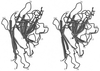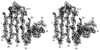issue contents
August 1999 issue
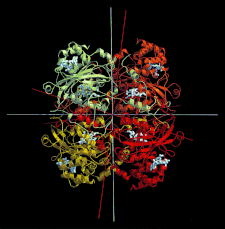
Cover illustration: A ribbon diagram of the polypeptide backbone for the entire tetramer (Mr = 260 000) of beef liver catalase containing four heme groups and four NADPH coenzyme molecules (p. 1383).
research papers
The crystal structures of the peanut-lectin complexes of methyl-β-galactose and N-acetyllactosamine and those involving other sugars demonstrate the important role of water molecules in generating carbohydrate specificity. A comparative study also provides a structural explanation for the exclusive specificity of peanut lectin for galactose at the monosaccharide level as against that of other related lectins for galactose and N-acetylgalactosamine.
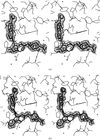
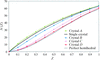
The structure of orthorhombic beef liver catalase crystals was determined by X-ray crystallography and studied as well by atomic force microscopy. Combination of the two techniques provided explanations of numerous features of the crystal growth process.
PDB reference: orthorhombic beef liver catalase, 4blc
Structures of five factor Xa inhibitors bond to bovine trypsin are described at 1.8 Å.
PDB references: trypsin–2-aminobenzimidazole complex, 1qa0; trypsin–2-{5-[amino(imino)methyl]-2-hydroxyphenoxy}-6-[3-(4,5-dihydro-1H-imidazol-2-yl)phenoxy]pyridine-4-carboxylic acid complex, 1qbn; trypsin–7-[(6-{[1-(1-iminoethyl)piperidin-4-yl]oxy}-2-methyl-1H-benzimidazol-1-yl)methyl]naphthalene-2-carboximidamide complex, 1qbo; trypsin–1-(2-{5-[amino(imino)methyl]-2-hydroxyphenoxy}-6-[3-(4,5-dihydro-1-methyl-1H-imidazol-2-yl)phenoxy]pyridin-4-yl)piperidine-3-carboxylic acid complex, 1qb1; trypsin–3,3′-[3,5-difluoro-4-methyl-2,6-pyridinediylbis(oxy)] bis(benzenecarboximidamide) complex, 1qb6; trypsin–7-[(2-{[1-(1-iminoethyl)piperidin-4-yl]oxy}-9H-carbozol-9-yl)methyl]naphthalene-2-carboximidamide complex, 1qb9
The crystal structures of three double-stranded RNA heptamers derived from the tRNAAla acceptor stem were refined as pseudo-merohedral twins at high resolution.
The frequency of occurrence of X in the consensus glycosylating sequence Asn-X-Ser/Thr in proteins and the conformational and hydrogen-bonding features of the triplet have been analysed using the PDB database.
The geometry of interaction of different aromatic side chains with the indole ring of tryptophan is presented.
A method for the simplification of the solution of MIR derivative sites and the improvement of isomorphism using a derivative backsoak is described. MIR sites are depleted, soak/backsoak difference Pattersons are simpler and the subset of sites may be used as a starting point for cross-phasing.
Geometrical data for Ca, Mg, Mn, Fe, Cu and Zn bonded to ligands which are analogues of amino-acid side chains have been extracted from the Cambridge Structural Database and tabulated. These could be useful in the determination, refinement or assessment of metalloprotein structures, or in the understanding of function.
Precipitants can be classified into three types for one particular protein. Combination of hard and soft precipitants has many advantages in protein crystallization.
The feasibility of two-wavelength MAD experiments and the optimal wavelength choice are discussed.
crystallization papers
Three different crystal forms of the minor group human rhinovirus serotype 2 (HRV2) hase been obtained. Crystals belonging to space group I222 diffracted beyond 1.8 Å resolution and contained 15 protomers in the asymmetric unit. Structure refinement on this crystal form is now in progress.
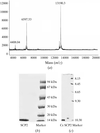
Purple acid phosphate from pig allantoic fluid has been crystallized in the space group P212121 with unit-cell dimensions a = 66.8, b = 70.3 and c = 77.1 Å. Using cryocooling techniques a complete data set to 1.55 Å resolution has been obtained.
Crystals of ROO, a terminal oxidase capable of reducing dioxygen to water, have been characterized. The non-crystallographic twofold axis within the homodimer is parallel to a twinning axis found in pseudo-merohedrally twinned crystals of ROO.
Crystals of potent inhibitor of blood platelet activation have been grown. Molecular replacement has not been successful in producing a trial structure, most likely because of poor diffraction.
Crystallization of rhodanese from A. vinelandii is presented.
The substrate-free E. coli tyrosine amintotransferase (eTAT) bound with a cofactor pyridoxal 5′-phosphate (PLP) was crystallized in trigonal space group P32. The low-resolution crystal structure shows that the substrate-free eTAT has a tertiary structure similar to the open form of aspartate aminotransferase.
PDB reference: tyrosine aminotransferase, 3tat
Sterol carrier protein 2 was expressed in Escherichia coli and purified to electrophoretic homogeneity. Single crystals were obtained suitable for high-resolution X-ray structure analysis.
An essential protein in bacterial cell-wall biosynthesis and vancomycin resistance in the clinically important bacterium E. faceium BM4147 has been expressed and crystallized in a form suitable for X-ray structure determination.
Crystals of rat m-calpain have been obtained in two forms. Diffraction data have been collected to 2.6 Å for the P1 form and to 2.15 Å for the P21 form.
Crystallization and crystallographic study of HIP/PAP, a human C-lectin overexpressed in primary liver cancer. Presentation of a molecular-replacement solution using the human lithostathine structure and the AMoRe software.


 journal menu
journal menu