issue contents
September 1998 issue
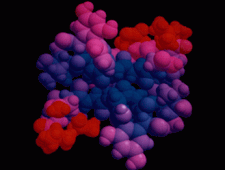
Cover illustration: The balhimycin dimer, with citrate and acetate ions in the binding pockets, colour coded on a scale where the most mobile atoms are red and the least mobile are blue. The binding pockets are clearly the most mobile, consistent with the idea that they can open and close to complex with cell-wall binding peptides. Courtesy of George Sheldrick.
research papers
The structure of the enzyme:inhibitor covalent adduct of factor D with 3,4-dichloroisocoumarin to a resolution of 1.8 Å is described. This represents the first structural analysis of factor D with a mechanism-based inhibitor.
Users of beamline X12-C at the National Synchrotron Light Source communicate with all the experimental apparatus, including both the data-collection equipment and the beamline components, through an easy-to-use graphical user interface (GUI). Important features of the system are modularity, ease of use, and automatic logging of experimental operations and parameters to facilitate nearly automatic data reduction.
A straightforward computer procedure is suggested to choose between several alternative solutions of the low-resolution phase problem.
A detailed description of the refined structure of E. coli dUTPase at 1.9 Å resolution shows a fold similar to other known structures of trimeric dUTPases, with a highly intertwined architecture and a disordered C-terminal arm. The use of optimized anomalous scattering measurements for the two heavy-atom derivatives led to a high quality initial electron-density map.
A modified phased translation function is applied to the location of small molecular fragments in noisy electron-density maps. Reciprocal-space implementation leads to speed improvements and some improved discrimination over real-space approaches.
The crystal structure has been determined at 2.0 Å resolution and detailed information on the two calcium binding sites has been obtained. This is essential for the elucidation of the specificity of tetranectin towards oligosaccharides.
Ultra high resolution structures of lysozyme reveal the variable nature of anion sites.
The 1.7 Å crystal structure of the non-psychrophidic cationic salmon trypsin comprise 4 molecules per asymmetric unit. The four independently refined molecules are compared to each other to the cold-adapted anionic salmon trypsin and the bovine trypsin, to search for the structural basis of the psychrophilic trypsin
A method is described for the determination of an anomalous scattering substructure using data at a single wavelength and the direct-methods program SnB.
The crystal structure of a fragment of fibritin, a predominantly coiled-coil structural protein from bacteriophage, has been determined to 1.85 Å resolution. Careful analysis of partial disorder associated with the crystal packing arrangement in the unusual space group P3 and appropriate modeling of the solvent structure were necessary for achieving good crystallographic refinement.
Strong information from self-rotation and Patterson functions is used to reduce the number of independent molecules to locate by molecular-replacement techniques.
Pseudo-translation symmetry in protein crystals of cyclophilin C and chorismate mutase and its use in structure determination is described.
This paper descibes the difficulties in the process of using the trial-and-error SYSTEM90 program to determine ab initio the structures of two small proteins App and rubredoxin with high-resolution data. Strategies to overcome the difficulties are discussed and the upgraded SYSTEM95 program was used successfully.
The X-ray structure of human lysozyme labelled with the 2′,3′-epoxypropyl β-glycoside of Man-β1,4-GlcNAc was determined by molecular replacement and refined to an R value of 0.168 at 2.1 Å resolution. Cell dimensions were a = 36.39, b = 116.38 and c = 30.91 Å (space group: P212121). The result explained the difference in molecular packing compared with wild-type human lysozyme and the origin of carbohydrate-residue recognition specificity at subsite B.
The chitobiose specific Ulex europaeus lectin II crystallizes in space group P3221 with unit-cell dimensions a = b = 105.54, c = 176.26 Å. The lectin forms a tetramer similar, but not identical to the PHA-L tetramer.
A three-parameter model is derived to describe the imperfections of protein crystals and their effect on rocking widths and diffracted-beam divergences. Measurements of frozen lysozyme crystals are presented in order to illustrate how the parameters describing crystal imperfection can be estimated.
The structure of glucoamylase from S. fibuligera has been solved at 1.7 Å resolution. Refinement from the initial molecular-replacement model involved the unrestrained automated refinement procedure (uARP) combined with the maximum-likelihood refinement program REFMAC. This is the first report of the three-dimensional structure of a yeast family 15 glucoamylase.
The growth of high-concentration fluctuations leads to phase separation and nucleation in supersaturated protein solutions. From NMR measurements of the induction time before crystallization, it can be concluded that the crystallization of lysozyme is a transient process and not a steady-state process.
A novel fiber-optic backscattering detector was employed to investigate nucleation in lysozyme solutions. The time evolution of the observed clusters can be employed in diagnosing conditions that favor protein crystallization.
The structure of the closed procapsid of the single-stranded DNA bacteriophage φX174 was determined to 3.5 Å resolution by molecular-replacement real-space averaging starting from a very poor model based on an electron microscopic reconstruction. Electron microscopy of thin-sectioned crystals was used to aid in the interpretation of the packing arrangement.
The structure of Ubc9, a sumo-conjugating enzyme, has been determined by molecular replacement to 2.8 Å resolution. The structure suggests how Ubc9 conjugates sumo rather than ubiquitin.
Using a difference Fourier map between tetragonal lysozyme bromide and chloride, five possible anion-binding sites have been identified.
A new software system for macromolecular structure determination is presented. The program covers nearly all aspects of macromolecular structure determination by X-ray crystallography and solution NMR.
X-ray topography and mosaicity measurements show that variations in solution conditions during protein crystal growth (an important feature of vapor diffusion and other popular growth methods) can produce crystal disorder. Several mechanisms may contribute to the observed disorder. These methods provide a very useful probe of protein crystal perfection and have been used to characterize the effects of variations in solution conditions during crystal growth on protein crystal perefection.
A crystal form of HIV-1 reverse transcriptase complexed to inhibitors which normally diffracts to 3.7 Å can be subjected to controlled dehydration to increase the diffraction limit to 2.2 Å. A range of different crystal forms have been identified and analysed, giving insight into the flexibility of this therapeutically important enzyme.
The structure of Ribonuclease Sa from Streptomyces aureofaciens has been solved in complex with the inhibitor barstar at 1.7 Å resolution. The nature of the interface of the complex is discussed and compared to that of the natural barnase–barstar system.
High-resolution X-ray data to 1.5 Å have been used to refine the crystal structures of dimeric and monomeric erabutoxin a. Significant protein discrete disorder and solvent order is described.
crystallization papers
The crystallization behavior of CcdB was investigated in detail. A novel crystal form, diffracting to 1.5 Å, proved to be suitable for structure determination.
Pig heart lipoamide dehydrogenase was crystallized and preliminarily X-ray analyzed by molecular replacement method.
Crystallization of DNA polymerase from the hyperthermophilic Archaeon Pyrococcus furiosus.
Bovine cytosolic brain-type creatine kinase hasd been crystallized. Diffraction data have been collected to 2.7 Å.
Crystallization and preliminary crystal analysis of cardosina, an aspartic proteinase found in flowers of the plant Cynara Cardunculus.
The hyperthermostable DNA polymerase (polymerase B family) from a marine Thermococcus archaeon has been crystallized, and a complete dataset was collected to 2.2 Å resolution. The structure of this enzyme will provide the first structural data on the archaeal and hyperthermostable classes of DNA polymerases.
The expression, purification, crystallization and preliminary X-ray characterization of the GlnK protein is described. Crystallization and preliminary X-ray characterization of the protein complexed with ATP is also described.
Crystallization of the hydrophilic ectodomain of the epidermal growth factor (EGF) receptor and low resolution X-ray stdies in combination with small angle X-ray diffraction and dynamic light-scattering techniques.
The thermophilic and thermostable family B DNA polymerase from the archaeon Sulfolobus solfataricus has been crystallized in the C2 space group with cell dimensions a = 187.4, b = 68.5, c = 125.8 Å and β = 107.8°. Crystals diffract up to 2.7 Å resolution.
The antigen-binding fragments (Fab) of one protective (13D9) and one non-protective (6B9) monoclonal antibody specific for the capsular polysaccharides of group B meningococci have been crystallized and undergone preliminary X-ray diffraction analysis. This is hoped to aid in the production of a vaccine for the disease meningococcal meningitis.
Portal proteins are essential in bacteriophage morphogenesis. Preliminary diffraction data of the crystallized portal protein gp6 from bacteriophage SPP1 confirmed its 13-fold symmetry.
Preliminary crystallographic studies of citrate synthase from an Antarctic psychrotolerant bacterium
Recombinant citrate synthase from a psychrotolerant bacterium, DS2-3R, recently isolated in Antarctica, has been crystallized. The crystals belong to space group P6122 or P6522, with cell dimensions a = b = 70.8, c = 307.8 Å. Knowledge of the structure of this enzyme will add to the understanding of cold activity and thermolability, and will be of biotechnological interest.
Pap1 is a fission yeast bZIP transcription factor that binds to non-consensus DNA sequences, TTACGTAA and TTAGTCA. Highly ordered crystals of Pap1-DNA complex that diffract up to 2.0 Å resolution have been obtained.
The water-soluble portion of rat heme oxygenase-1 has been expressed in E. coli and crystallized in the form of a complex with heme. The crystals belong to space group P41212 or P43212 and diffracted to better than 3.0 Å resolution.
Escherichia coli glutamate decarboxylase α and β have been expressed separately. Crystals of the latter (space group P31 or P32) have been used for a preliminary X-ray analysis at 2.5 Å resolution.
The article describes the first crystallization of a lectin bound to a synthetic multivalent ligand. Despite solution evidence that the protein is dimeric, a regular concanavalin A tetramer is found in the crystal with extensive contacts holding it together.
Rhamnogalacturonan acetylesterase from Aspergillus aculeatus has been crystallized in three different polymorphic modifications. Mass spectra showed that the ability to crystallize depends on the protein samples' degree of glycosylation. Native data sets have been collected to 1.55 Å for the orthorhombic and to 2.5 Å for the trigonal form.
Human serine hydroxymethlytransferase, a key enzyme in C1 metabolism and attractive anti-cancer target, has been overexpressed and successfully crystallized. The crystals diffract to at least 3 Å resolution and structure determination is under way.
Recombinant aspartate aminotransferase from an extreme thermophile has been crystallized in two different crystal forms. One form contains one dimer per asymmetric unit and diffracts to 2.1 Å resolution, while the other form contains two dimers and diffracts to 2.5 Å.
Crystals of the oxygenating subunit of 3,6-diketocamphane monooxygenase from Pseudomonas putida have been obtained using two different conditions.
short communications
Glutamate mutase was reconstituted with aquo- and cyanotobalamin, yielding inactive but stable enzyme, which was crystallized. One of its components was separately complexed with cofactor and crystallized.
The thermostable DNA binding protein HU from Thermotoga maritima has been purified and crystallized. A preliminary X-ray diffraction analysis has been reported.
Cycophilin A from the bovine parasite Trypanosoma brucei brucei had been cloned, expressed, purified and crystallized in the presence of cyclosporin A. The orthorombic crystals diffract to better than 2.1 Å.
The structure of an orthorhombic crystal form of pig plasma retinol-binding protein (RBP) has been determined at 1.65 Å resolution. New structural details in regard to the interaction of retinol with RBP and to the role of Cd2+ in promoting RBP crystallization are described.
A weakly bound inhibitor in the crystals of HIV-1 protease–inhibitor complex can be replaced by a more potent inhibitor. Structure of the resulting complex is similar to that obtained with the co-crystals of HIV-1 protease and the potent inhibitor.
Preliminary X-ray studies of two crystal forms of PNP from Cellulomonas sp.


 journal menu
journal menu






































































