issue contents
October 2015 issue
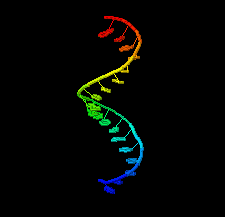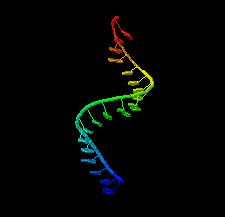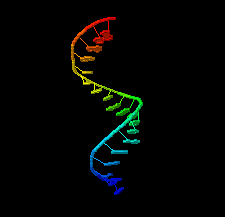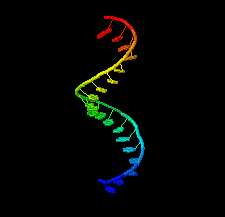
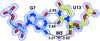
Cover illustration: 1.25 Å resolution structure of an RNA 20-mer that binds to the TREX2 complex (Valkov & Stewart, p. 1318).
research communications
Download citation


Download citation


The anthracycline biosynthetic enzyme DnmZ has been structurally characterized in ligand-free and thymidine diphosphate-bound forms.
The crystal structure of the response regulator RPA3017 from the photosynthetic bacterium R. palustris has been determined at 1.9 Å resolution. RPA3017 is part of a two-component system that involves two tandem bacteriophytochromes involved in red-light signaling.
Csm2 is a major component of type IIIA CRISPR ribonucleoprotein complexes that confer prokaryotes with immunity against phages and plasmids via an RNA-guided interference mechanism. The Csm2 protein from T. maritima was recombinantly expressed, purified and crystallized, and its structure was solved via cadmium single-wavelength anomalous diffraction phasing.
Open  access
access
 access
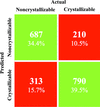
accessIn a large-scale study using data from the Protein Data Bank, some of the many reported findings regarding the crystallization of proteins were investigated.
KDM5B is an important new putative cancer-therapy target. Here, the binding properties and crystal structure of a nanobody raised against KDM5B are described.
PDB reference: NB17, 4zg1
The antibacterial compound QPT-1, the antibacterial drug moxifloxacin and the anticancer drug etoposide were each co-crystallized with DNA and S. aureus DNA gyrase. The base pairs at the ends of the DNA duplexes were modified to optimize the crystal contacts and to give diffraction data to between 2.45 and 3.15 Å resolution from six DNA-complex crystals.
RAB11(S20V), a constitutively active GTP-binding form of RAB11, was purified and crystallized. Crystals were obtained at 293 K and diffracted to a resolution of 2.4 Å; they belonged to space group I4, with unit-cell parameters a = 74.11, b = 74.11, c = 149.44 Å.
The crystal structure of the HECT domain of WWP2, an E3 ubiquitin–protein ligase, has been determined at 2.5 Å resolution and revealed a compact inverted T-shaped conformation.
PDB reference: HECT domain of human WWP2, 4y07
The resolution of the structure of 2,4′-dihydroxyacetophenone dioxygenase has been extended to 1.88 Å and it has been demonstrated by gel filtration and analytical ultracentrifugation that in situ chymotrypsinolysis, which is required for crystallization, converts the enzyme from a tetrameric form to a dimeric form. A putative model of the tetramer obtained by symmetric docking studies closely resembles the tetrameric assembly of the homologous RmlC-like cupin protein from R. eutropha.
PDB reference: 2,4′-dihydroxyacetophenone dioxygenase, 5bpx
Cellotetraose binding and cleavage by GH48 ExgS (glycosyl hydrolase 48 cellobiohydrolase S).
The structure of the death receptor 4–TNF-related apoptosis-inducing ligand (DR4–TRAIL) complex is presented and compared with those of DR5–TRAIL complexes. Isothermal titration calorimetry data are presented that demonstrate differences in the interactions of DR4 and DR5 with TRAIL.
PDB reference: DR4–TRAIL complex, 5cir
Frutalin, an α-D-galactose-binding lectin of biomedical interest from A. incisa seeds, has been crystallized and its preliminary X-ray diffraction analysis at 1.81 Å resolution is reported.
Crystallization and preliminary data collection to 2.2 Å resolution is reported for the C1 protein of Salmonella phage P22 in complex with a 23-mer DNA.
Open  access
access
 access
accessCitrate synthase from the thermophilic euryarchaeon T. acidophilum fused to a hexahistidine tag was purified and biochemically characterized. The structure of the unliganded enzyme at 2.2 Å resolution contains tail–active site contacts in half of the active sites.
PDB reference: citrate synthase, 4ybo
The X-ray crystal structure of the enteropathogenic E. coli gatekeeper protein SepL contains three X-bundle domains.
PDB reference: SepL, 5c9e
Open  access
access
 access
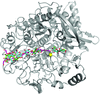
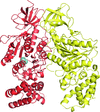
accessThe structure of the αβ heterodimer of the F-ATPase from the α-proteobacterium P. denitrificans has been determined at 2.3 Å resolution. It corresponds to the `open' or `empty' catalytic interface found in other F-ATPases.
Open  access
access
 access
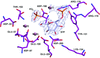
accessThe 1.25 Å resolution crystal structure of a 20 nt ribonucleotide that binds to the TREX2 complex with high affinity shows a double-stranded RNA duplex that contains C–A, U–U and C–C noncanonical base pairings together with canonical Watson–Crick A–U and G–C pairs and a G–U wobble.
PDB reference: RNA 20-mer, 5c5w
The response regulator NsrR from S. agalactiae, which is involved in nisin resistance, was crystallized, yielding crystals that diffracted to 1.4 Å resolution.
Open  access
access
 access
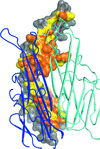
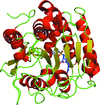
accessThe crystal structure of L. salivarius transketolase has been determined to high resolution in the presence and absence of thiamine pyrophosphate. The structures are presented with a brief comparison with other known transketolase structures.
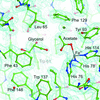
The crystal structure of the complex of porcine carboxypeptidase B with an arginine transition-state analogue is reported for the first time.
Open  access
access
 access
accessThe X-ray crystal structure of the dual-function fructose-1,6/sedoheptulose-1,7-bisphosphatase has been solved to 2.3 Å resolution in the presence of sedoheptulose-7-phosphate and the absence of adenosine monophosphate.
Crystals of porcine pancreatic elastase were cross-linked with glutaraldehyde, soaked with a novel inhibitor and characterized. This method enabled the soaking of a hydrophobic ligand into the protein crystals.
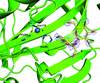
An engineered human carbonic anhydrase IX (CA IX) mimic was co-crystallized with the disaccharide sucrose. Its binding provides a novel `interaction' site that can be used to design targeted inhibitors of CA IX, a key marker of hypoxic tumors.
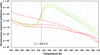
Over 250 proteins have been subjected to a standard dye-based thermal melt analysis, and the results suggest that at least a third of soluble protein samples can be made significantly more stable by changing the composition of their formulation.


 journal menu
journal menu

















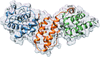



![[publBio]](/logos/publbio.gif)





