issue contents
December 2001 issue
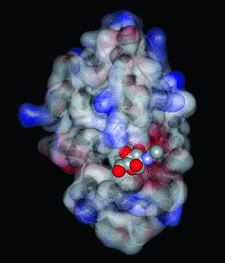
Cover illustration: The molecular surface of lysozyme with a CPK representation of bound N-acetylglucosamine. The protein surface is colored according to the electrostatic potential, where blue is positive and red negative (p. 1837).
research papers
Open  access
access
 access
accessA likelihood-based density-modification method is extended to include pattern recognition of structural motifs.
Open  access
access
 access
accessA map-likelihood function is described that can yield phase probabilities with very low model bias.
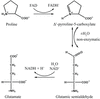
The structure of DAOCS was determined by isomorphous replacement from crystals twinned by merohedry.
The structure of a low molecular-weight pectate lyase was determined at 1.5 Å resolution. The structure is a β-helix domain motif.
PDB reference: pectate lyase, 1ee6
The high-resolution X-ray analysis of phospholipase A2 from D. russelli pulchella clearly brings out the differences between the two crystallographically independent molecules in the structure. The structure presents a unique case in which molecular association leads to blocking of the active site in one molecule while retaining the open geometry of the site in the other.
PDB reference: phospholipase A2, 1fb2
Variations of interactions of brome mosaic viruses in solution and the immediate formation of microcrystals were characterized by small-angle X-ray scattering.
Family 11 endo-β-1,4-xylanases degrade xylan, the main constituent of plant hemicelluloses. The structure of a family 11 endo-xylanase from Streptomyces sp. S38 at 2.0 Å is reported.
PDB reference: Xyl1, 1hix
An N-particle approach, combining loose-atom refinement with extensive application of geometrical restraints, allows optimization of a simple polyalanine test structure starting from random atom distributions.
The monoclinic crystal form of human peroxiredoxin 5 has been determined by the SIR method after detwinning. The presence of eight molecules in the asymmetric unit allows an analysis of the mobility.
PDB reference: Peroxiredoxin 5, 1h4o
The binding of N-acetylglucosamine to chicken egg lysozyme was investigated by high-resolution X-ray powder diffraction.
The structure of full-length grancalcin, a cytosolic Ca2+-binding protein, has been determined in the apo form and with one EF-hand motif within the dimer occupied by a Ca2+ ion.
The structure of the greylag goose oxy haemoglobin shows quaternary structural differences compared with that of its highland relative bar-headed goose. The four differing residues between the two species may be responsible for the differences in their oxygen affinity and quaternary structure.
PDB reference: greylag goose oxy haemoglobin, 1faw
Using Karle–Hauptman matrices as the basis of an algebraic procedure of phase determination is proposed. The method allows the determination of the sulfur/chlorine substructure of lysozyme based on the anomalous signal obtained from in-house measurements.
The structure of a recombinant major urinary protein has been determined at 1.75 Å resolution. The crystal structure is compared with the NMR-derived structure.
PDB reference: mouse major urinary protein, 1jv4
crystallization papers
Shikimate kinase from M. tuberculosis has been overexpressed and purified to homogeneity. Crystals of the enzyme in complex with MgADP diffract to 2.2 Å resolution.
The crystallization of antigen-binding fragments from two homologous monoclonal antibodies in the presence and absence of different carbohydrate haptens offers an opportunity to determine the sequence–structural basis of carbohydrate specificity.
Cytosolic Cu,Zn superoxide dismutase from S. mansoni has been crystallized in both orthorhombic and monoclinic space groups and data have been collected to 2.2 and 1.55 Å, respectively. Molecular-replacement solutions show that slight distortions to the crystal lattice relate one form to the other.
A complex of human calmodulin and adenylyl cyclase domain of edema factor, a pathogenic factor secreted by Bacillus anthrax, was crystallized in space groups I222, P3121 and P212121. The initial phases were obtained by selenomethionyl MAD using direct methods and improved by combining with two heavy-metal derivatives.
Crystallization and data collection are reported of a streptavidin crucial tryptophan mutation (Savm-W120K) that results in decreased affinity towards biotin and the dissociation of the tetramer into stable dimers.
Crystallization and preliminary X-ray analysis of glucose dehydrogenase from Haloferax mediterranei
Recombinant glucose dehydrogenase from the halophilic archaea H. mediterranei has been refolded, purified and crystallized. Two crystal forms have been obtained, one of which diffracts to greater than 2.0 Å resolution.
Transcobalamin is a cobalamin-binding protein present in mammalian plasma that facilitates the cellular uptake of vitamin B12. Crystals of human transcobalamin have been obtained and diffraction data have been collected to 3.2 Å resolution.
Crystals of Sso10b, an abundant non-specific double-stranded DNA-binding protein, from the hyperthermophilic archaeon S. solfataricus have been grown and preliminary X-ray data are reported.
The PAS domain of the NifL protein from A. vinelandii has been crystallized and X-ray diffraction data have been collected to 3.0 Å using synchrotron radiation. The native crystals belong to space group R32, with unit-cell parameters a = 65.0, c = 157.3 Å.
A haemoglobin-related protein composed of 109 residues (i.e. 30% shorter than conventional vertebrate haemoglobins) has been isolated from a nemertean worm neural tissue. Crystals of the recombinant protein allow location of the haem Fe atom in anomalous difference Patterson maps.
The heterodynamic periplasmic nitrate reductase from R. sphaeroides has been cloned, purified and crystallized. X-ray diffraction data have been collected to 3.1 Å resolution.
Two different liver basic fatty acid-binding proteins have been crystallized in forms suitable for X-ray diffraction studies. The crystals diffract to 2.0 and 2.5 Å resolution.
Crystals of the acetylxylan esterase from B. pumilus have been obtained by the hanging-drop method. The crystals diffract to 2.0 Å resolution and belong to space group R32.
Crystals of interleukin-10 in complex with the extracellular cytokine-binding domain of its high-affinity receptor have been obtained. A complete low-temperature native data set to 2.9 Å has been collected using synchrotron radiation.
The OXA-1 oxacillinase from E. coli has been crystallized from PEG at pH 7.5 in P1. Crystals diffract to 1.5 Å resolution and contain two monomers related by a pseudo twofold screw axis.
Maltose O-acetyltransferase has been crystallized in four crystal forms, including a trimethyl lead acetate cocrystal suitable for structure determination.
Crystallization conditions are described that allow recombinant obelin from O. longissima to form relatively large single light yellow crystals with excellent diffraction properties. A 1.1 Å data set has been collected and analysis is in progress.
The purification and crystallization of C. amalonaticus (2S,3S)-3-methylaspartic acid ammonia lyase is reported. Diffraction data have been collected to 2.1 and 1.5 Å, respectively, from two different crystal forms.
Crystals of a peptide corresponding to the first 669 residues of a 1320-residue multifunctional flavoprotein have been obtained and X-ray data have been collected to 2.15 Å resolution.
The C-terminal subdomain of rat 70 kDa heat-shock cognate protein has been cloned and expressed in E. coli. Preliminary crystallographic investigations have been carried out.
LCI, type of novel antibacterial polypeptide secreted by a Bacillus subtilis strain, has been expressed in E. coli DH5α with recombinant plasmid pBVAB16 and crystallized using PEG 4000 as a precipitant.
The N-terminal domain of E. coli Hsp100 ClpB has been crystallized. The crystal diffracted X-rays to 1.95 Å.
Crystallization and preliminary crystallographic analysis has been carried out on the most common polymorphic variants of the human intermediary metabolic enzyme alanine:glyoxylate aminotransferase, a deficiency of which causes the autosomal recessive disease primary hyperoxaluria type 1.
Aggregation of a membrane toxin was overcome by the use of the diamine buffer 1,3-diaminopropane. Glycerol inhibition of complex formation between protein and carbohydrate was shown to be insignificant in competitive binding assays conducted in the presence of glycerol prior to crystallization.
short communications
The acyl-homoserinelactone synthase EsaI has been crystallized in the space group P43, with unit-cell parameters a = b = 66.40, c = 47.33 Å. Multiple-wavelength anomalous diffraction using rhenium, a novel MAD atom, provided phases; the perrhenate-binding sites were identified in the model at 2.5 Å.
PDB reference: acyl-homoserinelactone synthase EsaI–rhenate complex, 1k4j
Anomalous scattering measurements reveal a K+ ion bound in proximity to the active site of the H. influenzae HslV protease.


 journal menu
journal menu