issue contents
July 1999 issue
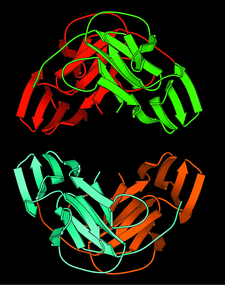
Cover illustration: The tetrameric arrangement of the mannose-specific plant lectin Scilla campanulata agglutinin, with each subunit in a different colour (p. 1264).
research papers
In a novel three-dimensional crystal of bacteriorhodopsin obtained by successive fusion of the vesicular assemblies, the native structure of the trimeric unit is preserved. One phospholipid bound to a crevice between monomers in the trimeric unit is suggested to act as a glue for formation of the trimer.
PDB reference: bacteriorhodopsin, 1bm1
The crystal structure of the HPt domain complexed with CheY was determined at 2.68 Å resolution. It revealed that the HPt domain has a rigid structure in contrast with CheY, which shows some significant conformational changes.
PDB reference: HPt–CheY complex, 1bdj
The three-dimensional structure of the mannose-specific bulb lectin Scilla campanulata agglutinin (SCA) has been determined and refined at 1.7 Å resolution. The SCA backbone fold contains a threefold symmetric β-prism made up of three antiparallel four-stranded β-sheets, each containing a putative saccharide-binding site.
PDB reference: mannose-specific bulb lectin, 1b2p
Haloalkane dehalogenase has been refined to 1.15 Å resolution. The structure is described in detail and an analysis of the protein's geometry and stereochemistry is presented. C—H O interactions are found to satisfy the hydrogen-bond acceptor capacity of main-chain carbonyl O atoms in the central β-sheet.
O interactions are found to satisfy the hydrogen-bond acceptor capacity of main-chain carbonyl O atoms in the central β-sheet.
PDB reference: haloalkane dehalogenase, 1b6g
The crystal structures of the deoxy- and carbonmonoxyhaemoglobin from D. akajei, a stingray, have been determined at 1.6 and 1.9 Å resolution, respectively. They are compared with the respective structures of human HbA.
Iterative rigid-body simulated-annealing refinement of even roughly positioned polyalanine models, employing successively fewer residues per rigid body and at successively higher resolution, can converge to an essentially correct polyalanine trace of the target structure. The application of the method to the structure determination of a 4-α-helical bundle is discussed.
PDB reference: A31P Rop mutant, 1b6q
Three investigators individually built and refined the same X-ray structure at 2.6 Å resolution. The various models showed different patterns of errors, and together with the final 1.8 Å structure, allowed testing of various error-finding tools.
The details of a triplet-phase data collection are presented. A total of about 850 triplet phases from tetragonal lysozyme were determined.
An evaluation of macromolecular crystal annealing techniques.
Description of SMILE2DICT, a program for the production of small-molecule dictionaries from SMILES representations.
crystallization papers
Crystals of a recombinant major urinary protein have been obtained. The crystals are monoclinic and diffract to 1.7 Å resolution.
A structural study of snake muscle fructose 1,6-bisphosphatase is presented.
High-resolution crystals of the R2 subdomain of the mouse c-Myb DNA-binding domain have been obtained by the vapour-diffusion method. Crystals of several R2 mutants at position 103 are also obtained and are isomorphous to the wild-type R2 crystals.
Carboxylesterase EST2 from A. acidocaldarius has been crystallized. The crystals diffract to 2.4 Å and belong to space group P41212 or P43212, with unit-cell parameters a = b = 78.8, c = 106.4 Å.
First report of purification and crystallization of the E. coli enzyme which catalyzes the second step of arginine biosynthesis and which may be a member of a new family of acylphosphate-forming enzymes.
Crystallization of D-glyceraldehyde-3-phosphate dehydrogenase from the archaeon M. fervidus is reported. Crystals diffract to 2.1 Å and belong to space group P21212.
PDB reference: D-glyceraldehyde-3-phosphate dehydrogenase, 1cf2
Apo and ligand-bound forms of the cytoplasmic molybdate-binding protein ModG from A. vinelandii have been crystallized in space groups P6322 and P321, respectively. X-ray data were collected on both forms to a maximum resolution of 2.0 Å.
The endo-polygalacturonase from the phytopathogenic fungus F. moniliforme has been crystallized. Crystals, containing at least three different glycoforms, diffract to 1.9 Å resolution and belong to the orthorhombic space group P212121.
The minimal Z-DNA binding domain Za of human adenosine deaminase (ADAR1) has been crystallized in complex with a double-stranded DNA fragment, d(TCGCGCG)2. Raman measurements show that the bound DNA substrate adopts the left-handed Z conformation.
The integrin α1 I domain was purified and crystallized. The crystals diffract to 2.0 Å and are suitable for X-ray structure determination.
Overexpression and crystallization of a type IIB aldolase: 2-dehydro-3-deoxygalactarate aldolase from E. coli.
Crystallization and preliminary characterization of the essential dengue virus NS3 serine protease complexed with a Bowman–Birk-type inhibitor from mung beans are reported.
Crystals of the RNA polymerase subunit RPB5 from S. cerevisiae have been obtained by vapour-diffusion techniques and preliminary diffraction data are presented here.


 journal menu
journal menu