issue contents
March 2014 issue

editorial
BIOLOGY | MEDICINE
Biological crystallography has never been more vibrant. Celebrating this, IUCrJ invites high-impact papers from across the whole spectrum of structural biology and medicine.
scientific commentaries
BIOLOGY | MEDICINE
A brief history is given of how X-ray diffraction data from crystals have been recorded. Today there are new possibilities, spawned by the availability of free electron lasers that produce powerful femtosecond long X-ray pulses.
research papers
BIOLOGY | MEDICINE
The structure solution of T. brucei cathepsin B from 80 in vivo grown crystals with an average volume of 9 µm3 obtained by serial synchrotron crystallography at a microfocus beamline is reported.
PDB reference: cathepsin B, 4n4z
BIOLOGY | MEDICINE
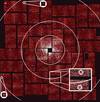
Bragg diffraction achieved from two-dimensional protein crystals using femtosecond X-ray laser snapshots is presented.
BIOLOGY | MEDICINE
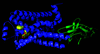
The plant purple acid phosphatase PPD1 forms a novel hexameric structure with a fibronectin III-like domain that is involved in DNA selectivity, binding and activation. The degradation of DNA by PPD1 implies a role for PPD1 in plant growth and repair and in pathogen defence.
PDB reference: purple acid phosphatase, 3zk4
CHEMISTRY | CRYSTENG
Download citation


Download citation


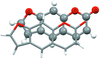
Multi-temperature single-crystal and powder diffraction experiments on 1-(2′-aminophenyl)-2-methyl-4-nitroimidazole show that this crystal undergoes an isomorphic phase transition with the coexistence of two phase domains over a wide temperature range. The anharmonic approach was the only way to model the resulting disorder.
CHEMISTRY | CRYSTENG
An enrichment ratio is derived from the decomposition of the crystal contact surface between pairs of interacting chemical species. The propensity of different contact types to form is investigated.
BIOLOGY | MEDICINE
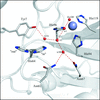
Human carbonic anhydrases are zinc metalloenzymes that catalyze the hydration and dehydration of CO2 and HCO3−, respectively. X-ray crystal structures of a variant of human carbonic anhydrase II in complex with four imidazole derivatives (imidazole, 1-methylimidazole, 2-methylimidazole and 4-methylimidazole) have been determined in order to identify the binding sites for such compounds, and a mechanism to explain the effects on catalytic activity is proposed.
CHEMISTRY | CRYSTENG
Download citation


Download citation


Multi-component crystals of the anti-inflammatory drug acemetacin were prepared by melt crystallization and their X-ray crystal structures solved using single-crystal and high-resolution powder X-ray diffraction (PXRD) data. The acemetacin–para-aminobenzoic acid adduct and the acemetacin piperazine salt are stable to hydration in the aqueous medium (up to 24 h).



 journal menu
journal menu




 access
access