issue contents
December 2015 issue
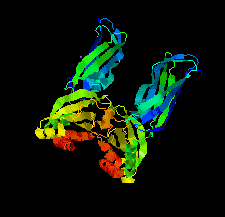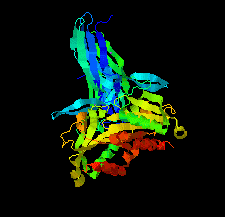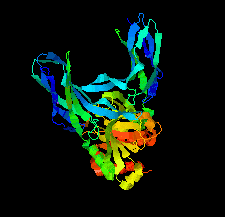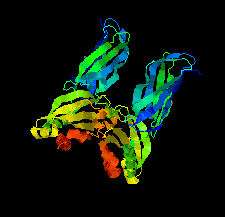
Cover illustration: Structure of Spo0M, a sporulation-control protein from Bacillus subtilis (Sonoda et al., p. 1488).
editorial
Free 

research communications
The C. trachomatis protease CT441 has been recombinantly produced and crystallized, and a diffraction data set has been collected to a resolution of 2.95 Å.
Diffraction-quality crystals of the ZnuA domain of pneumococcal AdcA were obtained using dried seaweed powder as a heterogeneous nucleating agent. X-ray diffraction data were collected to 2.03 Å resolution.
Open  access
access
 access
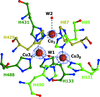
accessThe restoration of the native form of laccase from B. aclada from the type 2 copper-depleted form of the enzyme was investigated. Copper ions were found to be incorporated into the active site after soaking the depleted enzyme in a Cu+-containing solution.
PDB reference: Botrytis aclada laccase, 4x4k
Olivetolic acid cyclase from C. sativa L. has been overexpressed in E. coli, purified and crystallized. Diffraction data have been collected to a resolution of 1.40 Å.
Wild-type and an F34M+S405N variant of glyceraldehyde dehydrogenase from T. acidophilum were crystallized and X-ray diffraction data were collected for both enzyme variants to resolutions of 1.95 and 2.10 Å, respectively.
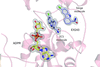
The crystal structure determination of a CRM1–RanGTP–SPN1–Nup214 quaternary nuclear export complex is reported. The process included protein-construct optimization, seeding, PEG-mediated crystal dehydration and additional post-mounting steps using an HC1c crystal humidifier.
The crystal structure of Spo0M was determined and the structure of the N-terminal half of Spo0M was compared with other proteins belonging to the arrestin clan. The FP domain of PI31 was also identified as a potential homologue of the C-terminal half of the Spo0M protein.
PDB reference: Spo0M, 5cl2
Open  access
access
 access
accessIn the present study, microseed matrix seeding was successfully applied to obtain a large number of crystals of the human sirtuin isotypes Sirt2 and Sirt3. These crystals appeared predictably in diverse crystallization conditions, diffracted to a higher resolution than reported in the literature and were subsequently used to study the protein–ligand interactions of two indole inhibitors.
The recombinant expression, purification, crystallization and X-ray crystallographic analysis of AerE from M. aeruginosa, an enzyme belonging to the cupin superfamily which has been deduced to participate in the biosynthesis of the 2-carboxy-6-hydroxyoctahydroindole moiety of aeruginosin, are described.
The catalytic domain of ChiL, a chitinase from the chitin-degrading bacterium C. shinanonensis, was expressed, purified and crystallized. Preliminary X-ray analysis shows that the crystal belonged to the orthorhombic space group P212121 and diffracted to 1.25 Å resolution.
Crystallographic studies show that diazaborines lacking a sulfonyl group can successfully inhibit E. coli enoyl-ACP reductase.
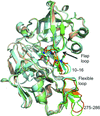
The crystal structures described here demonstrate the conformational flexibility of the active-site cavity of the plasmepsins. The interactions of the different moieties in the P1′ position of the inhibitors have to be taken into account in the design of new potent plasmepsin inhibitors.
addenda and errata
Free 

A correction is made to the article by Liu et al. [(2015), Acta Cryst. F71, 1063–1066].


 journal menu
journal menu


















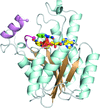

![[publBio]](/logos/publbio.gif)






