issue contents
January 1998 issue
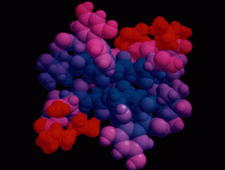
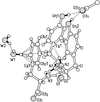
Cover illustration: The balhimycin dimer, with citrate and acetate ions in the binding pockets, colour coded on a scale where the most mobile atoms are red and the least mobile are blue. The binding pockets are clearly the most mobile, consistent with the idea that they can open and close to complex with cell-wall binding peptides. Courtesy of George Sheldrick.
international union of crystallography
Free 

topical reviews
The inherent features of the vapour-diffusion and microbatch methods for protein crystallization are discussed and guidelines for the conversion of crystallization conditions from one method to another are presented.
research papers
The overall molecular structures of the monoclinic and tetragonal forms of cytochrome b1 are essentially identical. in the monoclinic form, the asymmetric unit consists of 24 monomers (Mr = 450 kDa).
PDB reference: 1bfr
In the crystal structure of Tyr-Tyr-Phe dihydrate, one of the water molecules forms a hydrogen bond with the Phe side chain. In a database study four related examples in peptides are found.
The crystal structure of pepsin IIb from Atlantic cod has been refined with data to 2.16 Å resolution. The structure is very similar to pepsin from pig.
PDB reference: Pepsin IIb, 1am5
Structure of Bacteroides fragilis β-lactamase.
PDB reference: β-lactamase, 1bmi
The crystal structure of 36 kDa soluble lytic transglycosylase Slt35 from Escherichia coli has been determined at 1.7 Å resolution using wARP, a powerful procedure to improve and extend crystallographic phases. Without amny model building or electron-density map interpretation, the procedure resulted in phases which differed by only 21.7° from those calculated from the final Slt35 model. The weight WwARP, calculated by wARP, proved to be a better indicator of the phase improvement than the Rfree factor.
PDB reference: Transglycosylase Slt35, 1ltm
The effects of temperature increase on monoclinic and tetragonal lysozyme and on ribonuclease A single crystals were investigated by polarizing microscopy, X-ray diffraction and laser Raman spectroscopy. Although the crystal lattice of proteins is stabilized by only few intermolecular contacts, its breakdown with increasing temperature is primarily due to thermal unfolding of the proteins.
Iterative skeletonization was compared with the procedure of pseudo-atom introduction followed by refinement of the program suite DEMON/ANGEL. When non-crystallographic symmetry is present in the crystals, pseudo-atom introduction followed by refinement performs much better, leading to phase improvement.
crystallization papers
Due to their intrinsic flexibility NMR structures are often difficult to use in molecular replacement. The crystal structure of the p53 oligomerization domain was solved by molecular replacement using a previously determined NMR structure as a search template and refined at 1.5 Å resolution.
PDB reference: p53 (residues 326-356), 1aie
Orthorhombic crystals of the plant lectin Scilla campanulata agglutinin in complex with the disaccharide mannose-α1,6-D-mannose have been obtained which diffract X-rays to beyond 1.5 Å resolution at 277 K. SRS data to 1.6 Å resolution have been collected and the structure of the complex has been solved by molecular replacement.
We report the expression and crystallization of human Neutrophil Gelatinase Associated Lipocalin, a small glycoprotein, secreted by neutrophils, that binds mediators of mucosal immune responses.
The tetrameric 12S form of yeast phosphofructokinase, obtained by limited proteolytic cleavage of the native enzyme, was crystallized under a variety of conditions. The crystals have been characterized in the X-ray beam and are suitable for crystallographic studies.
The crystal structures of a monoclinic and hexagonal form of a Tet repressor–operator complex were solved by molecular replacement. The oligonucleotides show an obvious tendancy to form continuous DNA double helices.
High diffraction quality crystals of a TolB derivative tagged with six histidines have been obtained using the SAmBA software to design crystallization protocols.
Tyrosine aminotransferase from Trypanosoma cruzi has been crystallized from PEG 4000 at pH 6.8. The crystals belong to the monoclinic space group P21 and diffract to at least 2.7 Å.
Crystallization of human platelet profilin complexed with an oligo proline peptide. this structure will provide atomic detail about the localization of profilin, a function required for proper actin polymerization.
Sialidase L, the first sialidase with strict specificity, was crystallized. Threading and molecular-replacement calculations confirmed the existence of a bacterial sialidase-like domain.
CeIF, a processive endocellulase, member of the glycosyl hydrolases family 48, has been crystallized in the presence of a newly synthesized thiooligosaccharide.
Purification and crystallization of porcine quinolate phosphoribosyl transferase is described. Results from the preliminary X-ray diffraction studies are presented.
The RNA helicase domain of hepatitis C virus was crystallized and a native data set to 2.3 Å resolution was obtained. The crystals are suitable for structure determination.
Crystallization of the N—(1-D-carboxyethyl)-L-norvaline dehydrogenase from Arthrobacter sp. strain 1C in the presence or absence of cofactor and substrates.
A bleomycin (Bm) binding protein produced by Bm-producing Streptomyces verticillus was crystallized in a form suitable for X-ray diffraction analysis. The crystals diffract X-rays well and the diffraction intensity data was collected up to 1.5 Å resolution with a merging R value of 0.054.
Endo-β-1,4-mannanase from Pseudomonas fluorescens subspecies cellulosa has been crystallized. The crystals belong to space group R32 with cell dimensions a = b = 155.4 and c = 250.8 Å (hexagonal setting) and contain three molecules in the asymmetric unit.
The putrescine receptor (PotF) of the periplasmic putrescine transport system in E. coli has been crystallized. Diffraction data have been collected from the native crystals at 2.3 Å resolution, as well as from two heavy atom derivatives.
The flavoprotein component of E. coli sulfite reductase has been crystallized. A 3 Å resolution data set was collected using synchrotron radiation.
PAP-S, a ribosome-inactivating protein with three N-glycosylation sites, was crystallized under high protein concentration (100 mg ml−1) and high temperature (306 K). The crystals, diffracting to 1.8 Å, provide a chance to reveal the accurate structure of functionally significant glucosamine groups bound to the protein.
Histidine-containing phosphotransfer (HPt) domain at the C-terminus of the anaerobic sensor kinase ArcB has been cocrystallized with the chemotaxis response regulator CheY by a hanging-drop vapor-diffusion method. This is the first crystallization of a protein–protein complex formed by a transmitter domain of sensor kinase and a receiver domain of response regulator in the two-component signal transduction system.
Protein kinase CK2 (catalytic subunit) was successfully crystallized for the first time. The crystals obtained diffract X-ray to at least 2.0 Å resolution.
short communications
Bovine heart ubiquinol–cytochrome c reductase stabilized with sucrose monocaplate was crystallized to provide X-ray diffraction quality up to 2.8 Å resolution.
Crystals of human theta-class glutathione S-transferase have been grown and they have been characterized by collecting X-ray data to 3.2 Å resolution
It is shown that an R factor calculated from the probe structure with some omitted parts can be used to evaluate solutions of the molecular-replacement method.
Crystallization of several proteins is described using high concentrations of non-ionic surfactants as precipitants. At the surfactant concentrations used, glass formation is possible upon transfer to cryogenic temperatures.


 journal menu
journal menu