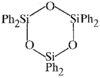
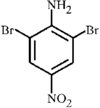
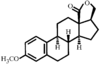
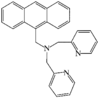
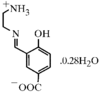
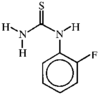
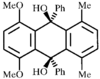
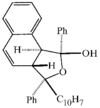
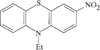
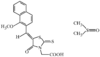
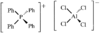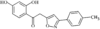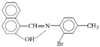issue contents
August 1998 issue
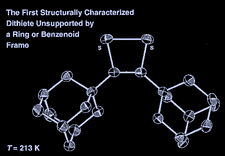
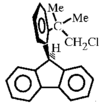
Cover illustration: 3,4-Bis(1-adamantyl)-1,2-dithiete: the First Structurally Characterized Dithiete Unsupported by a Ring or Benzenoid Frame, see Donahue & Holm, pages 1175-1178. One of the two independent molecules in the unit cell is shown with displacement ellipsoids plotted at the 50% probability level and H atoms omitted for clarity.
cif-access (inorganic compounds)
Download citation


Download citation


The crystal structure of the sanidine feldspar analogue potassium iron germanate, quenched from 873 K, has been determined in the C2/m space group using a flux-grown single crystal. A partial ordering of Fe is observed on the two tetrahedral sites, with a preferential occupancy for the T1 site (32%) over the T2 site (18%). This ordering is also reflected by the difference in the size of the tetrahedral sites [mean <T1-O> 1.765 (2) Å versus <T2-O> 1.749 (2) Å].
cif-access (metal-organic compounds)
Download citation


Download citation


The structure of bis(1,2-ethanediammonium) dichloride tetrachloromercurate, originally determined at 153 K [Wen et al. (1992). Inorg. Chim. Acta, 193, 77-85], has been redetermined with room-temperature data carefully corrected for absorption effects.
Download citation


Download citation


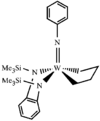
This compound was isolated as a deactivation product from olefin metathesis polymerization and its crystal structure was determined. The W atom has a square-pyramidal coordination geometry with the phenylimido ligand occupying the apical position. A relatively short W-N(imido) bond, 1.736 (3) Å, is interpreted as arising from the interaction of the lone pair of N(imido) and the W metal center.
Download citation


Download citation


This complex is isostructural with the analogous nickel complex but has slightly longer metal-oxygen bonds. The crystal is built up from infinite one-dimensional chains of hydrogen-bonded molecules.
Download citation


Download citation


The structure of [Co(C18H20N4O4)]ClO4.H2O is isomorphous with that of the corresponding tetrafluoroborate monohydrate [Mandel & Douglas (1989). Inorg. Chim. Acta, 155, 55-69]. The cobalt(III) atom has distorted octahedral coordination with trans carboxylato groups.
Download citation


Download citation


The crystal structure of tetraphenylphosphonium tetrachloroaluminate was determined at 150 K. Bond lengths and angles are normal.
Download citation


Download citation


The structure of bis(di-n-propyldithiocarbamato-S,S')nickel(II) has been reported previously in the rhombohedral space group R-3. The new triclinic polymorph shows similar structural features with a square-planar environment for the Ni atoms.
cif-access (organic compounds)
Download citation


Download citation


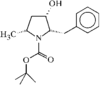
The pyrrolidine ring has an envelope conformation. The crystal packing is stabilized by an intermolecular hydrogen bond.
Download citation


Download citation


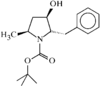
The pyrrolidine ring has a conformation intermediate between a twist and an envelope. The crystal packing is stabilized by an intermolecular hydrogen bond.
Download citation


Download citation


3C4H14N22+.P6O186−.3H2O contains tetragonal channels parallel to the a direction of the unit cell built up by four rows of P6O18 rings and completed by organic groups. The condensed phosphoric groups are centrosymmetric, while the three independent organic cations have no internal symmetry.
Download citation


Download citation


C4H12N22+.H2P2O72− has a typical layer structure composed of organic and inorganic groups centred on planes parallel to (100). The formula unit consists of one acidic anion and two crystallographically independent organic half cations. The H2P2O72− anions have no internal symmetry whereas the piperazinium cations are centrosymmetric.
inorganic compounds
Download citation


Download citation


The preparation of single crystals of the layered perovskite K2Nd2Ti3O10 and the characterization of its structure are described.
Download citation


Download citation


Na3Mn4Te2O12 has a superstructure of the CaFe2O4 structure type. A tripling of the b axis arises because of significant differences in the oxidation states of tellurium and manganese within their respective octahedra.
Download citation


Download citation


Ca9Nd(PO4)5(SiO4)F1.5O0.25 belongs to the britholite family and is isostructural with apatite. The neodymium ions are distributed over the two calcium sites but are mainly localized in calcium site II.
Download citation


Download citation


The structure of the mixed zinc cobalt diphosphate ZnCoP2O7 presents a low-dimensional system not previously observed in the diphosphates of 3d transition elements.
Download citation


Download citation


The crystal structure of pentalead molybdenum octaoxide is a 4:3:2 superstructure of tetragonal α-PbO (litharge).
Download citation


Download citation


Neutron powder diffraction studies confirm the results of a previous X-ray single-crystal study of the protonated compound. The water molecules form hydrogen bonds of different strengths. Deviation from a random distribution of the H atoms (H/D enrichment effect) has been established.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


addenda and errata
Download citation


Download citation


Free 

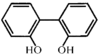
A re-examination of the structure of benzene-1,3,5-triacetic acid reported by Fitzgerald & Gerkin [Acta Cryst. (1997), C53, 967-969].
Free 

A response to the comment by Batten [Acta Cryst. (1998), C54, 1193].


 journal menu
journal menu