issue contents
February 2002 issue
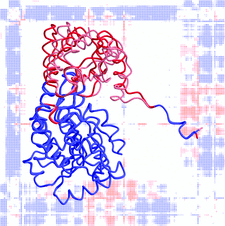
Cover illustration: Error-scaled difference distance matrix and least-squares superposition for the open and closed forms of aspartate aminotransferase. Conformationally invariant parts of the molecule are shown in blue, flexible parts in red (p. 195).
research papers
A genetic algorithm is described that divides a protein into rigid and flexible parts by automatic analysis of an ensemble of conformers.
The effect of osmotic shock on tetragonal lysozyme crystals is experimentally demonstrated. The network of channels and the charge of the crystal are responsible for a Gibbs–Donnan effect. This plays a critical role in the exchange of water, which leads to the formation of cracks.
The subtraction of the contribution of already positioned molecules from an observed Patterson map has proved to be useful for the determination of the correct orientation of the remaining molecules in the asymmetric unit of the crystal cell. For this purpose, a new procedure that incorporates a difference Patterson map in the evaluation of a vector-search rotation function has been developed.
The structure of equine apolactoferrin has been determined at 303 K. Both N and C lobes have been found in the closed conformations similar to those observed in various metal-saturated lactoferrin forms and in apolactoferrin at 277 K.
PDB reference: equine apolactoferrin, 1i6b
A second crystal structure of human mucosal addressin cell-adhesion molecule reveals that strand swapping can lead to dimer assembly and alters the conformation of the proposed integrin-binding surface.
PDB reference: MAdCAM-1, 1gsm, r1gsmsf
A method for calculating small-angle X-ray and neutron scattering from atomic detail models of solvated macromolecules is presented. The method also derives an analytical surface of the macromolecule which reproduces explicit-atom profiles well.
Conserved water molecules in the crystal structures of aspartic proteinases have been identified. Possible roles of these water molecules in the enzyme's structure and function have been analyzed.
The structural features of M. tuberculosis chaperonin-10 are reported and compared with the available chaperonin-10 structures. The structure also identifies binding sites for metal ions in the dome loop that have previously been shown to modulate the plastic states of M. tuberculosis chaperonin-10.
PDB reference: chaperonin-10, 1hx5
The structure determination and site-directed mutagenesis of the imipenemase SME-1 reveal the peculiar organization of its active site and the importance of the disulfide bridge found in the active-site cavity of the enzyme.
PDB reference: SME-1, 1dy6
A method which is able to locate fragments precisely over a wide range of resolutions for structure types from small organic molecules to proteins is described.
A set of quality standards that a fully refined protein structure should be able to meet is derived. Methods for detecting local structural errors are also evaluated and applied to a sample of recent structures from the Protein Data Bank.
crystallization papers
The preliminary X-ray crystallographic characterization of PelA from A. irakense is reported. This enzyme defines a new class of pectate lyases as it displays no homology to other bacterial, plant or fungal pectinases of known published structure.
The single-stranded DNA binding factor, PBF-2, is induced during pathogen infection of potato tubers and is implicated in defense gene regulation. Its DNA-binding component, p24, represents a conserved, ubiquitous family of plant-specific putative transcriptional regulators lacking strong similarity to proteins of known function.
Baculovirus P35 is a protein that prevents apoptosis in a diverse range of organisms. P35 stoichiometrically inhibits the apoptotic caspases in a cleavage-dependant mechanism that involves formation of a tight complex. An insect effector-caspase/P35 complex has been crystallized and its low-resolution structure is compared with the human initiator-caspase/P35 structure.
A soluble mutant (I6T/V47M/T69M) of the Rv2002 gene product (FabG3) of M. tuberculosis was crystallized. The X-ray diffraction quality of the crystal improved significantly after annealing/dehydration, enabling data collection to 1.8 Å resolution.
The use of substituted pentaerythritol compounds as precipitants and cryoprotectants for protein crystallography is described. These agents exhibit properties that lie between MPD and PEG and expand the repertoire of precipitants available for crystal growth.
The DNA decamers d(CCGGATCCGG) and d(CCGGCGCCGG) have been crystallized in order to investigate the effects of changing the two central base pairs of the DNA fragment d(CCGGGACCGG). Previous studies have already demonstrated that the structure of the former DNA fragment contains a DNA Holliday junction.
Crystals of the indoquinoline alkaloid cryptolepine complexed with the DNA fragment d(CCTAGG)2 have been grown in order to investigate a possible new type of intercalation between equal base pairs.
Human phenylethanolamine N-methyltransferase has been crystallized in a form suitable for structural studies. A major factor in obtaining crystals was the use of protein concentrations of greater than 90 mg ml−1.
E. coli B γ-glutamylcysteine synthetase has been crystallized. Native data have been collected to 2.8 Å resolution.
A 2.4 Å resolution data set was collected from a crystal of T. versicolor laccase. The initial electron-density maps indicate the presence of all four Cu atoms involved in electron transfer.
The homodimeric form of creatine amidinohydrolase from Actinobacillus has been crystallized by the hanging-drop vapour-diffusion method followed by macroseeding using PEG 6000 as a precipitant.
Two crystal forms of β-phosphoglucomutase have been obtained in space group P212121, with unit-cell parameters a = 53.67, b = 92.78, c = 111.60 and a = 53.21, b = 57.01, c = 76.11 Å. A complete three-wavelength MAD data set to 2.3 Å resolution has been collected from selenomethionine-substituted protein.
The single-stranded DNA-binding protein from M. tuberculosis has been crystallized in a trigonal form and an orthorhombic form. A mercury derivative of the trigonal crystals has been prepared.
Butyryl-ACP from E. coli, its selenomethionine (SeMet) variant and SeMet butyryl-ACP I62M have been crystallized. Crystals of the native protein and SeMet butyryl-ACP I62M belong to space group P212121 and diffract to beyond 1.4 Å, whereas those of native SeMet butyryl-ACP belong to space group P63 and diffract to at least 2 Å.
Crystals of peptide amidase from S. maltophilia, which selectively hydrolyses the C-terminal amide bond in peptide amides, have been obtained by sitting-drop vapour diffusion.
20K-cellulase, an endoglucanase from the thermophilic fungus M. albomyces, has been crystallized and full data were collected to 2.2 Å using synchrotron radiation. A methodology is described for growing single crystals of this endoglucanase.
The successful crystallographic analysis of glutathione amide reductase crystals is presented, leading to an initial structure solution by a molecular-replacement strategy. Even though glutathione amide reductase displays an altered substrate as well as cofactor specificity, it is seen to be a structural glutathione reductase homologue.
The membrane-bound NrfHA complex from W. succinogenes has been purified and crystallized in space group I422. MAD data has been collected at the iron Kα edge.
The molybdo-/iron–sulfur protein transhydroxylase was purified to homogeneity and crystals were grown which belong to space group P1. MAD data have been collected and structure solution is under way.
Dextranase has been expressed in P. pastoris with about 50% incorporation of selenomethionine. The enzyme has been crystallized in space group P21212, with unit-cell parameters a = 103.6, b = 115.3, c = 49.8 Å and one molecule per asymmetric unit. The crystals diffract to 2.0 Å.
The trehalose-6-phosphase synthase, OtsA, a UDP-sugar dependent glycosyltransferase from Escherichia coli, has been cloned, overexpressed and crystallized in two forms suitable for X-ray diffraction studies.
Aspartokinases catalyze the commitment step in the aspartate biosynthetic pathway. Aspartokinase III has been crystallized, in complex with MgADP and aspartate, and diffraction data collected.
The R1 plasmid-encoded toxic protein Kid from the parD system of E. coli has been overexpressed, purified, refolded and crystallized. Selenomethionine-incorporated crystals of Kid belong to space group P21, have unit-cell parameters a = 32.9, b = 45.0, c = 64.4 Å, β = 96.2° and diffract to a dmin of better than 1.8 Å on a synchrotron-radiation source.
Branching enzyme catalyzes the formation of the α-1,6 branch points in glycogen. This paper reports the crystallization and X-ray diffraction data collected to a resolution of 2.3 Å.
Two related crystal forms of an ABC-ATPase have been obtained in the absence of nucleotide. Experimental phases could be derived from single-wavelength anomalous diffraction data collected on an iodide derivative.
A combination of strategies leading to the successful structural determination of the catalytic domain of cytotoxic necrotizing factor 1 are presented. These strategies include successful expression of a partially soluble 33 kDa active CNF1 fragment, growth and optimization of crystals suitable for X-ray diffraction studies and determination of phases using a quadruple methionine-substitution mutant of the fragment.
Crystals of a recombinant form of CDP-D-glucose 4,6-dehydratase from Y. pseudotuberculosis have been grown in two crystal forms, both possessing pseudo-translational non-crystallographic symmetry.
Jack bean urease complexed with an antibody fragment has been crystallized, a partial 3.3 Å data set has been collected and a molecular-replacement solution for the urease has been determined.
short communications
The refined high-resolution structure of the D140N mutant of chitinase B from S. marcescens sheds light on the roles of Asp140 and Asp142 during catalysis.
PDB reference: chitinase B D140N mutant, 1goi


 journal menu
journal menu