issue contents
June 2003 issue
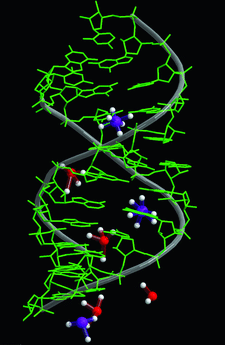
Cover illustration: An RNA dodecamer duplex containing symmetric loop from SRP domain IV demonstrates the interactions of hydrated Mg2+ ions (red) and Ca2+ ions (purple) (p. 1004).
research papers
Pseudo-rhombohedral packing led to a large asymmetric unit with three viruses and 180-fold non-crystallographic symmetry, which was used for phase determination, once the complicated rotation- and translation-function problems had been solved.
PDB reference: adeno-associated virus 2, 1lp3, r1lp3sf
The crystal structure of human phosphoserine phosphatase, an enzyme involved in the L-serine biosynthesis pathway, has been determined at a resolution of 1.53 Å. The structure presented here is in the open conformation and has a Ca2+ ion bound to the active site.
PDB reference: phosphoserine phosphatase, 1nnl, r1nnlsf
This structure of endothiapepsin complexed with the gem-diol inhibitor PD-135,040 has been refined anisotropically to 1.37 Å, allowing the identification of a number of short hydrogen bonds around the active site. The hydrogen-bonding pattern detailed is similar to that seen in atomic resolution structures of endothiapepsin co-complexed with single-hydroxyl inhibitors.
PDB reference: endothiapepsin–PD-135,040 complex, 1od1, r1od1sf
Comparative analysis of the structure of a myoglobin triple mutant determined from crystals grown in microgravity in two independent experiments on the Spacelab (Missions ISS 6A and ISS 8A) and on earth shows improved diffraction quality of the microgravity-grown crystals.
Four structures of C. cinereus peroxidase determined from crystals obtained under different physical conditions are described and used as a basis for an analysis of the structures at varying pH and temperature and with varying crystal packing. The structurally conserved water molecules have been identified and analysed.
The structure of a mutant of C. cinereus peroxidase showing altered thermostability and oxidative stability has been determined and structural explanations are provided for its altered properties.
PDB reference: thermostable mutant CiP, 1ly8, r1ly8sf
The crystal structure of the RNA dodecamer demonstrates that the symmetric loop in domain IV of SRP is greatly stabilized by hydrated metal ions. The structure also displays a novel RNA helical packing pattern of stacking at one end and abutting at the other.
PDB reference: 1lnt, r1lntsf
NDB reference: r(GCGUCAGGUCBrCG)/r(CGGAAGCAGBrCGC), AR0041
The structure of the lectin domain of a bacterial adhesin was determined using experimental phases derived from the anomalous dispersion signal of a selenium-containing analogue of a carbohydrate ligand.
PDB reference: F17a-G–β-Me-SeGlcNAc complex, 1o9v, r1o9vsf
A parallel-aware program MPI_FSEARCH has been implemented to perform molecular replacement using an exhaustive six-dimensional search.
Two proteins, xylanase of MW 32 kDa with five sulfurs and metal-free glucose isomerase of MW 44 kDa with eight sulfurs, were phased from the anomalous scattering signal of sulfur with a Bijvoet ratio of about 0.6%.
Crystallization trials of 456 T. maritima proteins have resulted in over 320 000 individual experiments. Analysis of this data shows that the number of initial screens can be reduced from 480 to 67 crystallization conditions without a significant impact on the number of proteins crystallized, that selenomethionine-containing proteins often crystallize in conditions which differ distinctly from those of their native counterparts and that the two-tiered shotgun method employed here is extremely useful for identifying those proteins which are likely to produce diffraction-quality crystals, thus increasing output.
crystallization papers
Atratoxin-b, a short-chain α-neurotoxin from mainland Chinese cobra venom has been purified and crystallized. Diffraction data to 1.56 Å resolution have been obtained at the X-ray Diffraction and Scattering Station of beamline U7B at the National Synchrotron Radiation Laboratory, Hefei, China.
PDB reference: atratoxin-b, 1onj, r1onjsf
Both the glutaredoxin from P. trichorpa in complex with glutathione and its selenomethionine derivative have been crystallized. A three-wavelength MAD data set was collected to a resolution of 1.73 Å, the positions of the Se atoms were identified and a high-quality electron-density map was obtained from the resulting phases.
Allergic reactions associated with house dust mites are an important source of morbidity in developed nations. A potent allergen from the dust mite D. farinae, Der f 2, has been crystallized in an effort to better understand the molecular basis of atopy.
Trigonal crystals of Langat virus E glycoprotein domain III grew in two distinct crystal forms from the same 1.8 M (NH4)2SO4, 0.1 M KOAc/NaOAc solution at pH 4.0. Native X-ray diffraction data were collected from the two forms to 2.9 and 3.35 Å resolution.
Monoclinic crystals of Jun a 1, the major allergen of pollen from the mountain cedar J. ashei, were grown from buffered ammonium acetate/PEG 4000 solutions. This is the first crystallization report of a cedar allergen and of a pectate lyase from a plant source.
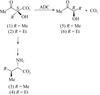
With the aim of understanding the mode of elongation of the side chain of peptidoglycan precursors by tRNA-dependent ligases in firmicutes, the FemX ligase from W. viridescens has been purified and crystallized. Native and SAD data have been collected to 1.7 and 2.1 Å, respectively.
The bacterial class B non-specific acid phosphatase AphA from E. coli has been crystallized. The enzyme is prone to polymorphism: three crystal forms in space groups I222 and C2 have been obtained. All crystals diffract X-rays to at least 2.0 Å.
A global regulator of gene expression in bacteria, Hfq protein, has been crystallized and X-ray diffraction data have been collected to 2.1 Å resolution.
p20, a thioredoxin-dependent thiol peroxidase from E. coli, has been crystallized. The crystal belongs to space group P212121, with unit-cell parameters a = 38.97, b = 58.97, c = 127.59 Å. A diffraction data set to 2.2 Å resolution has been collected.
To initiate the determination of the three-dimensional structure of a class A penicillin-binding protein, crystals of the transpeptidase domain of PBP1a from S. pneumoniae were prepared by limited proteolysis of the full-length molecule and purification by anion-exchange chromatography and gel filtration.
Crystals of the eukaryotic FeSOD from V. unguiculata have been obtained in a monoclinic form that diffract to 1.80 Å using synchrotron radiation.
Acetolactate decarboxylase has the unique ability to decarboxylate both enantiomers of acetolactate to give a single enantiomer of the decarboxylation product, (R)-acetoin. Three different crystal forms have been obtained from B. brevis, two of which diffract X-rays beyond 2 Å resolution and are suitable for structure determination.
The E. coli hypothetical protein TdcF has been crystallized in space group P21212. X-ray data were collected to 2.35 Å resolution.
AtCBL2, a calcineurin B-like protein from A. thaliana, was expressed in E. coli, purified and crystallized. A full set of X-ray diffraction data has been collected to 2.1 Å resolution using synchrotron radiation.
Three crystal forms are presented of the large subunit of a class Ib ribonucleotide reductase. The best diffracting crystals (tetragonal) were obtained after streak-seeding from one of the other crystal forms (orthorhombic).
An extracellular carbonic anhydrase from D. salina remains active over a broad range of salinities. Determination of the crystal structure using multiwavelength anamolous dispersion based on an intrinsic Zn ion at the active site is under progress.
This is the first report of diffraction data of the nucleoside hydrolases (NHs) from C. elegans and C. jejuni, representatives of mesozoa and bacteria, respectively. Their kinetic data suggest other functions and mechanisms than those of the well characterized protozoan NHs.
The replicative helicase loader protein G39P from the B. subtilis SPP1 phage has been purified and crystallized. Mass-spectrometric analysis has shown it to be very proteolytically sensitive and thus a truncated variant has been produced and crystallized that diffracts to higher resolution with improved reproducibility.
By switching from hanging-drop experiments to liquid–liquid diffusion in capillaries, well diffracting crystals could be obtained for leukotriene A4 hydrolase from S. cerevisiae.
The cloning, expression, purification and crystallization of the potential anti-parasitic drug target, purine nucleoside phosphorylase from S. mansoni, is described.
A method involving the evaporation of both water and acetic acid is used to crystallize isobutyrate kinase, a member of the ASKHA (acetate and sugar kinases/Hsc70/actin) superfamily of proteins.
Production, crystallization and preliminary crystallographic study of the major cat allergen Fel d 1
Fel d 1, the major cat (Felis domesticus) allergen, is one of the most important causes of allergic disease worldwide. Crystals of recombinant Fel d 1 have been obtained and diffraction data collected to 1.9 Å resolution.
The C-terminal part of C. elegans tropomodulin 1 was selected by homology and the recombinant protein was purified for crystallization. A nanoscreen produced the condition that led to crystals diffracting X-rays to 1.8 Å resolution.
GMP reductase 2 from human has been expressed in Escherichia coli, purified and crystallized. The crystals belong to space group P3221, with unit-cell parameters a = b = 110.6, c = 209.8 Å, α = β = 90, γ = 120°.
short communications
The crystal structure of human phosphoglucose isomerase complexed with the inhibitor 5-phosphoarabinonate has been determined to 2.5 Å resolution and helps to confirm the identity of Glu357 as the base catalyst in the isomerase mechanism.
The compatibility of detergents used to solubilize membrane proteins with the microbatch-under-oil crystallization technique was examined, specifically by testing whether significant quantities of detergent are lost owing to partitioning into the oil phase. For n-octyl-β-D-glucopyranoside and Fos-Choline-12, the loss of detergent is negligible.


 journal menu
journal menu