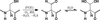issue contents
May 2007 issue
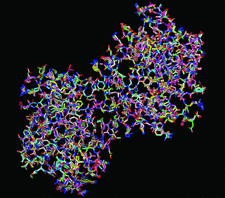
Cover illustration: The variation among 20 rebuilt versions of the structure of 1cqp [Kallen et al. (1999), J. Mol. Biol. 292, 1-9], obtained by automated iterative model-building, density modification and refinement (see p. 597), set a lower bound on the uncertainty in the coordinates of atoms in any one of the rebuilt models.
research papers
A comparison of the binding site of mexicain for E-64 with those of other E-64–cysteine protease complexes shows that highly conserved interactions are replaced by other interactions in which water molecules play an essential role.
PDB reference: mexicain–E-64 complex, 2bdz, r2bdzsf
Human hair was identified to be a versatile nucleant surface and was applied successfully for the crystallization of various proteins. Despite the rather complex mechanism of heterogeneous crystallization, it was discovered that the surface properties and the chemical composition together define the nucleation properties of the selected surface.
The vaccinia virus dUTPase structure shows an increased size of the central channel within the homotrimer.
PDB references: vaccinia virus dUTPase, 2okb, r2okbsf; 2okd, r2okdsf; 2oke, r2okesf; 2ol0, r2ol0sf; 2ol1, r2ol1sf
Arteriviruses form a family of enveloped, positive-sense RNA viruses that includes important agricultural pathogens such as EAV. The EAV nucleocapsid protein forms an arrangement of dimers in the crystal structure that may reflect its organization in the virus.
PDB reference: EAV N protein, 2i9f, r2i9fsf
The structure of a CBS-domain pair from human AMPK γ1 in complex with the physiological activator AMP and the pharmacological activator ZMP (AICAR) is presented.
Open  access
access
 access
accessHeterogeneity in ensembles generated by independent model rebuilding principally reflects the limitations of the data and of the model-building process rather than the diversity of structures in the crystal.
An evaluation of the geometrical restraints used in the refinement of protein structures is presented based on currently available protein and small-molecule structural databases.
The human formylglycine-generating enzyme was cocrystallized with bromide and iodide ions, which, among other positions, occupy an oxygen-binding pocket that is crucial for enzyme catalysis. Halide ions may substitute for xenon in the detection of oxygen-binding sites.
The crystal structure of a Rac1–GDP–Zn complex shows that Rac1 can coordinate zinc in a tetrahedral fashion by using residues in its biologically relevant switch I and switch II structural regions. Additionally, zinc can replace the normally coordinated magnesium without the perturbation of GDP from the small GTPase.
PDB reference: Rac1–GDP–Zn complex, 2p2l, r2p2lsf
A systematic study of the effect of substituting five different residues for large, solvent-exposed and flexible amino acids has been conducted using traditional and alternate reservoir screens. The results suggest a strategy that may enhance the success rates of preparation of X-ray quality crystals of proteins that are otherwise recalcitrant to crystallization in their wild-type form.
PDB references: RhoGDI CY mutant (K135Y,K138Y,K141Y), 2bxw, r2bxwsf; RhoGDI CH mutant (K135H,K138H,K141H), 2jhs, r2jhssf; RhoGDI CT mutant (K135T,K138T,K141T), 2jht, r2jhtsf; RhoGDI EA1 mutant (E154A,E155A), 2jhu, r2jhusf; RhoGDI EA2 mutant (E154A,E155A), 2jhv, r2jhvsf; RhoGDI FA mutant (E155A, E157A), 2jhw, r2jhwsf; RhoGDI FH1 mutant (E155H, E157H), 2jhx, r2jhxsf; RhoGDI FH2 mutant (E155H, E157H), 2jhy, r2jhysf; RhoGDI FS mutant (E155S, E157S), 2jhz, r2jhzsf; RhoGDI DY mutant (K138Y, K141Y), 2ji0, r2ji0sf
The crystallographic structure of the N114A mutant of the SH3 domain of the Abelson leukaemia virus tyrosine kinase complexed with a high-affinity peptide is reported. An X-ray diffraction data set was collected directly from crystals from the initial screening using the capillary counter-diffusion technique.
PDB reference: N114A Abl-SH32–p41 complex, 2o88, r2o88sf
It is shown that a high-pressure cryocooling method enables the phasing of macromolecular structures using either Xe or native S atoms. The method may even be used for crystals in capillaries.
essays
The origins of molecular and structural biology are briefly discussed in the context of the current status of the field. The future of structure biology is reviewed suggesting that in order to reach a new level of understanding of biological processes is necessary to include the notions of dissipative structures within the conceptual framework of non-equilibrium thermodynamics.


 journal menu
journal menu