issue contents
May 2020 issue
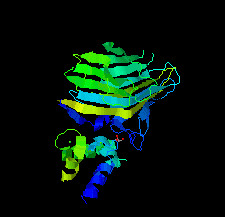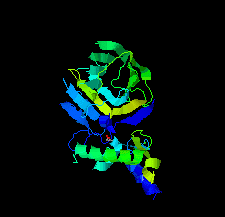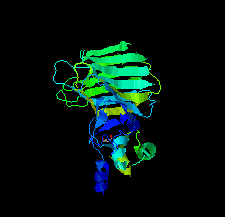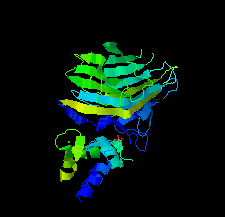
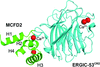
Cover illustration: The EF-hand protein MCFD2 complexed with the intracellular lectin ERGIC-53 involved in glycoprotein transport [Satoh et al. (2020), Acta Cryst. F76, 216-221]. The transmembrane intracellular lectin ER-Golgi intermediate compartment protein 53 (ERGIC-53) and the soluble EF-hand multiple coagulation factor deficiency protein 2 (MCFD2) form a complex that functions as a cargo receptor, trafficking various glycoproteins between the endoplasmic reticulum and the Golgi apparatus. Crystal structures of ERGIC-53CRD-MCFD2 complexes reveal that MCFD2, but not ERGIC-53-CRD, exhibits significant conformational plasticity that may be relevant to its accommodation of various polypeptide ligands.
research communications
 access
access

 journal menu
journal menu
















![[publBio]](/logos/publbio.gif)





