issue contents
January 2003 issue
Special issue on the Contribution of BioXAS to Structural Genomics
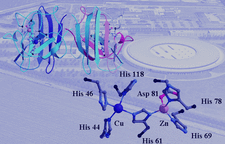
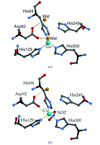
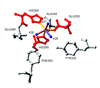
Cover illustration: Ribbon diagram and a close-up of the metal sites and the bridging His61 of CuZn bovine superoxide dismutase (see Hasnain & Strange, pages 9-15), and an artist's impression of SOLEIL, Saint Aubin, France (courtesy Chaix & Morel and associates).
facility information
research papers
Free 

An introduction to X-ray absorption spectroscopy and structural genomics.
Current results of a canonical structural genomics programme on yeast are presented. Contributions to the structural genomics of several structure-determination methods are discussed.
High-resolution X-ray crystallography and XAFS are a powerful combination for structure–function studies of metalloproteins, which make up a significant (∼30%) proportion of the known genomes.
Technical requirements and typical set-ups for both simple and complex biological XAS experiments are summarized.
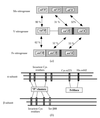
An overview of the activities of the Collaborative Computational Project Number 4 in protein crystallography is presented. Ideas and examples are given that may be applicable to the biological XAS community.
This paper aims at providing the theoretical background for full spectral fitting of the X-ray near-edge absorption energy region and beyond for the purpose of structural analysis, with an eye to biological applications.
Recent developments in the analysis and interpretation of EXAFS and XANES are reviewed.
The GNXAS method for multiple-scattering data analysis of EXAFS data and results recently obtained on molecular complexes relevant to biological matter are briefly reviewed and discussed.
This paper presents a new software procedure able to perform structural fits in the XANES energy range. The method is presented in detail with some applications: the metal K-edge of metal ions in water solution and the iron K-edge in the carbonmonoxy-myoglobin single crystal.
Overview of the biological applications of X-ray absorption near-edge structure at very low energy and presentation of a new method of calculating the spectra potentially applicable to metalloproteins.
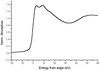
The influence of bond length and coordination number on simulated XANES spectra is studied and compared with experimental data for enzymes of the 2-His-1-carboxylate family and the metallo β-lactamase family.
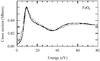
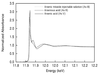
Third-generation synchrotron X-ray sources allow high-quality XAS data on biological samples to be obtained, where the metal concentration is in the 100 mM range.
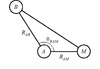
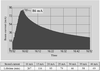
Crystallographic models of metal centres in metalloproteins at less than atomic resolution can be improved by applying EXAFS-derived distance restraints during the early stages of refinement. This is demonstrated for the Mo coordination of the FeMoco cluster of Klebsiella pneumoniae nitrogenase.
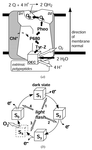
X-ray absorption spectroscopy facilitates detailed investigations on biological catalysis. The current status and perspectives of research on structural and oxidation-state changes at the metal site of biological metalloenzymes are discussed using water oxidation at the manganese complex of photosynthesis as an example.
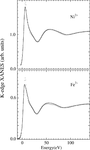
X-ray absorption spectroscopy, including extended X-ray absorption fine structure and X-ray absorption near-edge structure analysis, has been carried out at the Zn K edge of the N-terminal part of the integrase protein of the human immunodeficiency virus, type 2 (HIV-2), and of some zinc coordination compounds.
Various uses of X-ray absorption spectroscopy and imaging in pharmaceutical science are presented through a number of examples focused on low-molecular-mass species.
current events
Free 

Free 

international union of crystallography
Free 



 journal menu
journal menu