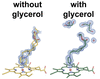issue contents
January 2023 issue
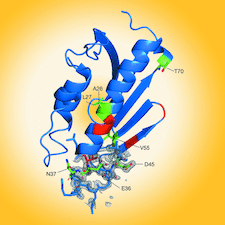
Cover illustration: Frataxin is a key protein in the biosynthesis of iron–sulfur clusters in mitochondria. The hereditary cardiodegenerative and neurodegenerative disease Friedreich's ataxia arises from either an inability to produce sufficient quantities or the production of a nonfunctional form of this protein. Rodrigues et al. [(2023), Acta Cryst. D79, 22–30] have studied frataxin from Drosophila melanogaster using both X-ray crystallography and NMR spectroscopy.
research papers
Open  access
access
 access
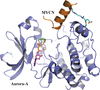
accessThe crystal structure of a covalently linked Aurora-A–MYCN complex is reported, enabling drug design and screening studies.
PDB reference: covalently linked Aurora-A–MYCN complex, 7ztl
Open  access
access
 access
accessA new protocol, DAQ-refine, for evaluating a protein model built from a cryo-EM map and applying local structure refinement is described.
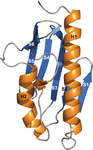
The structure of Drosophila melanogaster frataxin (Dfh) was derived by X-ray crystallography and studied by NMR spectroscopy. The crystal and solution structures are extremely similar. NMR spectroscopy was also used to identify the iron-binding location on helix 1 and strand 1 of Dfh.
PDB reference: Drosophila melanogaster frataxin, 7n9i
A scoring function has been developed for the prediction of protein complex interfaces based on neighboring amino acids.
Open  access
access
 access
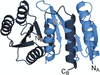
accessThe crystal structures of two permuted halves of a ribose-binding protein from Thermotoga maritima provide insights into the evolution of the periplasmic binding protein fold from a flavodoxin-like precursor by duplication and domain swapping.
Open  access
access
 access
accessAtomistic simulations enhance protein crystallography, yielding mechanistic insights into a protein kinase involved in the regulation of fundamental biological processes that include metabolism, development, memory and immune response.
This work shows that glycerol may compromise functional assays and crystallographic studies of pharmacological protein targets, consequently lowering the success rate of drug-discovery campaigns.
Open  access
access
 access
accessKey physical principles and methods for collecting crystallographic data from biomolecular systems at room temperature and, more generally, at temperatures between ∼200 and ∼350 K are reviewed and discussed.

 journal menu
journal menu