issue contents
September 2002 issue
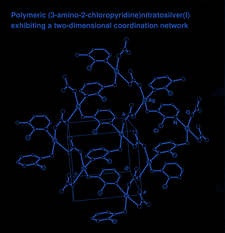
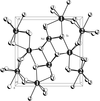
Cover illustration: A view of the two-dimensional coordination layer in (3-amino-2-chloropyridine)nitratosilver(I). See Tong, Chen & Ng [Acta Cryst. (2002), C58, m481-m482].
inorganic compounds
Download citation


Download citation


The MVTeO5 crystal structure consists of [VTeO5]n ribbons linked together by the M+ cations located in a distorted bicapped trigonal prism.
Download citation


Download citation


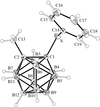
Each of the four title structures contains discrete [Cl3P=N=PCl3]+ cations and metal halide anions. The [NbCl6] and [TaCl6] compounds are isostructural.
Download citation


Download citation


Cs[Ag4Zn2(SCN)9] forms a continuous structure, where the Ag atoms and the thiocyanate S atoms form chains which run in the ac plane. These chains are bonded together through the Cs and Zn atoms. The structure also contains a disordered thiocyanate group.
Download citation


Download citation


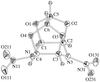
Na10[(UO2)(SO4)4](SO4)2·3H2O contains an unusual uranyl sulfate cluster that involves a chelating sulfate group in a pentagonal-bipyramidal coordination around the U atom.
Download citation


Download citation


Nd2Te3 crystallizes in the U2S3 structure type, with each of the two non-equivalent Nd atoms surrounded by seven Te atoms in polyhedra best described as a seven-octahedron and a monocapped trigonal prism.
Download citation


Download citation


The reported 2212-type superconductor provides proof of the chemical flexibility of layered cuprates. The data also reveal a remarkably large apical Cu—Cu distance, due to the presence of Tl and Sr in the intermediate layer.
Download citation


Download citation


The crystal structures of tetragonal [Co(H2O)6](H2PO2)2 and [Co0.5Ni0.05(H2O)6](H2PO2)2 have been determined. The crystals of hexaaquacobalt(II)/nickel(II) bis(hypophosphite) are a solid solution of CoII and NiIII hypophosphites. The packing of the Co(Ni) and P atoms is the same as in the structure of CaF2. The coordination number of six for metal is achieved by water molecules. The hypophosphite anion does not coordinate to the metal cation.
Download citation


Download citation


(PbCO3)2·BaF2 crystallizes in the ordered hydrocerussite-type structure, with a stacking sequence of [c(h)2]3 for the close-packed metal atoms.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu