Buy article online - an online subscription or single-article purchase is required to access this article.
A hexagonal modification (
P6
3mc) of (NH
4)
2SiF
6 was grown accidentally. This new modification has the unit-cell
c parameter doubled with respect to a previously reported trigonal form (
P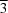 m
m1) of the title compound. The H atoms in the present structure are ordered. Three or even four F atoms are hydrogen-bond acceptors for each H atom. The structure is isostructural with modifications of (NH
4)
2MnF
6, K
2GeF
6 and Rb
2GeF
6.
Supporting information
Key indicators
- Single-crystal X-ray study
- T = 290 K
- Mean
![[sigma]](https://journals.iucr.org/logos/entities/sigma_rmgif.gif) (Si-F) = 0.001 Å
(Si-F) = 0.001 Å
- R factor = 0.018
- wR factor = 0.022
- Data-to-parameter ratio = 11.6
checkCIF results
No syntax errors found
ADDSYM reports no extra symmetry
 Alert Level C:
Alert Level C:
PLAT_420 Alert C D-H Without Acceptor N(2) - H(22N) ?
General Notes
REFLT_03
From the CIF: _diffrn_reflns_theta_max 29.98
From the CIF: _reflns_number_total 361
Count of symmetry unique reflns 191
Completeness (_total/calc) 189.01%
TEST3: Check Friedels for noncentro structure
Estimate of Friedel pairs measured 170
Fraction of Friedel pairs measured 0.890
Are heavy atom types Z>Si present yes
Please check that the estimate of the number of Friedel pairs is
correct. If it is not, please give the correct count in the
_publ_section_exptl_refinement section of the submitted CIF.
0 Alert Level A = Potentially serious problem
0 Alert Level B = Potential problem
1 Alert Level C = Please check
The present compound grew from a solution which was prepared by neutralization
of stoichiometric amounts of (NH4)2CO3 and H2PO3F in the molar
proportion 1:1. Most probably some of the fluorophosphate anions have
decomposed with production of HF which reacted with the glass of the beaker.
In addition to the structure which is reported here, the cubic modification
was also grown, in fact, in larger amount. Its lattice parameters corresponded
to those determined by e.g. Hanic (1966).
No restraints or constraints were applied. The inversion-twinning proportion
determined by refinement turned out to be insignificant.
Cell refinement: KM4B8; data reduction: JANA2000 (Petříček & Dušek, 2000); program(s) used to solve structure: JANA2000; program(s) used to refine structure: JANA2000; molecular graphics: ORTEPIII (Burnett & Johnson, 1996).
Crystal data top
| 2(NH4)·(SiF6) | Dx = 2.047 Mg m−3 |
| Mr = 178.15 | Mo Kα radiation, λ = 0.71069 Å |
| Hexagonal, P63mc | Cell parameters from 68 reflections |
| a = 5.8955 (10) Å | θ = 8.5–19.1° |
| c = 9.599 (1) Å | µ = 0.46 mm−1 |
| V = 288.93 (8) Å3 | T = 290 K |
| Z = 2 | Pyramid, colourless |
| F(000) = 180 | 0.40 × 0.35 × 0.30 mm |
Data collection top
Kuma-4
diffractometer | θmax = 30.0°, θmin = 4.0° |
| w/2θ scans | h = −7→7 |
| 1682 measured reflections | k = 0→7 |
| 361 independent reflections | l = −13→13 |
| 348 reflections with I > 3σ(I) | 3 standard reflections every 200 reflections |
| Rint = 0.020 | intensity decay: 1.5% |
Refinement top
| Refinement on F | All H-atom parameters refined |
| R[F2 > 2σ(F2)] = 0.018 | Weighting scheme based on measured s.u.'s w = 1/(σ2(F) + 0.0001F2) |
| wR(F2) = 0.022 | (Δ/σ)max = 0.0001 |
| S = 1.70 | Δρmax = 0.39 e Å−3 |
| 361 reflections | Δρmin = −0.39 e Å−3 |
| 31 parameters | |
Crystal data top
| 2(NH4)·(SiF6) | Z = 2 |
| Mr = 178.15 | Mo Kα radiation |
| Hexagonal, P63mc | µ = 0.46 mm−1 |
| a = 5.8955 (10) Å | T = 290 K |
| c = 9.599 (1) Å | 0.40 × 0.35 × 0.30 mm |
| V = 288.93 (8) Å3 | |
Data collection top
Kuma-4
diffractometer | Rint = 0.020 |
| 1682 measured reflections | 3 standard reflections every 200 reflections |
| 361 independent reflections | intensity decay: 1.5% |
| 348 reflections with I > 3σ(I) | |
Refinement top
| R[F2 > 2σ(F2)] = 0.018 | 31 parameters |
| wR(F2) = 0.022 | All H-atom parameters refined |
| S = 1.70 | Δρmax = 0.39 e Å−3 |
| 361 reflections | Δρmin = −0.39 e Å−3 |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Si | 0.333333 | 0.666667 | 0.25 | 0.01751 (12) | |
| F1 | 0.06201 (16) | 0.53101 (8) | 0.14734 (10) | 0.0276 (2) | |
| F2 | 0.19679 (9) | 0.39358 (19) | 0.35039 (10) | 0.0289 (3) | |
| N1 | 0.666667 | 0.333333 | 0.3731 (2) | 0.0235 (4) | |
| N2 | 0 | 0 | 0.5855 (2) | 0.0269 (5) | |
| H1n1 | 0.586 (3) | 0.172 (6) | 0.394 (4) | 0.089 (10)* | |
| H1n2 | −0.146 (4) | −0.073 (2) | 0.549 (3) | 0.071 (8)* | |
| H2n1 | 0.666667 | 0.333333 | 0.291 (13) | 0.19 (5)* | |
| H2n2 | 0 | 0 | 0.675 (8) | 0.10 (2)* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| N1 | 0.0239 (5) | 0.0239 (5) | 0.0227 (7) | 0.0119 (2) | 0 | 0 |
| N2 | 0.0247 (5) | 0.0247 (5) | 0.0315 (12) | 0.0123 (2) | 0 | 0 |
| Si | 0.01716 (15) | 0.01716 (15) | 0.0182 (2) | 0.00858 (7) | 0 | 0 |
| F1 | 0.0221 (4) | 0.0301 (3) | 0.0279 (4) | 0.0110 (2) | −0.0072 (2) | −0.00360 (14) |
| F2 | 0.0297 (3) | 0.0244 (4) | 0.0308 (4) | 0.0122 (2) | 0.00440 (15) | 0.0088 (3) |
Geometric parameters (Å, º) top
| Si—F1 | 1.700 (1) | Si—F2 | 1.695 (1) |
| Si—F1i | 1.700 (1) | Si—F2i | 1.695 (1) |
| Si—F1ii | 1.700 (1) | Si—F2ii | 1.695 (1) |
| | | |
| H1n1—N1—H1n1iii | 115 (3) | F1—Si—F2 | 89.68 (5) |
| H1n1—N1—H1n1iv | 115 (3) | F1—Si—F2i | 179.22 (5) |
| H1n1—N1—H2n1 | 104 (2) | F1—Si—F2ii | 89.68 (5) |
| H1n1iii—N1—H1n1iv | 115 (3) | F1i—Si—F1ii | 89.77 (5) |
| H1n1iii—N1—H2n1 | 104 (2) | F1i—Si—F2 | 89.68 (5) |
| H1n1iv—N1—H2n1 | 104 (2) | F1i—Si—F2i | 89.68 (5) |
| H1n2—N2—H1n2v | 103 (3) | F1i—Si—F2ii | 179.22 (5) |
| H1n2—N2—H1n2vi | 103 (3) | F1ii—Si—F2 | 179.22 (5) |
| H1n2—N2—H2n2 | 115 (2) | F1ii—Si—F2i | 89.68 (5) |
| H1n2v—N2—H1n2vi | 103 (3) | F1ii—Si—F2ii | 89.68 (5) |
| H1n2v—N2—H2n2 | 115 (2) | F2—Si—F2i | 90.87 (6) |
| H1n2vi—N2—H2n2 | 115 (2) | F2—Si—F2ii | 90.87 (6) |
| F1—Si—F1i | 89.77 (5) | F2ii—Si—F2i | 90.87 (6) |
| F1—Si—F1ii | 89.77 (5) | | |
| Symmetry codes: (i) −y+1, x−y+1, z; (ii) −x+y, −x+1, z; (iii) −y+1, x−y, z; (iv) −x+y+1, −x+1, z; (v) −y, x−y, z; (vi) −x+y, −x, z. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1n1···F1vii | 0.85 (3) | 2.49 (4) | 2.975 (2) | 117 (3) |
| N1—H1n1···F1vi | 0.85 (3) | 2.65 (4) | 2.962 (2) | 103 (3) |
| N1—H1n1···F2iii | 0.85 (3) | 2.28 (2) | 2.972 (1) | 139 (3) |
| N1—H1n1···F2vi | 0.85 (3) | 2.28 (2) | 2.972 (1) | 139 (3) |
| N2—H1n2···F1viii | 0.82 (3) | 2.58 (3) | 3.023 (1) | 115 (2) |
| N2—H1n2···F1ix | 0.82 (3) | 2.58 (3) | 3.023 (1) | 115 (2) |
| N2—H1n2···F2v | 0.82 (3) | 2.29 (3) | 3.022 (2) | 149 (3) |
| N1—H2n1···F1x | 0.8 (1) | 2.45 (7) | 2.962 (2) | 124 (3) |
| N1—H2n1···F1i | 0.8 (1) | 2.45 (7) | 2.962 (2) | 124 (3) |
| N1—H2n1···F1vi | 0.8 (1) | 2.45 (7) | 2.962 (2) | 124 (3) |
| N2—H2n2···F2viii | 0.86 (8) | 2.62 (5) | 3.241 (2) | 130 (1) |
| N2—H2n2···F2xi | 0.86 (8) | 2.62 (5) | 3.241 (2) | 130 (1) |
| N2—H2n2···F2xii | 0.86 (8) | 2.62 (5) | 3.241 (2) | 130 (1) |
| Symmetry codes: (i) −y+1, x−y+1, z; (iii) −y+1, x−y, z; (v) −y, x−y, z; (vi) −x+y, −x, z; (vii) x−y+1, x, z+1/2; (viii) x−y, x, z+1/2; (ix) y−1, −x+y−1, z+1/2; (x) x+1, y, z; (xi) y, x, z+1/2; (xii) −x, −y, z+1/2. |
Experimental details
| Crystal data |
| Chemical formula | 2(NH4)·(SiF6) |
| Mr | 178.15 |
| Crystal system, space group | Hexagonal, P63mc |
| Temperature (K) | 290 |
| a, c (Å) | 5.8955 (10), 9.599 (1) |
| V (Å3) | 288.93 (8) |
| Z | 2 |
| Radiation type | Mo Kα |
| µ (mm−1) | 0.46 |
| Crystal size (mm) | 0.40 × 0.35 × 0.30 |
| |
| Data collection |
| Diffractometer | Kuma-4
diffractometer |
| Absorption correction | – |
No. of measured, independent and
observed [I > 3σ(I)] reflections | 1682, 361, 348 |
| Rint | 0.020 |
| (sin θ/λ)max (Å−1) | 0.703 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.018, 0.022, 1.70 |
| No. of reflections | 361 |
| No. of parameters | 31 |
| No. of restraints | ? |
| H-atom treatment | All H-atom parameters refined |
| Δρmax, Δρmin (e Å−3) | 0.39, −0.39 |
Selected bond lengths (Å) top| Si—F1 | 1.700 (1) | Si—F2 | 1.695 (1) |
| Si—F1i | 1.700 (1) | Si—F2i | 1.695 (1) |
| Si—F1ii | 1.700 (1) | Si—F2ii | 1.695 (1) |
| Symmetry codes: (i) −y+1, x−y+1, z; (ii) −x+y, −x+1, z. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1n1···F1iii | 0.85 (3) | 2.49 (4) | 2.975 (2) | 117 (3) |
| N1—H1n1···F1iv | 0.85 (3) | 2.65 (4) | 2.962 (2) | 103 (3) |
| N1—H1n1···F2v | 0.85 (3) | 2.28 (2) | 2.972 (1) | 139 (3) |
| N1—H1n1···F2iv | 0.85 (3) | 2.28 (2) | 2.972 (1) | 139 (3) |
| N2—H1n2···F1vi | 0.82 (3) | 2.58 (3) | 3.023 (1) | 115 (2) |
| N2—H1n2···F1vii | 0.82 (3) | 2.58 (3) | 3.023 (1) | 115 (2) |
| N2—H1n2···F2viii | 0.82 (3) | 2.29 (3) | 3.022 (2) | 149 (3) |
| N1—H2n1···F1ix | 0.8 (1) | 2.45 (7) | 2.962 (2) | 124 (3) |
| N1—H2n1···F1i | 0.8 (1) | 2.45 (7) | 2.962 (2) | 124 (3) |
| N1—H2n1···F1iv | 0.8 (1) | 2.45 (7) | 2.962 (2) | 124 (3) |
| N2—H2n2···F2vi | 0.86 (8) | 2.62 (5) | 3.241 (2) | 130 (1) |
| N2—H2n2···F2x | 0.86 (8) | 2.62 (5) | 3.241 (2) | 130 (1) |
| N2—H2n2···F2xi | 0.86 (8) | 2.62 (5) | 3.241 (2) | 130 (1) |
| Symmetry codes: (i) −y+1, x−y+1, z; (iii) x−y+1, x, z+1/2; (iv) −x+y, −x, z; (v) −y+1, x−y, z; (vi) x−y, x, z+1/2; (vii) y−1, −x+y−1, z+1/2; (viii) −y, x−y, z; (ix) x+1, y, z; (x) y, x, z+1/2; (xi) −x, −y, z+1/2. |

 Subscribe to Acta Crystallographica Section E: Crystallographic Communications
Subscribe to Acta Crystallographica Section E: Crystallographic Communications
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
[email protected] for assistance.
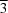 m1) of the title compound. The H atoms in the present structure are ordered. Three or even four F atoms are hydrogen-bond acceptors for each H atom. The structure is isostructural with modifications of (NH4)2MnF6, K2GeF6 and Rb2GeF6.
m1) of the title compound. The H atoms in the present structure are ordered. Three or even four F atoms are hydrogen-bond acceptors for each H atom. The structure is isostructural with modifications of (NH4)2MnF6, K2GeF6 and Rb2GeF6.
 journal menu
journal menu











![[Figure 1]](https://journals.iucr.org/e/issues/2001/10/00/wn6052/wn6052fig1thm.gif)




Until now, two modifications of (NH4)2SiF6 have been known (ICSD; Bergerhoff et al., 1983; PDF-2, 2001): trigonal P3m1 (Schlemper & Hamilton, 1966; PDF-2 44–1424 and 72–1548) and cubic Fm3 m (Hanic, 1966; PDF-2 07–0013, 72–1552 and 72–1759). The trigonal and cubic modifications are also known by their respective mineralogical names bararite and cryptohalite. The cubic modification is stable at room temperature (Schlemper & Hamilton, 1966). In the cubic modification, the positions of the H atoms were determined; they were not determined in the trigonal modification. According to both, the neutron-diffraction study (Schlemper & Hamilton, 1966) and the electron-diffraction study (Vainshtein & Stasova, 1956) the H atoms in the cubic modification are disordered.
In this study, two modifications were grown in the same beaker, viz. hexagonal and cubic. The crystals of each modification were quite easily distinguishable in cross-polarized light as well as by their habit. While the habit of the cubic modification was cubic, the hexagonal modification grew as trigonal or hexagonal pyramids. The typical size of both crystal modifications was several tenths of a millimetre.
Attempts at chemical analysis were hindered by the small amount of available crystals. An electron microprobe analysis [Jeol JXA733 (Jeol Ltd., Tokyo, Japan) with X-ray analyzer KEVEX (Delta Class Analyzer; Kevex Instruments, San Carlos, California, USA)] was not successful in the determination of the proportion of constituent elements P, Si, O and F because of the fluffy grains on the surface of the sample. These fluffy grains contained predominantly Si, F and K. It should be noted that the microprobe analysis was not performed on the sample that was used for the structure determination. Nevertheless, the satisfactory result of the structure analysis shows that (NH4)2SiF6 can also form a superstructure, either as a transitory state or stabilized by traces of K.
The previously determined trigonal modification shows similarities with the present structure in the orientation of the Si—F bonds with regard to the unit-cell axes. It should be noted that the structure determined in this study is isostructural with one of the known modifications of K2GeF6 (Bode & Brockmann, 1952) and Rb2GeF6 (ibid.). The structure is also isostructural with (NH4)2MnF6 (Kaskel & Straehle, 1997) where the positions of the H atoms were determined.
The H atoms were readily seen in the difference Fourier maps and could be easily refined with no applied constraints or restraints. All the H atoms are ordered. Each N atom in the structure is surrounded by six F atoms with fairly similar N···F distances. The distribution of four H atoms among six surrounding F atoms results in the distances between the H and F atoms being unequal despite the regularity of the N···F distances. Table 2 lists the closest N···F distances and angles.