issue contents
May 1998 issue
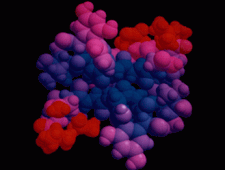
Cover illustration: The balhimycin dimer, with citrate and acetate ions in the binding pockets, colour coded on a scale where the most mobile atoms are red and the least mobile are blue. The binding pockets are clearly the most mobile, consistent with the idea that they can open and close to complex with cell-wall binding peptides. Courtesy of George Sheldrick.
research papers
Native, zinc-removed, and mercury-replaced models for carboxypeptidase A have been refined at high resolution, and compared with older models available for this enzyme.
Anisotropic B-factor refinement compared with normal mode calculations.
Structure of Bacillus cereus β-lactamase II.
PDB reference: ZincII β-lactamase, 1bme
With molecules as extended as DNA helices, rotation/translation molecular replacement searches are greatly assisted if one abandons a formal separation of rotation versus translation, and instead searches systematically for the best-established variables first. For stacked helical rods of DNA, this means (1) orientation of rods in space (two rotations), then (2) position of rods in the in the unit cell (two translations), and only then (3) helix substructure within a rod.
The crystal structure of the complex of bovine pancreatic phospholipase A2 with a transition-state analogue inhibitor has been determined and compared with the free enzyme.
PDB reference: Phospholipase A2, 1mkv
The trigonal form of the recombinant bovine pancreatic phospholipase A2 has been re-refined at a higher resolution of 1.72 Å.
PDB reference: Phospholipase A2, 1mkt
X-ray structure of azurin I obtained from Alcaligenes xylosoxidans NCIMB 11015 revealed the copper coordination geometry of Cu—N (His46) 2.0 Å, Cu—N (His117) 1.9 Å, Cu—S (Cys112) 2.2 Å, Cu—S (Met121) 3.1 Å and Cu—O (Gly45) 2.5 Å. C—O (Gly45) is 0.25 Å shorter than its counterpart in azurin II.
PDB reference: Azurin I, 1rkr
The three-dimensional structure and associated solvent of human carboxyhemoglobin at 2.2 Å resolution are compared with other R-state and T-state human hemoglobin structures.
PDB reference: Carboxyhemoglobin, 1aj9
Structure-factor amplitudes reduced from Laue diffraction images were used to refine a protein crystal structure at 1.7 Å resolution. The strategies for obtaining accurate and complete Laue data sets are shown.
PDB reference: Restrictocin, 1aqz
Two double mutants (F14N/V21T and F14N/V86T) of CheY have been analysed by X-ray crystallography and urea denauration. The F14N mutation stabilizes the protein by −12.1 kJ mol−1 by reverse hydrophobic effect an N-cap formation, while the V to T mutations compensate the destabilization caused by burying a polar group by the formation of new hydrogen bonds.
Large diffracting crystals of alcohol dehydrogenase from Sulfolobus solfataricus were grown in microgravity. Improvement of size and diffraction resolution limit was also observed.
Improved real-space quality indices correlate well with the subtle errors remaining in fully refined macromolecular structure. They can be used to identify the residues or atoms with the largest errors.
A method for locating heavy atoms in the unit cell of macromolecular crystals by a full-symmetry translation function is described.
crystallization papers
The extracellular ribonuclease RNase from S. aureofaciens is inhibited by barstar. A complex of RNase with wild-type barstar has been crystallized and a preliminary investigation undertaken.
The preliminary crystallographic characterization of two Fab fragments (Fab3A2, Fab6A) of two monoclonal antibodies raised against epitopes of hCG, is reported here. The Fab3A2 antibody recognizes a linear epitope on the C-terminal peptide of peptide of the β-subunit of hCG and the Fab6A a conformational epitope of hCG.
The alanine dehydrogenase from Phormidium lapideum has been crystallized in space group R32, cell dimensions a = b = 123.1, c = 184.8 Å, with a monomer in the asymmetric unit.
Urease from the soil bacterium Bacillus pasteurii has been crystallized both in the native and inhibited form in space group P6322. With the use of synchrotron radiation the crystals provide high-resolution diffraction (2.00 Å for the native and 1.65 Å for the inhibited enzyme) and good data completeness.
Homoserine dehydrogenase from Saccharomyces cerevisiae has been crystallized in three different forms. Crystals grown in the presence of the co-factor NAD+ diffract to 2.2 Å resolution, crystals grown in the presence of L-homoserine and an NAD+ analogue diffract to 2.7 Å and crystals of the apo-enzyme have a diffraction limit of 3.9 Å resolution.
A novel maltogenic amylase, exhibiting an α-1,6-glycosidic bond formation, was crystallized, and a data set to 2.7 Å resolution was obtained.
Crystals of PeIL, a member of a new family of pectate lyases, have been grown and are suitable for structure determination. Data have been collected and processed to 2.3 Å resolution.
The estrogen receptor hormone binding domain has been crystallized as an intact GST fusion. The crystals have been characterized by electron microscopy.
The NADP-dependent β-keto acyl carrier protein reductase from E. coli has been crystallized by the hanging-drop method of vapour diffusion using PEG of average molecular weight 1450.
Acetyl xylan esterase core protein has been crystallized. Diffraction data have been collected to 2.3 Å resolution.
Human p58 killer cell inhibitory receptor (specific for HLA-Cw1,3,7) has been crystallized in space group P6122 (or P6522). Crystal growth was optimized by addition of octyl-β-glucoside.
Crystals have been grown from a recombinant form of hORF6 that is a member of the novel antioxidant enzyme family. A native data set has been collected to 2.0 Å resolution.
Porphobilinogen synthase catalyzes the condensation of two identical molecules of 5-aminolevulinic acid to give a pyrrole derivative. The enzyme from E. coli gives tetragonal crystals which diffract to 3.0 Å resolution.
Bowman–Birk inhibitor from barley seeds has been crystallized and X-ray data have been collected to 1.9 Å.
Crystallization of 3-isopropylmalate dehydrogenase has been achieved by the vapor-diffusion method using ammonium sulfate with addition of 2-methyl-2,4-pentanediol.
The thermostable catechol 2,3-dioxygenase of Bacillus stearothermophilus has been purified and crystallized. A preliminary X-ray diffraction study has been reported.
Recombinant thiaminase I from Bacillus thiaminolyticus has been crystallized. Three orthorhombic crystal forms have been characterized.
A soluble form of the MHC class I homologue MICA has been expressed and crystallized.
A native data set has been collected to 2.3 Å resolution on a new crystal form for the vanadium-dependent bromoperoxidase from Corallina officinalis. A model for the oligomeric arrangement of the molecule has been deduced from the self-rotation functions, and the heavy-atom positions of a mercury derivative.
short communications
Two crystal forms of ligand-free hGSTM2-2 were grown which diffract to 2.5 and 3.0 Å, respectively.
Extending explicit solvent definition results in improved map quality in previously ill-defined regions.
Three crystal forms of the biosynthetic threonine deaminase from E. coli, an allosteric tetramer with key regulatory functions, are described. Diffraction datasets to 2.8 Å resolution (native) and 2.9 Å resolution (isomorphous uranyl derivative) have been collected and map interpretation is in progress.
Cytochrome P450cam from Pseudomonas putida has been crystallized using a combination of site-directed mutagenesis and the hanging-drop vapour-diffusion method. X-ray data collection of camphor-bound (C334A)P450cam at 100 K is described.
A recombinant form of mammalian casein I δ containing the catalytic domain and an auto-inhibitory domain was expressed in E. coli, purified and crystallized. A conserved intermolecular contact suggests the formation of dimers which would inhibit the activity of this protein kinase.
fast communications
A human uroporphyrinogen decarboxylase from E. coli has been crystallized. Diffraction data have been collected to 3 Å resolution.
A flash-annealing technique has been developed which utilizes brief thawing and rapid freezing cycles of glycerol kinase crystals in a cold nitrogen stream. This method increases the diffraction limits and simultaneously decreases the mosaicity of the crystals.


 journal menu
journal menu