issue contents
April 2004 issue

Cover illustration: This birefringence orientation image, produced using the Metripol imaging system, illustrates the symmetry of the alignment of the acicular crystals around the centre of a lysozyme spherulite (p. 696).
research papers
Individual line defects, inclusions and other microdefects were observed in the phase-contrast X-ray diffraction images of a tetragonal lysozyme crystal which had a 4 4 0 rocking-curve width of ∼16.7 arcsec. It was revealed that dehydration led to a significant change in the microstructures and a nearly threefold increase in the width of the 4 4 0 rocking curve.
The phase transition of a triclinic lysozyme crystal was measured by X-ray diffraction and the structure of the transformed crystal was determined at 1.1 Å resolution.
This paper reports the crystal structure of the response regulator Spo0F in complex with manganese and explores the differing possibilities for the involvement of manganese and magnesium in the phosphoryl transfer.
PDB reference: metal-bound Spo0F, 1pey, r1peysf
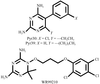
Structural data are reported for the first examples of the tetrahydroquinazoline antifolate (6R,6S)-2,4-diamino-6-(1-indolinomethyl)-5,6,7,8-tetrahydroquinazoline and its trimethoxy analogue (6R,6S)-2,4-diamino-6-(3′,4′,5′-trimethoxybenzyl)-5,6,7,8-tetrahydroquinazoline as inhibitor complexes with dihydrofolate reductase (DHFR) from human (hDHFR) and Pneumocystis carinii (pcDHFR) sources.
The crystal structure of the γδ T-cell ligand T10 was determined by molecular replacement using data from highly pseudo-merohedrally twinned monoclinic crystals that display pseudo-translation and non-crystallographic symmetry. The detection and treatment of twinning in T10 crystals and an analysis of the effect of the twinning on refinement is presented. A search of the PDB revealed several cases where possible twinning in monoclinic crystals may have gone unnoticed.
PDB reference: γδ T-cell ligand T10, 1r3h, r1r3hsf
The crystal structures of two lectins from the roots of pokeweed (P. americana) have been solved by molecular replacement. Structural features are observed in the relative arrangement of the carbohydrate-binding sites and the C-terminal regions.
A new crystal form of translin, an octameric human DNA/RNA-binding protein, has been solved at 2.2 Å resolution.
PDB reference: translin, 1j1j, r1j1jsf
Open  access
access
 access
accessA new structure of the duplex d(CGCAATTGCG)2 is presented. It contains Co2+ and diffracts to 1.6 Å. The results obtained are compared with four structures of the same duplex previously reported. The central hexamer sequence CAATTG has a practically constant conformation under all conditions, whereas the terminal base pairs differ in each case.
PDB reference: d(CGCAATTGCG), 1s23, r1s23sf
NDB reference: BD0066
The structure of the 115-amino-acid residue protein DsvC was determined by a combination of long-wavelength sulfur phasing and radiation-damage-induced phasing.
PDB reference: γ subunit of dissimilatory sulfite reductase, 1sau, r1sausf
Open  access
access
 access
accessAn automated birefringence technique for assessing the content of crystallization droplets, in particular precipitates, is described and discussed.
crystallization papers
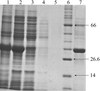
Glycolipid-transfer protein (GLTP) is a 24 kDa basic cytosolic protein that facilitates the transfer of glycolipids between bilayer membranes in vitro, but its in vivo function is unknown. Here, the crystallization and preliminary X-ray analysis of bovine GLTP are reported for the first time.
The anti-malaria vaccine candidate Pvs25 forms crystals that diffract to 2.3 Å and several complete data sets have been collected. The reductive methylation of lysine residues markedly improved the diffraction quality of the crystals.
Uridine phosphorylase from S. typhimurium has been crystallized. A preliminary X-ray diffraction data set indicates that the crystals belong to space group P61(5).
The human oncoprotein SET/TAF-1β has been crystallized by the sitting-drop method. A complete diffraction data set was collected to 2.8 Å resolution using synchrotron radiation.
Hyperthermostable NAD-dependent glutamate dehydrogenase from Pb. islandicum has been crystallized. The crystals diffract to a maximum resolution of 2.8 Å.
The common edible mushroom (A. bisporus) lectin has been crystallized in a form suitable for X-ray diffraction studies. The crystals diffract to 2.2 Å resolution.
Galectins are lectins involved in cell adhesion and growth regulation in animal cells. Cgl-1 and Cgl-2 of the fungus C. cinereus are the first family members from outside the animal kingdom. The gene design, expression, purification and crystallization of these two prototype galectins are described.
The hatching enzyme of medaka fish, high choriolytic enzyme has been crystallized by the hanging-drop vapour-diffusion method using PEG 10 000 as the precipitant.
Open  access
access
 access
accessKaiB and Cys-substituted KaiB mutants from the thermophilic cyanobacterium T. elongatus BP-1 have been expressed as GST-fusion proteins in Escherichia coli, purified and crystallized.
A human homologue of plant pathogenesis-related protein, which is specifically localized to the Golgi membranes, has been crystallized and crystals that diffract to 1.5 Å resolution are reported.
A conserved hypothetical protein from the hyperthermophilic crenarchaeon P. aerophilum has been expressed, purified and crystallized. Two methionine residues have been introduced by mutagenesis in order to provide SeMet-substituted crystals for MAD phasing.
Oligoribonuclease (Orn), an essential 3′-to-5′ exoribonuclease which degrades small oligoribonucleotides, has been crystallized under three different conditions.
The DH/PH tandem of PDZ-RhoGEF and C-terminally truncated RhoA have been overexpressed in Escherichia coli as TEV protease-cleavable fusion proteins containing GST and a hexahistidine tag at the N-termini, respectively. The nucleotide-free DH/PH–RhoA complex was purified by gel filtration and crystallized.
The NAD(H)-binding domain of the membrane protein transhydrogenase from E. coli has been crystallized. Crystals diffract to 1.9 Å resolution.
The E. faecalis cytolysin repressor CylR2 forms part of a new quorum-sensing system.
Type III polyketide synthase PKS18 from M. tuberculosis has been crystallized in order to understand its unusual starter molecule specificity.
Rap1GAP, the founding member of a family of GTPase-activating proteins for the small GTPase Rap1, has been overexpressed, purified and crystallized. Data collection from native crystals is reported.
The major cold-shock proteins of Bacillus subtilis and Bacillus caldolyticus are small oligonucleotide/oligosaccharide-binding fold proteins that have been described as binding single-stranded nucleic acids. Here they have been crystallized in the presence of the deoxyhexanucleotide (dT)6.
The crystallization and preliminary X-ray analysis of a soluble form of the lytic transglycosylase MltA that lacks the membrane anchor is presented.
Two crystal forms of 2-keto-3-deoxygluconate kinase from T. thermophilus have been obtained and characterized by X-ray diffraction.
Single crystals of the catalytic domain of death-associated protein kinase and its complexes with two inhibitor molecules have been prepared using the sitting-drop vapour-diffusion method. Both inhibitor molecules were clearly identified in the resulting difference Fourier map.
Crystals of asparagine synthetase A from the archaea P. abyssi, an enzyme structurally related to asparaginyl-tRNA synthetase, were grown by vapour diffusion; they belong to space groups C2 and P21 and diffract to 2.3 and 3.0 Å, respectively.
Open  access
access
 access
accessY75N mutant M. pusillus pepsin has been cocrystallized with human renin inhibitor and the crystals have been analysed.
Acyl carrier protein synthase from S. aureus was crystallized in two crystal forms. Complete data sets were collected to resolutions of 1.65 and 1.8 Å, respectively.
The tRNA pseudouridine synthase TruD from E. coli has been overexpressed, purified and crystallized. The orthorhombic crystals diffract to 2.4 Å resolution using synchrotron radiation.
Recombinant M. tuberculosis thioredoxin reductase has been expressed, purified and crystallized. Diffraction data were collected to 3 Å resolution and preliminary X-ray diffraction analysis is described.
Open  access
access
 access
accessWild-type and mutant (C59R+S108N) dihydrofolate reductase–thymidylate synthase (PfDHFR-TS) enzymes from P. falciparum complexed with NADPH, dUMP and either Pyr39 or Pyr30, pyrimethamine derivatives which retain binding affinities to the mutant enzyme, have been crystallized using the microbatch method. A preliminary study on the use of baby oil in microbatch crystallization is also discussed.
Open  access
access
 access
accessCo-crystallization of the third KH domain of human hnRNP K with a 15-mer ssDNA gave rod-shaped crystals belonging to the trigonal space group P3121 (unit-cell parameters a = 54.0, c = 149.7 Å) and diffracting to 2.4 Å resolution.
The purification, crystallization and X-ray diffraction analysis of NNA7, an anti-blood group Fab fragment, are described along with initial phasing information.
short communications
The crystal structure of bovine carbonic anhydrase II (BCA II) has been determined at 1.95 Å resolution by molecular replacement.
PDB reference: bovine carbonic anhydrase II, 1v9e, r1v9esf
Perturbing protein structural models in the direction of their lowest-frequency normal modes can make them favourable templates for difficult molecular-replacement problems.
addenda and errata
Free 

Free 

Free 



 journal menu
journal menu