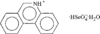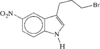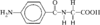issue contents
February 1999 issue
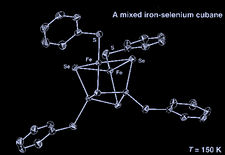
Cover illustration: Tris(tetraethylammonium) tetra-mu3-selenido-tetrakis[(thiophenolato-S)iron], see Hauptmann, Schneider, Chen & Henkel, pages 192-194. Displacement ellipsoids are shown at the 50% probability level.
cif-access (metal-organic compounds)
Download citation


Download citation


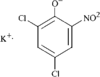
In the crystal structure of the the title compound, each K+ ion is coordinated to five planar 2,4-dichloro-6-nitrophenolate anions through the O atoms. All the O atoms are involved in the sixfold coordination of potassium.
Download citation


Download citation


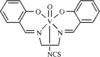
The title compound, [VO(salen)(NCS)], was prepared by the reaction of [VO(salen)]NO3 with KSCN. The structure was determined in order to observe the coordination of an isothiocyanate ion trans to an oxo O atom. There are two independent complex molecules in the crystal and they have similar distorted octahedral coordination geometries, with V—NCS distances of 2.218(6) and 2.300(5)Å.
cif-access (organic compounds)
Download citation


Download citation


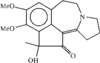
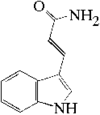
The single-crystal structure of the novel title compound, an analogue of the alkaloid hainanensine which has significant antitumor activity, was determined by X-ray structure analysis. nobreak Hydrogen bonds connect the molecules in pairs.
Download citation


Download citation


The crystal structure of phenathridinium hydrogenselenate monohydrate has been determined by single-crystal X-ray diffraction. The title compound was characterized using IR spectroscopy, powder diffraction and differential scanning calorimetry. No indication of unusual dielectric properties was found.
Download citation


Download citation


The structure of the title compound was determined previously by Jones & Templeton [Acta Cryst. (1958), 11, 484] at T = 278 K using the Weissenberg technique, and twice by Nahringbauer [Acta Chem. Scand. (1970), 24, 453-462] at T = 278 and 93 K. The present redetermination at T = 40 K provides data of higher precision, reflected in s.u.'s which are about one third those of Nahringbauer.
Download citation


Download citation


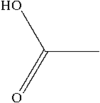
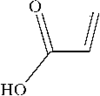
The title compound belongs to the family of α,β-unsaturated carboxylic acids having a Z conformation. The packing is characterized by layers of O—H⋯O hydrogen-bonded dimers.
Download citation


Download citation


The condensation reaction of 1,5-benzodiazepin-2-one with ethyl hydrazono-α-bromoglyoxylate in the presence of triethylamine is peri and regioselective, as indicated by the structures of the title cycloadducts. Moreover, these structures indicate that the dipole addition occurs in a unique way on the C=N double bond.
Download citation


Download citation


The absolute configuration of the title compound has been determined. The structure contains two independent molecules with very similar conformations. Torsion angles about corresponding bonds differ by less than 10°. The solvent molecule is not involved in short intermolecular contacts.
Download citation


Download citation


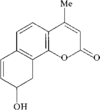
Coumarin derivatives are used as antifungal compounds, anticoagulants and laser dyes. In the title compound, the dihydrobenzene ring adopts a half-chair conformation and the hydroxyl group is involved in an O—H⋯O hydrogen bond. The inversion-related molecules are stacked 3.446 (4) Å apart along the c axis.
Download citation


Download citation


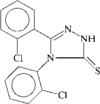
In the title compound, one of the two chlorophenyl rings is disordered with the two different orientations nearly related by a 180° rotation around its bond with the triazole ring. The planar triazole ring makes dihedral angles of 81.1(2) and 78.4(4)° with the two phenyl rings. Around the inversion centre, the molecules exist as an N—H⋯ hydrogen-bonded dimer.
Download citation


Download citation


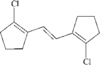
In the title compound, the C=C double bond exists in an E configuration. The cyclopentenyl ring adopts a flattened envelope conformation. In the solid state, the molecules are stacked along the a axis.
Download citation


Download citation


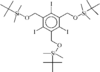
The title compound has two crystallographically independent molecules in the asymmetric unit. In molecule B, all three tert-butyldimethylsilyloxymethyl side chains are positioned on the same side of the aromatic ring plane, but only two of them are on the same side in molecule A.
Download citation


Download citation


3-(3-Bromopropyl)-5-nitro-1H-indole, C11H11BrN2O2, and 3-(3-hydroxypropyl)-5-nitro-1H-indole, C11H12N2O3, both have their propyl subunits in the synclinal–synclinal conformation. Bond distances to the terminal substituent on the propyl group are: Br—C 1.955(2) and O—C 1.428(2)Å.
inorganic compounds
Download citation


Download citation


Ammonium cobalt orthophosphate hydrate, a synthetic analogue of the ineral niahite, forms two-dimensional four-connected nets of [CoO6] octahedra sharing vertices. Each square mesh is capped by [PO4] tetrahedra sharing an edge and a vertex.
Download citation


Download citation


The structure of Ba3Mn2O8 has been refined using powder neutron diffraction. It consists of MnO43- tetrahedra separated by barium ions in six- and 12-fold coordination.
Download citation


Download citation


A synthetic route to single crystals of monoclinic NH4VO(NH3)PO4 has been found. The results of the single-crystal structure analysis are discussed.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu