issue contents
December 2022 issue
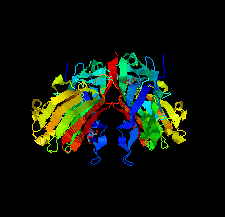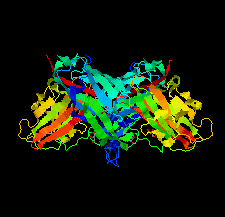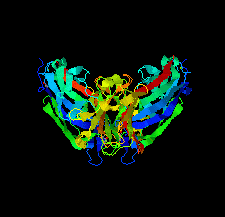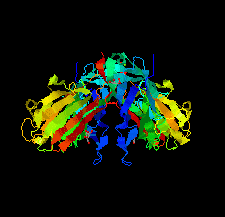
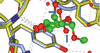
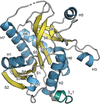
Cover illustration: The purine nucleoside phosphorylase from Geobacillus stearothermophilus is a thermostable protein of potential interest for the biocatalytic synthesis of antiviral nucleoside compounds. The structure of the protein was determined and found to include a His tag in the active site [Given et al. (2022), Acta Cryst. F78, 416–422]. This study shows that while tags are useful in the purification of proteins, we need to ensure that they do not have unwanted effects on protein structure and function.
scientific comment
 access
accessresearch communications
 access
access
 journal menu
journal menu













![[publBio]](/logos/publbio.gif)





