issue contents
July 2004 issue
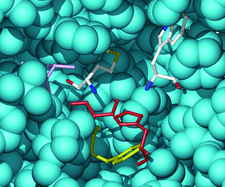
Cover illustration: The active site of P. fluorescens aryl esterase is formed by catalytic residues Ser94 and His251 (red) with the N atoms of Trp128 and Met95 (CPK colors) forming the oxyanion hole. Val121 (magenta) marks the acyl binding site and Phe93 (yellow) marks the binding site of the alcohol moiety of the substrate (p. 1237).
research papers
The structure of MPS is described in detail and compared with that of DDG aldolase.
PDB reference: M. commelinae MPS, 1izc, r1izcsf
Open  access
access
 access
accessThe structures of two crystal forms of ribonuclease Sa2 were determined at 1.8 and 1.5 Å. One form contains a complex of two molecules interacting through their active sites, in which the aromatic ring of a flipped-out active-site tyrosine residue of one molecule occupies the substrate-binding site of the second molecule.
The A. nidulans DNA apophotolyase structure has been determined at 1.8 Å resolution. The kinetics of in vitro reconstitution of the apoprotein have been determined and structural changes associated with X-ray-induced FAD reduction characterized.
PDB references: DNA photolyase, 1owl, r1owlsf; 1owm, r1owmsf; 1own, r1ownsf; 1owo, r1owosf; 1owp, r1owpsf
The PCM6 structure provides the first three-dimensional structure of a plant calmodulin and shows differences from animal and protozoa calmodulin structures in its crystal form, central helix conformation and molecular-surface properties.
PDB reference: potato calmodulin PCM6, 1rfj, r1rfjsf
Open  access
access
 access
accessA derivation of a maximum-likelihood single-wavelength anomalous diffraction phasing function with simple algorithmic characteristics for refinement of models containing anomalous scatterers.
Several types of search models prepared with alignments of different accuracy were compared in MR trials. The analysis shows, that models based on more accurate alignments have a higher success rate in MR.
The X-ray crystal structure of P. fluorescens aryl esterase has been solved to a resolution of 1.8 Å. It shows highest structural similarity to non-heme bacterial haloperoxidases, rather than to other esterases.
PDB reference: aryl esterase, 1va4, r1va4sf
Direct methods have been combined with the SOLVE/RESOLVE procedure to improve SAD phasing of proteins.
The local maxima of promolecular electron-density distributions are obtained at various degrees of smoothing for a protein structure. They are hierarchically related and are interpreted in terms of protein fragments.
structural genomics papers
The crystal structure of the putative DNA-binding protein SP_1288 from S. pyogenes has been determined by X-ray crystallography to a resolution of 2.3 Å using anomalous diffraction data at the Se peak wavelength.
PDB reference: SP_1288, 1s7o, r1s7osf
crystallization papers
Recombinant SOS3 from A. thaliana has been overexpressed in E. coli, purified and crystallized by the hanging-drop vapour-diffusion method. A preliminary X-ray data set has been collected at 3.2 Å resolution.
A new crystal form of a family 11 xylanase shows diffraction limits of better than 1 Å resolution.
The expression, refolding, purification and crystallization of mouse H-2Kk in complex with both an octapeptide from Influenza A virus and a nonapeptide from simian virus SV40 are reported.
A C-terminal fragment of TonB from E. coli consisting of amino-acid residues 147–239 (TonB-92) has been purified and crystallized.
E. coli K12 YgjK protein, a member of GH family 63, has been overexpressed, purified and crystallized using the vapour-diffusion method.
The crystallization and preliminary X-ray analysis of the α-L-arabinofuranosidase from A. kawachii, a family 54 glycoside hydrolase, is described. The crystals of recombinant protein expressed in P. pastoris belong to space group P212121 and diffracted to a resolution of 1.82 Å.
Xenobiotic reductase B from P. fluorescens I-C catalyzes the NADPH-dependent, nitrite elimination from nitroglycerin to yield 1,3-dinitroglycerol. Crystallization and analysis of the X-ray data for this FMN-containing enzyme is reported.
The human adrenal gland protein AD-004 has been expressed and purified from E. coli and crystallized in space group P61 or P65. The crystals diffracted to high resolution.
Insect-cell-expressed interleukin-22 has been crystallized using hanging-drop vapor-diffusion experiments at 310 K. A complete low-temperature 2.6 Å resolution data set has been collected using synchrotron radiation.
A recombinant peptide deformylase (PDF) of thermophilic bacterium T. thermophilus HB8 has been crystallized by the hanging-drop vapour-diffusion method using PEG 4000 as a precipitant. The crystals belonged to the tetragonal space group P41 or P43 with unit-cell parameters of a = b = 62.58, c = 105.27 Å.
Two truncated versions of the voltage-dependant calcium channel β2a subunit have been expressed in E. coli, purified and crystallized in two forms with orthorhombic and tetragonal symmetries.
The protein biosynthesis elongation factor 3 from yeast has been expressed in E. coli and yeast strains and well diffracting crystals have been obtained.
A thermophilic acylphosphatase from P. horikoshii was crystallized in space group P3221, with unit-cell parameters a = b = 85.65, c = 75.51 Å. A complete diffraction data set was collected to 1.6 Å.
Open  access
access
 access
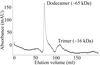
accessAnti-TRAP protein from B. subtilis was crystallized. The arrangement of peaks in the self-rotation function together with gel-filtration data suggest that the protein exists as a trimer or dodecamer in solution.
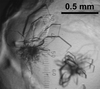
The 65 kDa trypsin-activated fragment of the active Cry4Aa mutant from B. thuringiensis subsp. israelensis, which is specifically toxic to mosquito larvae, has been purified and crystallized. The crystals belong to space group C2221 and diffract to 2.90 Å resolution.
The mitochondrial seryl-tRNA synthetase from B. taurus has been cocrystallized with the substrates ATP and serine using PEG 8000 and lithium sulfate as precipitants. The crystals, which belong to space group C2221, diffracted X-rays to a resolution of 1.6 Å.
The purification, crystallization and X-ray diffraction analysis of a Fab fragment of monoclonal neuroantibody αD11 against nerve growth factor are described. Initial phasing by Patterson search techniques and key features of the crystal-packing motif are discussed.
The dimerization domain of the Drosophila transcription factor responsible for sexual differentiation, Doublesex, has been expressed and crystallized; phases have been solved by single-wavelength anomalous dispersion.
Open  access
access
 access
accessTomatinase, a saponin-detoxifying enzyme belonging to family 3 of the glycosyl hydrolases from the fungal tomato pathogen S. lycopersici, has been purified from culture medium and crystallized. The crystals belong to space group P21212 are predicted to contain 46% solvent and diffract to at least 3.0 Å resolution.
short communications
The properties of ionic strength reducers are exploited in order to optimize protein purification and crystallization.
Anomalous diffraction data at the zinc absorption edge (λ = 1.2823 Å) were used to distinguish between zinc and calcium ions.
PDB reference: 3,4-dihydroxy-2-butanone 4-phosphate synthase, 1snn, r1snnsf
Open  access
access
 access
accessNative, bacterial-peptide-bound and heavy-atom derivative crystals of the 2F13F1 module pair of human fibronectin have been crystallized in a tetragonal space group (P4212, P41212 or P43212), with unit-cell parameters a = b = 37, c = 108 Å. Data were collected to high-resolution limits of between 2.2 and 1.7 Å. Six different tests were performed to test for possible twinning in the crystals.
addenda and errata
Free 



 journal menu
journal menu