issue contents
March 1995 issue
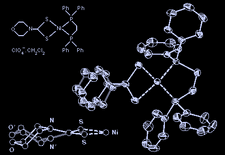
Cover illustration: [1,2-Bis(diphenylphosphino)ethane-P,P'](4-morpholine-carbodithioato-S,S')nickel({II}) Perchlorate Dichloromethane Solvate, see Akilan, Sivakumar, Venkatachalam, Ramalingam, Chinnakali & Fun, pages 368--370. The two different orientations of the morpholine ring, related by a rotation of 180°, are shown. The displacement ellipsoids are shown at the 30% probability level.
inorganic compounds










metal-organic compounds








































organic compounds














































































































 journal menu
journal menu
































































































