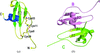issue contents
June 2008 issue
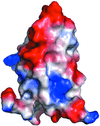
Cover illustration: The hybrid cluster protein from Desulfovibrio vulgaris showing the three domains (p. 665). The hybrid Fe-S cluster lies at the interface of the three domains in the centre of the picture, whilst the cubane Fe-S cluster is bound near the N-terminus of domain1 (coloured in green).
research papers
Open  access
access
 access
accessThe structure of mouse IP-10 shows a novel tetrameric association.
PDB reference: mouse IP-10, 2r3z, r2r3zsf
A method of automatically fitting nucleic acid double helices into electron density of high to medium resolution is presented and tested.
The crystal structure of M. tuberculosis phosphoribosyl-ATP pyrophosphohydrolase, the second enzyme in the histidine-biosynthetic pathway, is presented. The structural and inferred functional relationships between M. tuberculosis phosphoribosyl-ATP pyrophosphohydrolase and other members of the nucleoside-triphosphate pyrophosphatase-fold family are described.
The structure of the Fab fragment f3p4 selected from the HuCAL GOLD library is reported.
PDB reference: Fab fragment f3p4, 2v7n, r2v7nsf
The crystal structure of the enzymatically active domain of Ply500 has been solved at 1.8 Å resolution.
PDB reference: enzymatically active domain of Ply500, 2vo9, r2vo9sf
The crystal structure of human α-enolase (hENO1) has been determined for the first time and discloses distinct surface properties despite its high sequence similarities to other enolases. The surface properties provide an insight into its unique activities, including plasmin(ogen) and DNA binding, which are closely related to its roles in numerous diseases such as metastatic cancer, autoimmune disorders, ischaemia and bacterial infection.
PDB reference: human α-enolase, 3b97, r3b97sf
An evaluation is made of the protonation states of surface Asp and Glu residues in the C1 domain of cMyBP-C at 1.3 Å resolution. Extrapolations are made of the X-ray diffraction resolution required and/or of the specimen volume required for neutron protein crystallography to determine such states precisely.
PDB reference: cMyBP-C C1 domain, 3cx2, r3cx2sf
Hybrid cluster proteins (HCP), whose function has yet to be determined, possess a similar fold to carbon monoxide dehydrogenases which also have a related metal–sulfur cluster at the equivalent hybrid cluster position. In HCP the precise nature of the hybrid cluster appears to be dependent on the absence or presence of oxygen during the purification and crystallization procedures.
PDB reference: hybrid cluster protein, 1w9m, r1w9msf
The crystal structures of apo histidinol-phosphate aminotransferase from C. glutamicum, of the internal PLP aldimine adduct and of a pyridoxamine 5-phosphate–enzyme complex were determined at resolutions of 2.2, 2.1 and 1.8 Å, respectively. The residues lining the substrate-binding pocket were studied by site-directed mutagenesis.
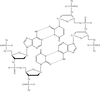
Microporous molecular sieves were used as a hetero-epitaxic nucleant for protein crystallization.
short communications
The crystal structure of an archaeal alanine:glyoxylate aminotransferase which has an amino-acid sequence that shows no similarity to any other known alanine:glyoxylate aminotransferases was determined.
PDB reference: alanine:glyoxylate aminotransferase, 2zc0, r2zc0sf
Open  access
access
 access
accessHuman Fc fragments containing the L234F/L235E/P331S triple mutation exhibit a dramatic decrease in their binding to several effector molecules (CD64, CD32A, CD16 and C1q). The three-dimensional structure of such a mutated fragment reveals that these broad-ranging functional effects are not caused by major structural rearrangements in the Fc moiety.
PDB reference: human Fc fragment, 3c2s, r3c2ssf


 journal menu
journal menu