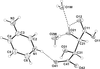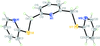issue contents
January 2010 issue
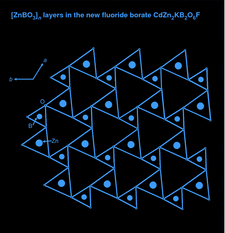
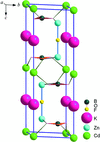
Cover illustration: A single [ZnBO3]n layer in CdZn2KB2O6F, viewed along the c axis, showing the BO3 (smaller) and ZnO3 (larger) triangles connected to each other via shared corners. See Zhang, Jiao, Shen, Shen & Wang [Acta Cryst. (2010), C66, i1-i3].
international union of crystallography
Free 

inorganic compounds
Download citation


Download citation


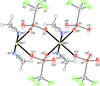
CdZn2KB2O6F consists of (001) layers with corner-sharing ZnO3F tetrahedra and BO3 triangles. The Cd atoms occupy six-coordinated interlayer sites and the K atoms are located in interlayer channels.
Download citation


Download citation


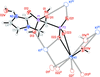
The cation disorder in the crystal structure of the mixed valent lithium/cobalt(II/III) phosphate Li4.03Co1.97(P2O7)2 is elucidated both from the analysis of the structural data and by means of the charge-distribution method which validate the proposed structural model.
Download citation


Download citation


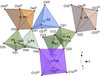
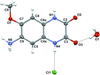
KBa7Mg2B14O28F5, with a nominal 7:1 composition of BaB2O4 to KMg2F5, is formed by isolated B7O14 groups and double perovskite units.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu