | C53—C54—H54 | 120.1 |
| C5—N4—N41 | 125.21 (12) | C54—C55—C56 | 118.62 (15) |
| C3—N4—N41 | 128.75 (12) | C54—C55—H55 | 120.7 |
| N1—C5—N4 | 108.48 (12) | C56—C55—H55 | 120.7 |
| N1—C5—C52 | 122.00 (12) | N51—C56—C55 | 122.45 (15) |
| N4—C5—C52 | 129.52 (12) | N51—C56—H56 | 118.8 |
| C36—N31—C32 | 116.86 (14) | C55—C56—H56 | 118.8 |
| N31—C32—C33 | 123.55 (14) | C12—C11—C16 | 120.23 (16) |
| N31—C32—C3 | 116.92 (13) | C12—C11—C14 | 120.31 (15) |
| C33—C32—C3 | 119.52 (13) | C16—C11—C14 | 119.45 (15) |
| C32—C33—C34 | 118.16 (15) | N13—C12—C11 | 179.34 (19) |
| C32—C33—H33 | 120.9 | C12—N13—Cd1 | 169.75 (17) |
| C34—C33—H33 | 120.9 | N15—C14—C11 | 178.6 (2) |
| C35—C34—C33 | 118.94 (17) | N17—C16—C11 | 179.2 (3) |
| | | |
| C5—N1—N2—C3 | 0.54 (16) | N31—C32—C33—C34 | −0.3 (2) |
| Cd1—N1—N2—C3 | −169.83 (11) | C3—C32—C33—C34 | 178.93 (14) |
| N1—N2—C3—N4 | −1.04 (16) | C32—C33—C34—C35 | 0.2 (2) |
| N1—N2—C3—C32 | 179.22 (13) | C33—C34—C35—C36 | 0.3 (3) |
| N2—C3—N4—C5 | 1.15 (15) | C32—N31—C36—C35 | 0.9 (2) |
| C32—C3—N4—C5 | −179.12 (13) | C34—C35—C36—N31 | −0.9 (3) |
| N2—C3—N4—N41 | 177.58 (13) | C56—N51—C52—C53 | 2.1 (2) |
| C32—C3—N4—N41 | −2.7 (2) | Cd1—N51—C52—C53 | −175.53 (11) |
| N2—N1—C5—N4 | 0.17 (16) | C56—N51—C52—C5 | −177.38 (13) |
| Cd1—N1—C5—N4 | 172.77 (8) | Cd1—N51—C52—C5 | 4.96 (15) |
| N2—N1—C5—C52 | −179.76 (12) | N1—C5—C52—N51 | 1.57 (19) |
| Cd1—N1—C5—C52 | −7.16 (17) | N4—C5—C52—N51 | −178.35 (13) |
| C3—N4—C5—N1 | −0.79 (15) | N1—C5—C52—C53 | −177.92 (14) |
| N41—N4—C5—N1 | −177.38 (12) | N4—C5—C52—C53 | 2.2 (2) |
| C3—N4—C5—C52 | 179.14 (13) | N51—C52—C53—C54 | −1.1 (2) |
| N41—N4—C5—C52 | 2.5 (2) | C5—C52—C53—C54 | 178.35 (14) |
| C36—N31—C32—C33 | −0.2 (2) | C52—C53—C54—C55 | −0.8 (3) |
| C36—N31—C32—C3 | −179.47 (13) | C53—C54—C55—C56 | 1.5 (3) |
| N2—C3—C32—N31 | 161.22 (14) | C52—N51—C56—C55 | −1.3 (2) |
| N4—C3—C32—N31 | −18.5 (2) | Cd1—N51—C56—C55 | 176.20 (12) |
| N2—C3—C32—C33 | −18.0 (2) | C54—C55—C56—N51 | −0.5 (3) |
| N4—C3—C32—C33 | 162.26 (14) | | |
| Symmetry code: (i) −x+1, −y+1, −z+1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N41—H41A···N31 | 0.86 | 2.19 | 2.889 (3) | 137 |
| N41—H41B···N15ii | 0.90 | 2.27 | 3.023 (3) | 141 |
| C56—H56···N17iii | 0.93 | 2.62 | 3.230 (4) | 123 |
| Symmetry codes: (ii) x, y, z+1; (iii) −x+2, −y+1, −z. |
Selected geometric parameters (Å, °) top| Parameter | | (I) (M = Zn; X = N) | (II) (M = Cd; X = C) | (III) (M = Cd; X = C) |
| M—N1 | | 2.0772 (19) | 2.2553 (13) | 2.3032 (15) |
| M—N13 | | 2.223 (2) | 2.3638 (17) | 2.3797 (19) |
| M—N51 | | 2.1800 (19) | 2.3915 (14) | 2.3219 (15) |
| X11—C12 | | 1.313 (4) | 1.298 (3) | 1.395 (2) |
| X11—C14 | | 1.325 (4) | 1.318 (3) | 1.403 (2) |
| X11—C16 | | | | 1.402 (2) |
| C12—N13 | | 1.139 (3) | 1.145 (2) | 1.140 (2) |
| C14—N15 | | 1.120 (3) | 1.140 (2) | 1.142 (2) |
| C16—N17 | | | | 1.141 (3) |
| | | | |
| N1—M—N13 | | 91.26 (8) | 90.29 (6) | 92.09 (6) |
| N1—M—N51 | | 76.80 (7) | 71.07 (5) | 71.86 (5) |
| N13—M—N51 | | 89.58 (7) | 92.62 (6) | 90.83 (5) |
| C12—X11—C14 | | 120.5 (3) | 121.65 (18) | 120.31 (15) |
| C12—X11—C16 | | | | 120.13 (16) |
| C14—X11—C16 | | | | 119.45 (15) |
| X11—C12—N13 | | 168.9 (3) | 172.8 (2) | 179.34 (19) |
| X11—C14—N15 | | 169.1 (3) | 172.9 (2) | 178.6 (2) |
| X11—C16—N17 | | | | 179.2 (3) |
| | | | |
| N2—C3—C32—N31 | | 173.7 (2) | 171.72 (15) | 161.22 (14) |
| N1—C5—C52—N51 | | 2.5 (3) | 2.1 (2) | 1.57 (19) |
| N2—N1—M—N13 | | -96.37 (16) | -90.8 (2) | -93.03 (14) |
| | | | |
| Pyrazole/N31–C36 | | 8.01 (12) | 9.01 (9) | 18.16 (8) |
| Pyrazole/N51–C56 | | 6.93 (12) | 7.26 (9) | 7.03 (8) |
| N31–C36/N51–C56 | | 5.38 (16) | 3.34 (12) | 16.17 (8) |
Hydrogen bonds and short intermolecular contacts (Å, °) top| Compound | D—H···A | | D—H | H···A | D···A | D—H···A |
| (I) | N41—H41A···N31 | | 0.94 | 2.12 | 2.896 (3) | 139 |
| N41—H41B···N15i | | 0.94 | 2.27 | 3.101 (3) | 140 |
| C34—H34···N15ii | | 0.93 | 2.56 | 3.357 (4) | 144 |
| | | | | | |
| (II) | N41—H41A···N31 | | 0.88 | 2.14 | 2.886 (2) | 143 |
| N41—H41B···N15iii | | 0.90 | 2.38 | 3.190 (2) | 150 |
| C34—H34···N15iv | | 0.93 | 2.52 | 3.300 (3) | 142 |
| | | | | | |
| (III) | N41—H41A···N31 | | 0.86 | 2.19 | 2.889 (3) | 137 |
| N41—H41B···N15v | | 0.90 | 2.27 | 3.023 (2) | 141 |
| C56—H56···N17vi | | 0.93 | 2.62 | 3.230 (4) | 123 |
| Symmetry codes:
(i) x+1, y, z-1;
(ii) -x, -y+2, -z+1;
(iii) x-1, y-1, z;
(iv) -x+1, -y+2, -z;
(v) x, y, z+1;
(vi) -x+2, -y+1, -z. |

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
[email protected] for assistance.
 N and C—H
N and C—H N hydrogen bonds and π–π stacking interactions generates three-dimensional framework structures in (I) and (II), and a sheet structure in (III). The photoluminescence spectra of (I)–(III) indicate that the energies of the π–π* transitions in the coordinated triazole ligand are modified by minor changes of the ligand geometry associated with coordination to the metal centres.
N hydrogen bonds and π–π stacking interactions generates three-dimensional framework structures in (I) and (II), and a sheet structure in (III). The photoluminescence spectra of (I)–(III) indicate that the energies of the π–π* transitions in the coordinated triazole ligand are modified by minor changes of the ligand geometry associated with coordination to the metal centres.
 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
 journal menu
journal menu











