issue contents
January 2012 issue
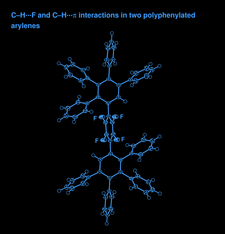
Cover illustration: Twice the asymmetric unit of 2',3',5',6'-tetrafluoro-2,2'',3,3'',4,4'',5,5''-octaphenyl-1,1':4',1''-terphenyl, with displacement ellipsoids at the 70% probability level. See Budy, Nichol & Loy [Acta Cryst. (2012), C68, o23-o27].
editorial
Free 

international union of crystallography
Free 

inorganic compounds
Download citation


Download citation


In the structure of PbCa2[Al8O15], AlO4 tetrahedra and AlO5 trigonal bipyramids in a 3:1 ratio constitute the framework [Al8O15]6− aluminate anion through corner- and edge-sharing. Pb2+ and Ca2+ cations exhibit coordination numbers of 7 and 6, respectively.
Download citation


Download citation


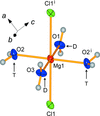
The structure of the title compound is built up from MgCl2(H2O)4 octahedra with a trans configuration. Each complex is situated on a crystallographic twofold axis, with the rotation axis aligned along one H2O—Mg—OH2 axis. They are connected by a complex network of O—H⋯Cl hydrogen bonds. The structure contains two-dimensional sections that are essentially identical to those in the reported tetrahydrates of CrCl2, FeCl2, FeBr2 and CoBr2, but they are stacked in a different manner in MgCl2·4H2O compared with the transition metal structures.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu